Serum testosterone concentration is closely associated with the disease outcome of COVID-19 in male and female hamsters
Firstly, 8-week male and female hamsters were intranasally inoculated with 1 × 104 plaque forming unit (PFU) of SARS-CoV-2 prototype, Beta, Delta and XBB.1.9.2.1 variants, respectively. Generally, the male hamsters showed higher mortality, more body weight loss and higher viral RNA load in lung tissues than the female hamsters from 0 to 9 days post infection (dpi) of all the tested SARS-CoV-2 strains (Supplementary Fig. S1). The recovered body weight of the surviving hamsters at 9 dpi indicated an alleviation of acute COVID-19 (Supplementary Fig. S1). In a parallel experiment, six male and female hamsters were intranasally inoculated with 1 × 104 PFU of prototype SARS-CoV-2, and were euthanized at 0 dpi, 3 dpi, 5 dpi, 7 dpi, and 9 dpi, respectively. Serum TET concentration, body weight loss and viral RNA levels in lung tissues were detected at each time point (Fig. 1a). In the male hamsters, approximately 2- to 3-fold increase of serum TET concentration were detected from 0 to 9 dpi, which was negatively related to the percentage of body weight loss (Fig. 1b) and was positively related to the viral RNA levels in lung tissues (Fig. 1c). Although the female hamsters showed approximately 10-fold lower baseline of serum TET concentration than the male hamsters at 0 dpi, a significant increase of serum TET concentration was detectable from 0 to 9 dpi (Fig. 1d). Contrary to male hamsters, serum TET concentration was positively related to the percentage of body weight loss (Fig. 1e), and was negatively related to the viral RNA levels in lung tissues (Fig. 1f) in the female hamsters. Similar to the prototype SARS-CoV-2, the relationship between serum TET concentration and key disease parameters was also observed in the male and female hamsters that infected with the Beta, Delta, and XBB.1.9.2.1 variants, respectively (Supplementary Fig. S2).
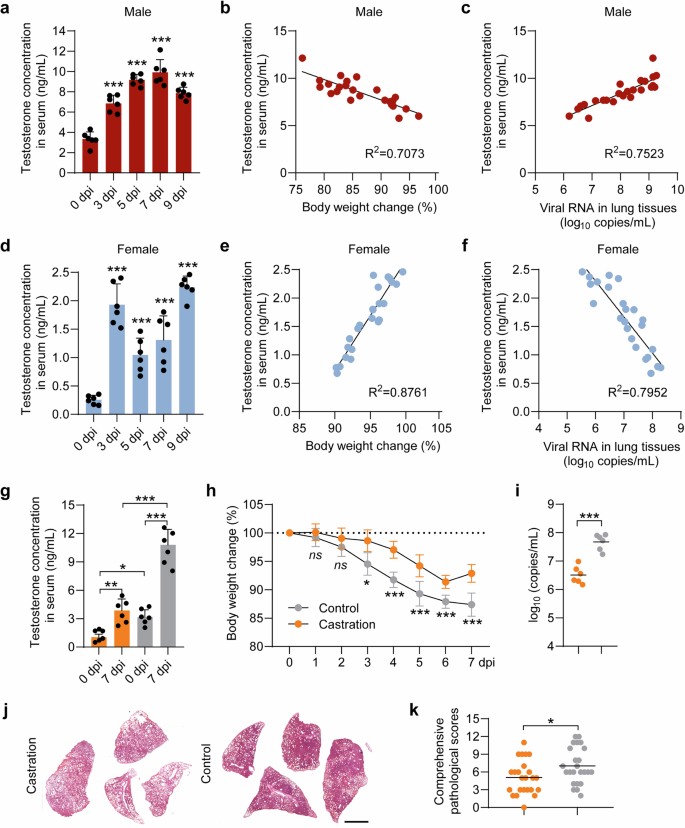
Serum TET concentration is closely associated with the disease outcome of SARS-CoV-2 infection in hamsters. Male and female hamsters were intranasally inoculated with 1 × 104 PFU of prototype SARS-CoV-2. For sample collection, hamsters were euthanized at 0, 3, 5, 7, and 9 dpi, respectively. TET concentration in the serum samples of (a) male and (b) female hamsters was measured by ELISA (n = 6). Percentage of body weight changes at indicated time points were recorded and viral RNA levels in lung tissues were measured by RT-PCR (n = 6). The linear relationship between testosterone concentration in the serum and (b, e) percentage of body weight changes, and (c, f) viral RNA levels in lung tissues was shown. g TET concentration in the serum samples of castrated hamsters and intact controls was measured by ELISA (n = 6). Castrated and intact male hamsters (controls) were intranasally inoculated with 1 × 104 PFU of prototype SARS-CoV-2. h Percentage of body weight changes were recorded from 0 to 7 dpi (n = 6). i Viral RNA levels in tissue homogenates of lungs collected from prototype SARS-CoV-2 infected hamsters were measured by RT-PCR (n = 6). j Representative H&E staining images of lung tissues collected from castrated hamsters and controls sacrificed at 7 dpi were presented (Bar = 2 mm). Lung images for all of the hamsters were shown in Fig. S4. k Comprehensive pathological scores for lung sections were determined based on the severity and percentage of injured areas of each lung lobe. For each group, 24 lung lobes were collected from six individual hamsters and were scored. Lung pathological scores for all of the hamsters were shown in Table S1. One-way ANOVA followed by Dunnett’s multiple-comparisons test comparing each time point to baseline (0 dpi) for (a, d); simple linear regression for (b, c, e, f); two-way ANOVA with Bonferroni’s post hoc test for planned pairwise comparisons (g) and for between-group comparisons at individual time points across 1–7 dpi for (h); and unpaired two-tailed Student’s t-test for (i, k). P-values *P **P ***P
To confirm the causality of male sex bias, serum TET concentration and disease severity in male hamsters of acute COVID-19, six male hamsters were castrated and intranasally inoculated with 1 × 104 PFU of prototype SARS-CoV-2, Beta, Delta and XBB.1.9.2.1 variants, respectively. Compared to the intact controls, the castrated hamsters showed approximately 3-fold lower serum TET concentration (Fig. 1g) and rescued body weight loss (Fig. 1h). Furthermore, we anatomized all of the castrated hamsters and intact controls at 7 dpi and obtained their lungs for analysis of viral load and lung injury. The castrated hamsters showed over 10-fold decrease of viral RNA levels in lung tissues than the intact controls (Fig. 1i). Alleviated disease severity was also observed in the castrated hamsters that intranasally inoculated with 1 × 104 PFU of Beta, Delta, and XBB.1.9.2.1 variants of SARS-CoV-2, respectively (Supplementary Fig. S3). The results of hematoxylin and eosin (H&E) and Masson Trichrome staining for the lung lobes collected from intact controls showed typical features of severe pneumonia, including pulmonary consolidation and alveolar destruction, diffusive inflammation, protein-rich fluid exudate, hyaline membrane formation and severe pulmonary hemorrhage (Fig. 1j and Supplementary Figs. S4 and S5). In contrast, critical lung injury was attenuated in the castrated hamsters (Fig. 1j and Supplementary Figs. S4 and S5). The severity of lung pathogenesis was quantified by comprehensive pathological score based on alveolar septum thickening and consolidation, hemorrhage, exudation, pulmonary edema, and mucous, recruitment and infiltration of inflammatory cells among all the hamster lung lobes. As expected, the castrated hamsters showed a lower lung pathological score than intact controls (Fig. 1k and Supplementary Table S1). In addition, low serum TET concentration is associated with Long COVID at 6 weeks post infection of prototype SARS-CoV-2 in both male and female hamsters, which indicating the multifaceted nature of TET in different course of COVID-19 (Supplementary Fig. S6). Overall, the above results revealed that serum TET concentration is closely associated with diseases outcome of COVID-19 in male and female hamsters.
The disease outcome of extrinsic TET treatment is dose-dependent in male and female hamsters
As castration was able to decrease serum TET concentration and alleviate the severity of COVID-19 in male hamsters, we further investigated the disease outcomes of SARS-CoV-2-infected male and female hamsters with extrinsic TET treatment (Fig. 2a). The prototype SARS-CoV-2-infected hamsters were daily treated with different doses of TET (high dose: TET-H, 1 mg/kg; middle dose: TET-M, 0.2 mg/kg; low dose: TET-L, 0.04 mg/kg per dose) via intraperitoneal injection from 0 to 5 dpi. The prototype SARS-CoV-2-infected hamster without extrinsic TET treatment were set as control group. Physical and health examinations had been undertaken for 7 days by recording their survival rate and percentage of body weight changes. In the male hamsters, SARS-CoV-2 infection caused one death out of 16 hamsters, whereas, two, five and seven out of 16 hamsters deceased in the TET-L, TET-M and TET-H groups respectively (Fig. 2b). The control male hamsters and those in TET-L, TET-M and TET-H groups exhibited the mean body weight loss of 11.2 ± 2.3%, 16.6 ± 2.5%, 21.3 ± 1.7% and 23.4 ± 2.3% at 7 dpi, respectively (Fig. 2c). In the female hamsters, only high dose of extrinsic TET treatment (TET-H) killed three out of 16 hamsters (Fig. 2d). The control female hamsters and those in TET-L, TET-M and TET-H groups exhibited the mean body weight loss of 5.9 ± 3.0%, 3.5 ± 2.4%, 8.7 ± 1.8% and 11.8 ± 2.6% at 7 dpi, respectively (Fig. 2e). To evaluate the severity of lung injury and viral load in respiratory tract organs, all the survived hamsters were euthanized at 7 dpi. The results of H&E and Masson Trichrome staining for lung lobes and comprehensive pathological score of the male hamsters showed that extrinsic TET treatment caused a dose-dependent deterioration of lung pathological changes after SARS-CoV-2 infection (Fig. 2f, g, Supplementary Figs. S7–8 and Supplementary Table S2). In the female hamsters, low dose of extrinsic TET treatment relieved lung pathological changes, whereas middle and high doses of extrinsic TET treatment resulted in more severe lung injury (Fig. 2f, g, Supplementary Figs. S9–10 and Supplementary Table S3).
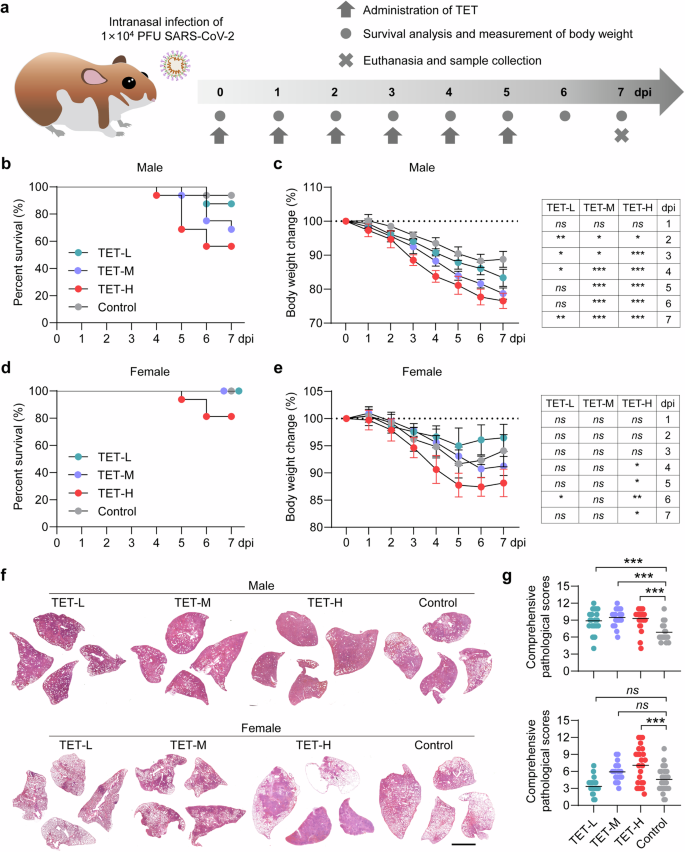
TET treatment affects the disease severity of SARS-CoV-2 infection in male and female hamsters. a Schematic diagram of SARS-CoV-2 infection and animal operations. Male and female hamsters were intranasally inoculated with 1 × 104 PFU of prototype SARS-CoV-2, and then received different doses of intraperitoneal injection of testosterone (TET) from 0 to 5 dpi (once per day; TET-H, 1 mg/kg per dose; TET-M, 0.2 mg/kg per dose; TET-L, 0.04 mg/kg per dose). Age-paired male and female SARS-CoV-2-infected hamsters without treatment were set as control groups. b, d Survival rate (n = 16) and c, e percentage of body weight changes (n = 6) were daily recorded from 0 to 7 dpi. The survived hamsters were euthanized at 7 dpi for pathological, virological, and immunological analysis. f Representative H&E staining images of lung tissues collected from hamsters sacrificed at 7 dpi were presented (Bar = 2 mm). Lung images for all of the hamsters were shown in Figs. S7–10. g Comprehensive pathological scores for lung sections were determined based on the severity and percentage of injured areas of each lung lobe. For each group, 24 lung lobes were collected from six individual hamsters and were scored. Lung pathological scores for all of the hamsters were shown in Tables S2 and S3. Two-way ANOVA with Dunnett’s post hoc test for between-group comparisons at individual time points across 1–7 dpi for (c, e); one-way ANOVA followed by Dunnett’s multiple-comparisons test for (g). P-values *P **P ***P
Excessive release of inflammatory cytokines, abnormal perturbation of antiviral innate immune response and high viral load in respiratory tract organs are considered as typical manifestations of critical COVID-19 in both human patients and animal models. Therefore, we evaluated the mRNA levels of inflammatory cytokines, including interleukin-6 (IL-6), interferon gamma (IFN-γ), and tumor necrosis factor alpha (TNF-α), as well as interferon alpha (IFN-α), interferon-stimulated gene 15 (ISG15), and myxovirus resistance 1 (MX1) in lung tissues collected at 7 dpi. In the male hamsters, TET treatment increased the mRNA levels of IFN-γ, IL-6 and TNF-α (Fig. 3a–c). Although the mRNA levels of IFN-α were not significantly changed, middle and high doses of TET treatment down-regulated the mRNA levels of ISG15 and MX1 (Fig. 3d–f). In the female hamsters, low dose of TET treatment decreased the mRNA levels of IL-6, IFN-γ, and TNF-α, whereas high dose TET treatment showed an opposite effect on these inflammatory cytokines (Fig. 3g–i). Furthermore, the female hamsters with high dose TET treatment showed a declined up-regulation of IFN-α, ISG15, and MX1 mRNA levels than those with low dose TET treatment (Fig. 3j–l).
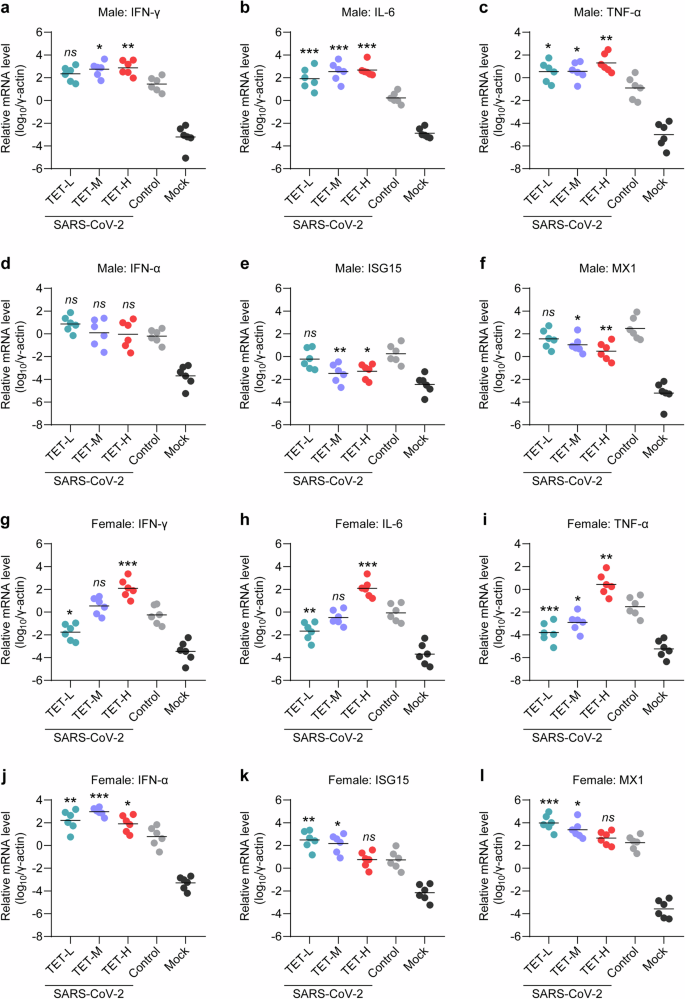
Fold changes of inflammation and innate immune response-associated genes in lung tissues of SARS-CoV-2-infected male and female hamsters after TET treatment. The mRNA levels of inflammatory cytokines include a IFN-γ, b IL-6, and c TNF-α in the lung tissues that collected from the male hamsters at 7 dpi and g–i those collected from the female hamsters at 7 dpi were measured by RT-PCR to determine the fold changes (n = 6). The mRNA levels of (d) IFN-α and representative ISGs include (e) ISG15 and (f) MX1 in the lung tissues collected from the male hamsters at 7 dpi and (j-l) those collected from the female hamsters at 7 dpi were also measured by RT-PCR to determine the fold changes (n = 6). These mRNA levels were normalized to the housekeeping gene γ-actin. One-way ANOVA followed by Dunnett’s multiple-comparisons test was used to compare each TET group with the control group. P-values *P **P ***P
Afterwards, we asked whether extrinsic TET treatment could affect viral load. To that end, we analyzed viral mRNA levels in respiratory tract organs, including turbinate, trachea, and lung by RT-PCR that amplifies SARS-CoV-2 open reading frame 1ab (ORF1ab) for detection of viral RNA load in the homogenized tissues collected at 7 dpi. Compared to the control group, extrinsic TET treatment caused a dose-dependent increase of viral RNA levels in turbinate, trachea and lung of the male hamsters, respectively (Fig. 4a–c, left panel). The male hamsters in TET-H group showed 5- to 10-fold increase of viral RNA levels than the control group in all of the three detected organs. The female hamsters in TET-L and TET-M groups showed no significant change or decreased viral RNA levels in these respiratory tract organs than the control group (Fig. 4a–c, right panel). Whereas the female hamsters in TET-H group showed over 5-fold increase of viral RNA levels. In addition, the titers of live virus particles in the respiratory tract tissue samples showed a similar trend with the results of viral RNA levels (Fig. 4d–f). The results of immunohistochemistry staining for SARS-CoV-2 nucleocapsid protein in hamster lung tissues confirmed that TET treatment improves replication of viral load (Supplementary Fig. S11). In the primary human alveolar epithelial cell model, TET treatment enhanced the expression of SARS-CoV-2 receptor ACE2 and co-receptor TMRPSS2, which was consistent with the RT-PCR results of hamster lung tissues (Supplementary Fig. S11). These data suggested that TET deteriorates the severity of COVID-19 in the hamster model by arguments of pulmonary viral load. Similar results were also found in human lung epithelial cells, lung organoids, and mouse models.18,20,22
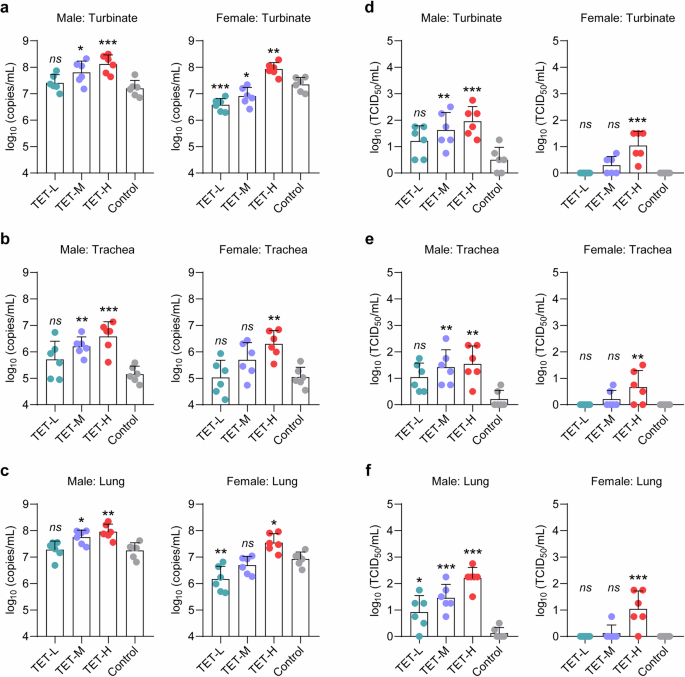
Viral load in respiratory tract organs of SARS-CoV-2-infected male and female hamsters after TET treatment. Viral RNA levels in respiratory tract organ tissues, including a turbinate, b trachea, and c lung of SARS-CoV-2 infected male and female hamsters with TET treatment were collected from at 7 dpi and measured by RT-PCR (n = 6). The primers were used to amplify SARS-CoV-2 ORF1ab gene. d–f Viral titers of these samples were detected by a titration method of gradient dilution and cytopathic effect observation (n = 6). One-way ANOVA followed by Dunnett’s multiple-comparisons test was used to compare each TET group with the control group. P-values *P **P ***P
In addition, we also detected the concentrations for TET, DHT, and E2 in the serum samples collected at 7 dpi, respectively. The hamsters without SARS-CoV-2 infection or extrinsic TET treatment were set as mock group. In both of the male and female hamsters, extrinsic TET treatment corresponded to a dose-dependent increase of TET and DHT levels and a decrease of E2 levels (Fig. 5d–f). Remarkably, the female hamsters with moderate COVID-19 (control and TET-L groups) showed lower TET/DHT levels and/or higher E2 levels than those with middle to critical COVID-19. Taken together, extrinsic TET treatment usually causes deteriorating disease outcome in male hamsters with SARS-CoV-2 infection. Low dose extrinsic TET treatment is beneficial to the female hamsters, however, middle to high doses is harmful.
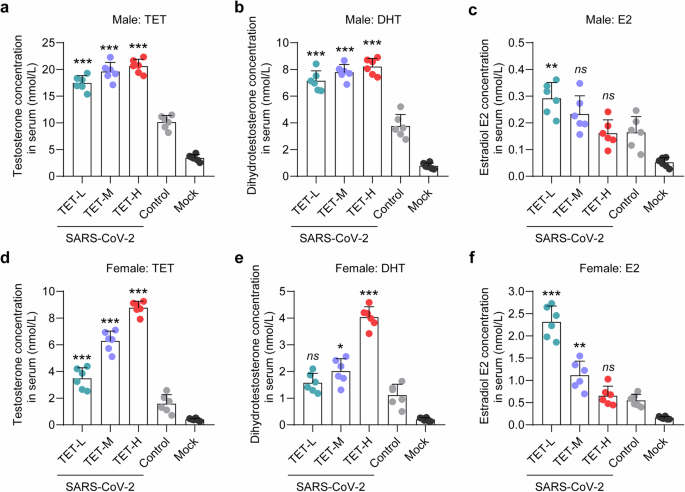
Sex hormone concentration in serum of SARS-CoV-2-infected male and female hamsters after TET treatment. The concentrations of (a, d) TET, (b, e) DHT and (c, f) E2 in the serum samples collected at 7 dpi were measured by ELISA, respectively (n = 6). One-way ANOVA followed by Dunnett’s multiple-comparisons test was used to compare each TET group with the control group. P-values *P **P ***P
Pulmonary transcriptome analysis reveals key clues for the pathological mechanisms of TET-associated disease enhancement in hamster model
Furthermore, we performed bulk and single-cell RNA sequencing to delineates the pathological signatures of TET treatment in male hamsters. Lung tissues were collected from mock and prototype SARS-CoV-2-infected male hamsters with or without 6-dose TET-H treatment were collected at 7 dpi, respectively. Principal component analysis (PCA) showed highly similar transcriptional profiles between the mock and mock-TET groups, whereas clear separation was observed between the COVID-19 and COVID-19-TET groups, indicating a TET-dependent transcriptional divergence pattern (Supplementary Fig. S12a). KEGG pathway enrichment revealed distinct functional alterations across groups. Prior to infection, calcium signaling was reduced in the mock-TET group compared with the mock group. Notably, the COVID-19-TET group displayed markedly elevated pathogenic signaling, including the IL-17, HIF-1α, and apoptosis, suggesting that TET might enhance cytokine storm priming and cell death (Supplementary Fig. S12b). To further investigate the alteration at single-cell resolution, we performed scRNA-seq and identified 17 cell subsets based on canonical marker genes (Supplementary Fig. S12c, d). Cell-type compositions differed substantially among the four groups (Supplementary Fig. S12e). In the mock and mock-TET groups, frequencies of the ciliated epithelial cells, monocytes, alveolar macrophages, and B cells were reduced by TET treatment, and these populations declined even further after infection (Supplementary Fig. S12f). In contrast, myeloid dendritic cells (mDCs), plasmacytoid dendritic cells (pDCs), and interstitial macrophages (IMs) exhibited greater post-infection expansion in the COVID-19-TET group (Supplementary Fig. S12f), indicating a TET-driven reshaping of the pulmonary immune landscape in acute COVID-19. Consistent with the results of animal experiments, pulmonary RNA-Seq analysis confirmed that TET exacerbates inflammation and reprograms immune cell dynamics during SARS-CoV-2 infection, provided key clues for the pathological mechanisms of TET-associated disease enhancement. Inhalation delivery of cocktail RNA interference (RNAi) against IL-17 and HIF-1α signaling reduced death, body weight loss, and severe pneumonia in prototype SARS-CoV-2-infected male and female hamsters with or without TET treatment (Supplementary Fig. S13). These validation experiments suggested that TET-associated disease enhancement is reversible.
FINA therapy ameliorated critical COVID-19 in male hamsters
Considering the disease outcomes of SARS-CoV-2 male hamsters with castration or extrinsic TET treatment, we further questioned whether the pharmacological target of TET could counteract the severity of COVID-19. FINA is a widely used TET inhibitor and has been approved by the FDA for the treatment of diseases that are associated with abnormally elevated TET. Therefore, male hamsters were intranasally inoculated with 1 × 104 PFU of prototype SARS-CoV-2 and received sequential FINA therapy. The SARS-CoV-2-infected hamsters were orally administrated with 6-doses of FINA (once per day, 1 mg/kg per dose) from 0 to 5 dpi, or 3-doses of FINA from 3 to 5 dpi (Fig. 6a). The SARS-CoV-2-infected hamsters without FINA therapy were set as controls. Physical and health examinations were undertaken for 7 days to record percentage of body weight changes. All the SARS-CoV-2-infected hamsters with or without FINA therapy survived at 7 dpi. The control hamsters exhibited progressive mean body weight loss of up to 12.1 ± 3.7% from 0 to 7 dpi (Fig. 6b). Whereas, the hamsters with 3-doses and 6-doses of FINA therapy exhibited body weight loss of 8.1 ± 1.5% and 3.4 ± 1.7% at 7 dpi (Fig. 6b).
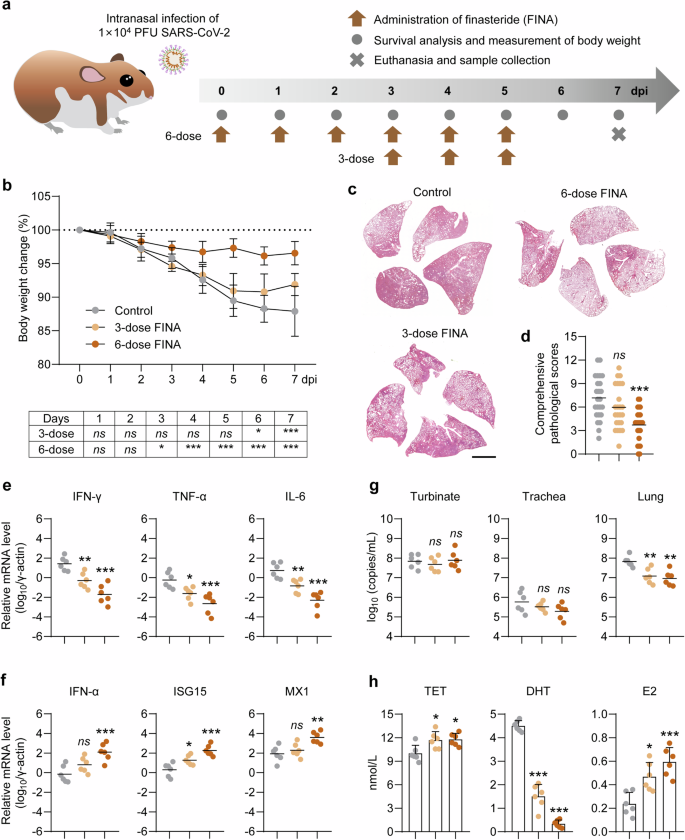
Evaluation for the therapeutic effect of FINA in the male hamsters with SARS-CoV-2 infection. a Schematic diagram of SARS-CoV-2 infection and animal operations. Male hamsters were intranasally inoculated with 1×104 PFU of prototype SARS-CoV-2, and then received 6-dose oral administration of finasteride (FINA) from 0 to 5 dpi, or 3-dose FINA from 3 to 5 dpi. The infected hamsters without FINA therapy were set as controls. b Percentage of body weight changes of the hamsters were recorded from 0 to 7 dpi (n = 6). The survived animals were euthanized at 7 dpi for pathological analysis. c Representative H&E staining images of lung tissues collected from hamsters sacrificed at 7 dpi were presented (Bar = 2 mm). Lung images for all of the hamsters were shown in Figs. S14 and 15. d Comprehensive pathological scores for lung sections were determined based on the severity and percentage of injured areas of each lung lobe. For each group, 24 lung lobes were collected from six individual hamsters and were scored. Lung pathological scores for all of the hamsters were shown in Table S4. Fold changes for the mRNA levels of e inflammatory cytokines, f IFN-α and ISGs in lung tissues were detected by RT-PCR (n = 6). These mRNA levels were normalized to the housekeeping gene γ-actin. g Viral RNA levels in tissue homogenates of turbinate, trachea, and lung collected at 7 dpi were measured by RT-PCR (n = 6). h The concentrations of TET, DHT and E2 in the serum samples collected at 7 dpi were measured by ELISA (n = 6). Two-way ANOVA with Dunnett’s post hoc test for between-group comparisons at individual time points across 1–7 dpi for (b). One-way ANOVA followed by Dunnett’s multiple-comparisons test (each finasteride dose vs Control) for (d–h). P-values *P **P ***P
To evaluate the severity of lung injury and viral load in respiratory tract organs, all of the hamsters were euthanized at 7 dpi. The results of H&E and Masson Trichrome staining of lung lobes and comprehensive pathological scores showed that SARS-CoV-2-induced lung pathological changes were largely suppressed by 6-dose FINA therapy (Fig. 6c, d, Supplementary Figs. S14, 15 and Supplementary Table S4). Partial relief of lung injury was observed in the male hamsters with 3-dose FINA therapy. In a parallel experiment, reduced dosage of FINA (0.2 or 0.04 mg/kg per dose) cannot fully protect against the body weight loss and lung injury caused by prototype SARS-CoV-2 infection (Supplementary Fig. S16). Of note, FINA therapy resulted in a dose-dependent down-regulation of IL-6, IFN-γ, and TNF-α (Fig. 6e), as well as up-regulation of IFN-α, ISG15, and MX1 (Fig. 6f) mRNA levels in lung tissues. We further analyzed viral replication in respiratory tract organs, including turbinate, trachea, and lungs by RT-PCR that amplifies SARS-CoV-2 ORF1ab for the detection of viral RNA loads in the homogenized tissues collected at 7 dpi. In contrast of the hamsters without therapy, lower levels of viral RNA in turbinate, trachea, and lung were detected in the hamsters with 3-doses and 6-doses FINA therapy, but only the decreases in lungs were statistically significant (Fig. 6g). Although FINA therapy slightly increased serum TET levels, remarkable decreases of serum DHT levels were coupled with increase of E2 levels at 7 dpi (Fig. 6h). In addition, 6-doses FINA therapy (1 mg/kg per dose) can rescue male hamsters from lethal COVID-19 caused by the highly pathogenic Beta, Delta and XBB.1.9.2.1 variants of SARS-CoV-2 (Supplementary Figs. S17 and 18), suggesting that host-target therapy can largely overcome the challenge of rapid viral mutation and immune escape. Overall, these data demonstrated the potent and broad-spectrum therapeutic efficacy of FINA in male hamsters with SARS-CoV-2 infection.
Pulmonary transcriptome analysis of FINA therapy reveals key clues for the mechanisms of FINA therapy in hamster model
To investigate the underlying therapeutic mechanisms of FINA, we performed bulk and single-cell RNA sequencing on lung tissues of male hamsters with or without 6-dose FINA therapy (COVID-19 and FINA groups). Age-paired mock male hamsters without SARS-CoV-2 infection were set as health controls. PCA showed that samples under each group were well separated and the FINA group was closer to the mock group (Fig. 7a). SARS-CoV-2 infection resulted in down-regulation of 3090 genes and up-regulation of 3087 genes, however, 2127 and 2462 of them were reserved by FINA therapy, respectively (Fig. 7b). The results of Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Set Enrichment Analysis (GSEA) revealed that signaling pathways include neutrophil extracellular trap (NET) formation, p53, IL-17 and HIF-1 were up-regulated in the COVID-19 group and down-regulated by FINA therapy, whereas Wnt, Rap1 and calcium signaling pathways etc. were down-regulated in the COVID-19 group and up-regulated by FINA therapy (Fig. 7c). In addition, the regulatory network and heat map of differentially expressed genes (DEGs) in five representative biological process and signaling pathways were displayed (Fig. 7d and Supplementary Fig. S19).
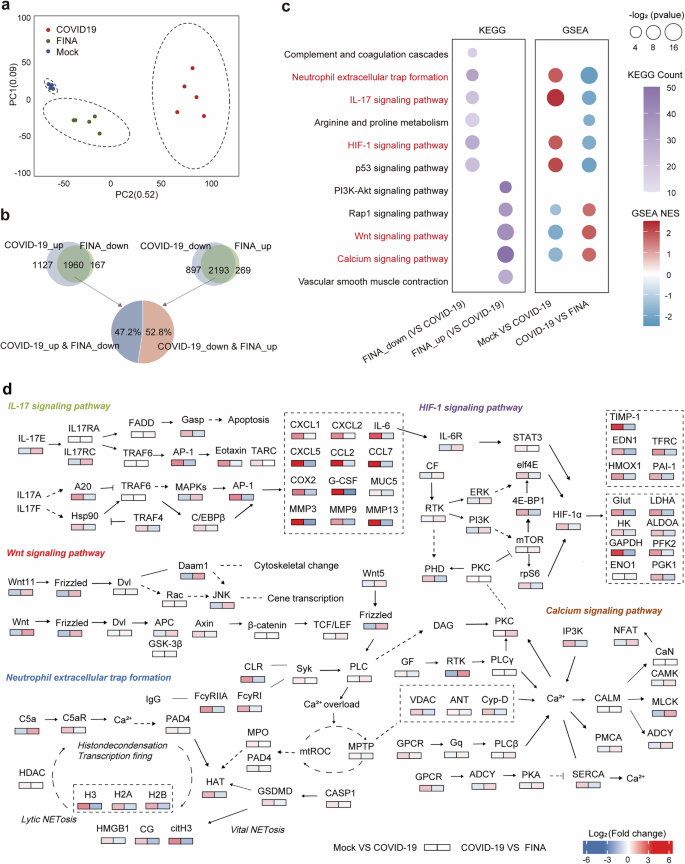
Pulmonary transcriptome analysis of FINA therapy in male hamsters with SARS-CoV-2 infection. a Principal component analysis (PCA) for the transcriptome analysis of lung tissues. Health controls, SARS-CoV-2 infected hamsters with or without FINA therapy were colored in blue (Mock), red (COVID-19) and green (FINA), respectively (n = 5). b Number of DEGs in comparisons between the mock and COVID-19 groups, and between the COVID-19 and FINA groups. The pie chart displays the percentage of significantly up-regulated (in red) or down-regulated (in blue) genes in the overlapping DEGs. c Enrichment analysis based on KEGG database. The first column shows the up-regulated and down-regulated pathways that were significantly altered in overlapping DEGs based on enriched KEGG pathways, with the color intensity on behalf of the counts of genes. The second column shows the results of the GSEA. Pathways in which gene expression was increased or decreased are shown in red and blue, respectively. The color intensity is proportional to the NES calculated using the GSEA. d The diagram shows the changes and interactions of genes in the key signaling pathways after SARS-CoV-2 infection and FINA therapy. The intensity of the colors indicates average changes in genetic levels. In two columns, dot size is used to presented p value of the pathways, with only p value
Afterwards, the protein-protein interaction (PPI) network was performed to explore interactions between proteins regulated by the core genes. Molecular complex detection (MCODE) component identified 20 genes enriched in the Wnt and calcium signaling pathways within the network (Supplementary Fig. S20a). Ten hub genes were subsequently defined by overlapping six cytoHubba algorithms (Supplementary Fig. S20b, c), six of which were confirmed by scRNA-seq to be reversed by FINA treatment (Supplementary Fig. S20d). Notably, these hub genes, especially Fzd2, Wnt2, and Ntrk3, are predominantly expressed in fibroblasts, with Fzd2 additionally showing high expression in epithelial cells, indicating that FINA may counteract disease progression by modulating proliferation and growth programs primarily in fibrotic and epithelial compartments (Supplementary Fig. S20e).
Next, single-cell RNA sequencing was further performed to determine how FINA modulates cell fate alterations in response to SARS-CoV-2 infection. FINA treatment reversed the SARS-CoV-2-induced decrease in alveolar macrophages and monocytes, as well as the increase in IMs and dendritic cell populations (Supplementary Fig. S21a). At the level of cell-cell communication, SARS-CoV-2 infection primarily reshaped macrophage-centered interaction networks, particularly those involving IMs and their crosstalk with other pulmonary cell types (Supplementary Fig. S21b). These alterations were largely reversed by FINA treatment (Supplementary Fig. S21b). These findings suggest that FINA may counteract disease progression by modulating macrophage fate and their interactions with other cell types, prompting further investigation into the functional heterogeneity of macrophage subpopulations. Enrichment analysis of marker genes from the three macrophage subclusters revealed that IMs were uniquely characterized by prominent activation of inflammatory pathways, including the IL-17 signaling pathway and Chemokine signaling pathway, in contrast to the other subsets (Supplementary Fig. S21c). Collectively, these findings demonstrate that FINA alleviates SARS-CoV-2-induced lung injury by reshaping IM fate and dampening macrophage-centered inflammatory signaling. Overall, these findings indicate that FINA may alleviate SARS-CoV-2-induced lung injury mainly through reprogramming macrophage fate and attenuating inflammatory signaling associated with IMs.






