Characteristics of the study population
We enrolled 353 individuals living in 83 households in the study from February 2021 through January 2022, with an average household size of 4.3 individuals (range (1–16), IQR (3–5)). Descriptive sociodemographic characteristics of the individuals included in the study are found in Supplementary Table S1 that presents in column 1, all survey participants, and in column 2, only those who had 10 or more follow-up visits. These data demonstrate a female-to-male ratio of 0.98, and that most participants were under 30 years old (median age: 28.7). Regarding education, 36.5% of the STR inhabitants had completed secondary school or were currently pursuing it and 5.1% had completed or were currently pursuing a degree. Occupationally, 28.9% of the inhabitants worked in indoor activities and 24.4% in outdoor related activities.
Using the STR census data from 2021 (383 inhabitants) and the number of reported malaria cases (https://datosabiertos.gob.pe/dataset/vigilancia-epidemiol%C3%B3gica-de-malaria) in 2021 (n = 11) and 2022 (n = 35), the APIs were 23 and 91, respectively. For context, the human census and APIs for the other two villages of the San Jose de Lupuna community (San Jose and San Pedro) are included (Supplementary Table S2).
The epidemiological survey conducted in STR (Supplementary Table S1) revealed that the ownership rate of any type of mosquito net (raw cotton, nylon, or insecticide-treated nets) among all participants ranged from 99.7% to 100%. Among the 109 individuals who completed between 10 and 12 follow-up visits, 89% reported always using a mosquito net before sleeping, while the remaining 11% used it on more than half of the occasions. No cases were reported of individuals who never used a mosquito net, and despite the high historical prevalence of malaria (more than 80% of the population has had malaria in their lifetime), only 5.4% of individuals had malaria diagnosed by microscopy in the previous year.
The household characteristics of the study participants who completed the questionnaire (n = 83), are presented in Supplementary Table S3a. Most individuals resided in houses constructed with wooden walls (87%), dirt floors (61%), and tin roofs (99%). Most inhabitants lack access to essential public services, most households lacked access to electricity at home, unless equipped with a power generator that provided a limited supply. The primary sources of drinking and cooking water were springs and river water. Sanitation facilities were inadequate, with many participants relying on pit latrines or other unsanitary practices. The principal fuel source for cooking was firewood (89%).
A bivariate analysis, conducted only on those participants with 10–12 follow ups, for whom PCR P. vivax status was recorded, in Supplementary Table S3b, found that individuals engaged in outdoor-related occupations had significantly higher P. vivax infection rates compared to those with indoor occupations or who were not working (35% vs. 21%, p = 0.012). Adults aged 30–50 and over 50 years were more frequently infected than children under 15 (p = 0.006). Poor housing conditions were also associated with infection: participants living in dwellings with unfinished or wood-based flooring had higher infection rates than those with cement floors (p = 0.004). Likewise, inadequate sanitation facilities were linked to increased prevalence, with 63% of infected individuals residing in homes lacking basic sanitation (p = 0.004).
Rate ratios from a Poisson regression, with variable selection performed using a stepwise procedure based on the Akaike Information, detected several additional factors, independently associated with PCR-detected P. vivax infection (Supplementary Table S3c). Female participants had a lower incidence rate compared to males (IRR = 0.62; 95% CI: 0.39–1.00; p = 0.049). The risk of infection increased with age, particularly among individuals aged 30–50 (IRR = 3.19; 95% CI: 1.66–6.19; p p = 0.004). Outdoor-related occupations (IRR = 2.28; 95% CI: 1.33–3.98; p = 0.003), migration (IRR = 0.31; 95% CI: 0.13–0.76; p = 0.003), and inadequate sanitation (IRR = 0.48; 95% CI: 0.28–0.80; p = 0.006) were also significant. Living in a fully enclosed house strongly reduced infection risk (IRR = 0.02; 95% CI: 0.00–0.15; p p p = 0.091).
Prevalence of malaria infection in the study population
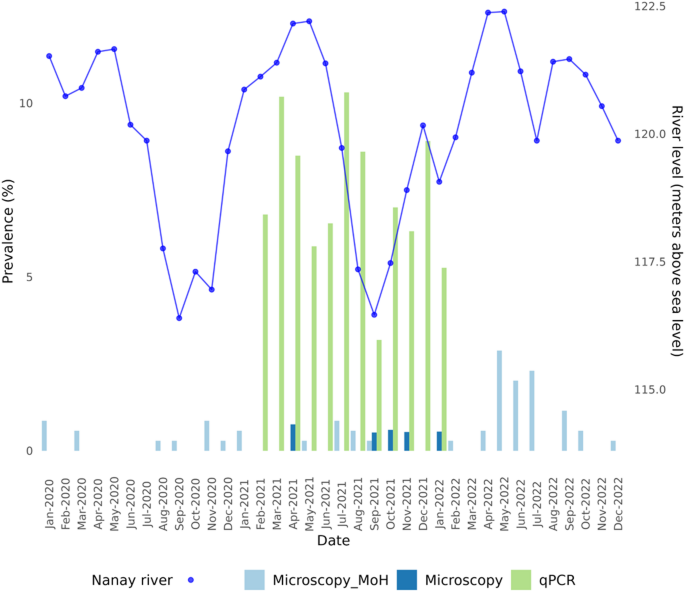
Malaria in STR is seasonal, with cases increasing during the months when the river reaches its peak. Additionally, in the months with the lowest river levels (August-October), there is another, often smaller, increase in cases detected by microscopy (Fig. 3), possibly related to the formation of mosquito breeding sites, i.e., pools of water left behind as the river recedes.
Malaria prevalence diagnosed by microscopy in our surveys was less than 1%, with cases reported in April 2021 and between September 2021 and January 2022. In contrast, the prevalence of qPCR detected cases was 5x higher in the months of microscopy detected malaria infections. Furthermore, malaria infections were detected during every survey, using qPCR (Table 1). The PCR diagnosis was conducted at the conclusion of the 12-month study period only in those individuals with 10 to 12 months of follow-up. A total of 1201 tests in 109 individuals were performed by qPCR, resulting in the detection of 88 Plasmodium vivax infections, two Plasmodium falciparum infections, and no mixed infections. Of these, microscopy detected four P. vivax infections and one P. falciparum infection, yielding a ratio of 17:1 submicroscopic/microscopic malaria infections. In the period from February 2021 to December 2022, 31% (34/109) of the individuals tested had experienced at least one P. vivax infection. Of these, 88% (30/34) were not detectable by microscopy throughout the entire study period. Notably, 32.4% of PCR-positive individuals had more than one positive result over the 12-month follow-up period, indicating potential persistence of low-density infections or repeated exposure to infectious mosquito bites.
The Scott-Knott clustering method was used to evaluate the average number of mosquitoes per cluster across months. Similar to Tukey’s test, this method has the advantage of not producing intermediate or overlapping groups, making it easier to identify distinct groupings. Using a boxplot analysis, results from the Scott-Knott test showed a marked decline in PCR-detected P. vivax infections across all transmission intensity groups starting in June 2021. During the high transmission period (Feb–May 2021), infection counts were notably higher in the high and medium transmission groups, with several extreme values. In contrast, from June to November 2021, the low transmission season, cases were nearly absent across all groups. A moderate resurgence was observed in December 2021 and January 2022, particularly in the medium group (Supplementary Figure S1).
Temporally (by month) Ny. darlingi abundance was compared with PCR-detected P. vivax infections (malaria prevalence) using a Pearson correlation analysis; results were not significant (Pearson’s r = 0.14, p = 0.653) (Supplementary Figure S2).
Malaria cases by microscopy and PCR in Santa Rita, Peru, from 2020–2022. The bright blue line represents the monthly level of the Amazon River at the mouth of the Nanay River. Dark blue histograms represent microscopy results from the present study; light blue are those detected by the Ministry of Health (see text). Molecular diagnostic tests (green histograms) were performed only for participants with 10 to 12 months of follow-up.
Mosquito species composition and molecular identification
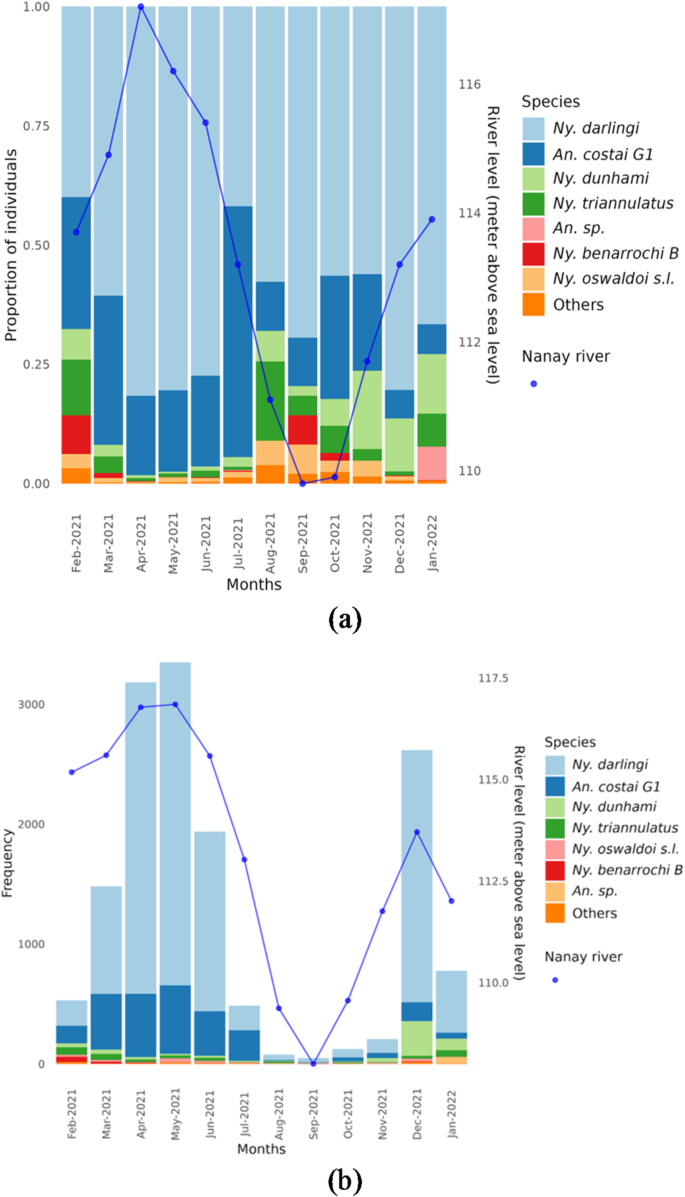
A total of 14,842 female anopheline mosquitoes were collected. Nyssorhynchus darlingi was the most abundant species (10,994 individuals; 74% of the total). During the rainy months (December-June), 9,142 (61.6%) Ny. darlingi were collected and at the initial increase in the river level (December-February) 1,842 (21.1%) Ny. darlingi were collected. April and May exhibited the highest abundance of anophelines, coinciding with the peaks of the Nanay river. The second most abundant species, An. forattinii/near costai (2,583 individuals; 17% of the total), was collected mainly during the rainy season although unexpectedly, during July (dry season), it was the most abundant mosquito species collected (Fig. 4a, b). The results of the COI sequence analysis of a subsample of 25 individual An. forattinii/near costai together with Anopheles COI sequences from GenBank, identified An. near costai G1 (24 of 25) and An. near costai G2 (1/25). A comparison of the An. near costai G1 consensus sequence with the single An. near costai G2 sequence identified a 4.5% pairwise difference.
Nyssorhynchus dunhami was predominantly collected at the beginning of the wet months, December-January, but seasonally, it accounted for 3.6% during the rainy months and 6.1% during the dry (Figs. 4, 5 and 6; Supplementary Table S5). This species was identified in 2018 in the Peruvian Amazon and as a secondary malaria vector for the first time throughout its Amazonian Basin distribution12. Other species such as Ny. triannulatus (1.9%), Ny. oswaldoi s.l. (0.8%), and Ny. benarrochi B (0.5%), were collected in low quantities during both wet and dry months. The species composition decreased during the dry months (July-November). Nyssorhynchus rangeli, Ny. tadei, and other species of Anopheles were collected in all months in smaller quantities (Supplementary Tables S4 and S5). Unique COI sequences used to identify mosquito species molecularly were deposited in GenBank [Accession ids: PQ530459 – PQ530500].
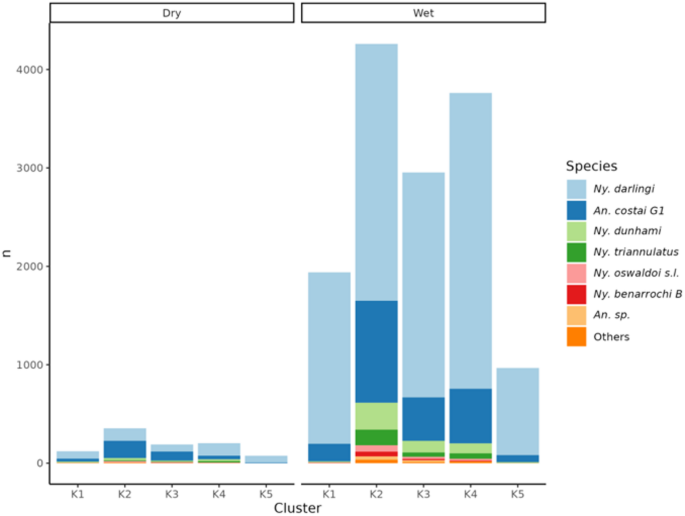
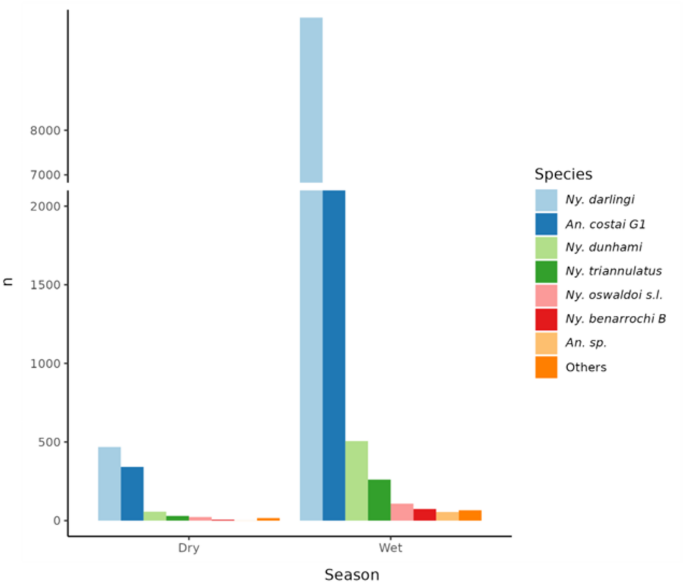
The analysis of captured anophelines shows that Ny. darlingi was the most abundant species overall, in all clusters (over 50%; Fig. 5) and in both seasons (76% in wet, 50% in dry; Fig. 6). The results of the chi-square test of percent abundance of Ny. darlingi (76% rainy, 50% dry) and An. near costai G1 (16% rainy, 36% dry) of all mosquitoes collected by season, was highly significant (chi-square = 12.87, p = 0.0003). During the rainy season (December-June), the clusters with the highest abundance were K2 (31%) and K4 (27%), corresponding to forests and degraded forest patches, respectively. Additionally, these clusters recorded a higher species composition, including An. near costai G1, Ny. dunhami, Ny. triannulatus, Ny. oswaldoi, Ny. benarrochi B, Ny. rangeli, and other species of the genus Anopheles, compared with the remaining landscapes (Fig. 5). Clusters K3 (21%) and K2 (14%), representing cropland and household landscapes respectively, recorded lower abundances of anophelines but were higher than flood-prone areas (K5: 7%), which had both low abundance and species composition. During the dry season (June-November), abundances decreased in all clusters.
The abundance of mosquitoes is directly correlated with seasonality; during the wet season (December-June), the highest abundances (71%) and species composition (17 in total) were recorded, whereas in the dry season, both measures decreased (29%, 13 species) (Fig. 6). Nyssorhynchus darlingi was the most abundant species in each season, followed by An. near costai G1. Other species such as Ny. dunhami, Ny. triannulatus, Ny. oswaldoi s.l., and Ny. benarrochi B were also collected in both seasons, but in greater abundance during the rainy season.
The monthly Human Biting Rate (HBR) of Ny. darlingi varies greatly. April and May recorded the highest peaks, with 650 and 674 bites/person/night (bpn), respectively, unlike the months of August and September, which registered the lowest rates, 11 and 9 bpn, respectively (Table 2). In December, a rate of 526 bpn was recorded, decreasing to 130 bpn in January. This may be attributed to the rising river level in December followed by a decrease in January, affecting breeding sites and consequently adult mosquito abundance. The second most abundant species, An. near costai G1, is highest between April and May (HBR of 528 and 571 respectively), and almost disappears in August and September (HBR of 8 and 5, respectively) (Table 2).
Infection rates of Ny. darlingi and An. near costai G1
Of the 10,901 Ny. darlingi mosquitoes tested for Plasmodium by ELISAs, 21 were positive. Nineteen of 21 Ny. darlingi were infected with P. vivax, and 2 with P. falciparum; no mixed infections were detected. Most infected Ny. darlingi (57%) were detected in April and May. These months also show Entomological Inoculation Rates (EIR) above 1 infected mosquito biting (Apr: 1.0 and May: 2.0) (Table 2). The analysis of Ny. darlingi entomological indices by cluster shows relatively high HBR, the highest being in K4 (degraded forest) in May, recording 262.5 bpn and in April, 201 bpn, along with the highest EIR (1.0 in both months). Household landscapes, cluster K1, recorded an HBR of 189.3 bpn and an EIR of 1.0 in May, whereas the flood-prone K5 cluster recorded an EIR of 0.5 in March and May (Table 3).
Of 2,602 An. near costai G1 tested individually by real-time PCR39, 10 were positive. Four of 10 were infected with P. vivax, 4 with P. malariae/brasilianum, and 2 could only be identified as Plasmodium spp. The greatest number of infected An. near costai G1 was found in clusters K4 (n = 4, degraded forest) and K2 (n = 3, forest), when the Nanay River is rising in December (Table 2; Supplementary Table S6; Fig. 4a, b). Also, one infected An. near costai G1 was identified in each of K1 (household landscapes), K3 (cropland) and K5 (seasonally flood-prone). By cluster, the highest EIR values of 1.00 were in degraded forest (K4) in September and flood-prone areas (K5) in November. In addition, there were three clusters with EIR of 0.50: degraded forest (K4) in August; household landscape (K1) and cropland, both in December (Supplementary Table S6). Considering all Plasmodium-infected mosquitoes, infected Ny. darlingi and An. near costai G1 were detected mainly in the wet months i.e., 28/31; the IRR for the rainy season was 2.2 times higher than that for the dry season (p
Parity rate and longevity of Ny. darlingi
A total of 1,331 Ny. darlingi females were dissected (7% of those collected), with a monthly mean parous rate of 64.3% (range 51.3 ± 81.1). No significant differences were found between months, although the highest parity rates (81%) were in May and August, whereas the lowest rates were in December and January (48.7% and 52.5%, respectively). For nulliparous females, the rate was 32.8% (range 18.9 ± 52.5), with the lowest monthly mean rate in August (19%) and the highest in January (52.5%). No significant differences were found between wet and dry months. The age of Ny. darlingi populations ranged from 2.94 to 11.61 days. Daily survival rates ranged between 71 and 92% (mean of 81.9%), indicating a high probability of daily survival for this species. Ovary dissections were not performed in October and November due to the low quantity of collected mosquitoes (Table 4).
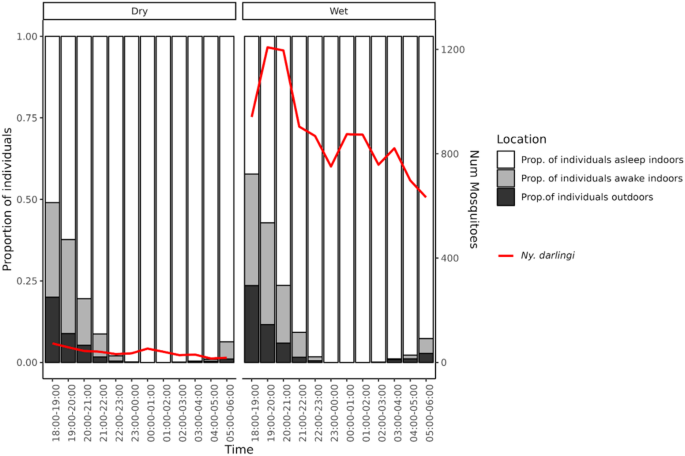
Sleeping behavior and mosquito abundance
In STR, 40% of Ny. darlingi mosquitoes were captured outdoors between 18:00 and 22:00 h, when people were still outdoors or were indoors but awake. The majority of infected Ny. darlingi mosquitoes (81%) were captured between 18:00 and 00:00 h, when many people were unprotected by mosquito nets (Fig. 7). The chi-square test was used to compare the distribution of individuals across three activity states (outdoors, awake indoors, and asleep) between the rainy and dry seasons at specific time intervals. Results indicate that, at the onset of the night (18:00–21:00 h), the proportion of individuals who were awake indoors and/or outdoors was significantly higher during the rainy season compared to the dry season. In contrast, a significantly higher proportion of individuals were asleep during this same time period in the dry season (p 5).
The analysis of the comparison of the mean proportion of time individuals spent outdoors between 18:00 and 22:00 h among those who tested PCR-positive and PCR-negative for Plasmodium vivax showed no statistically significant difference between the two groups (p > 0.05), suggesting that outdoor exposure during this time window does not differ substantially based on P. vivax infection status.
Distribution of malaria infections, Ny. darlingi abundance, and infected mosquitoes
A Poisson GLM found that Ny. darlingi counts were significantly associated with both geographic cluster and season. Taking household landscapes as the reference category, mosquito abundance was markedly higher in degraded patches (IRR = 1.72; 95% CI: 1.63–1.83), forested areas (IRR = 1.51; 95% CI: 1.42–1.60), and crop zones (IRR = 1.30; 95% CI: 1.22–1.38). In contrast, flood-prone areas were associated with significantly lower mosquito counts (IRR = 0.52; 95% CI: 0.48–0.57). Seasonal variation was also pronounced, with mosquito counts nearly five times higher during the wet season compared to the dry season (IRR = 4.94; 95% CI: 4.70–5.19; p
In the southeastern sector of STR there was a higher abundance of Ny. darlingi collected by HLC and human Plasmodium infections detected mainly by qPCR (Fig. 8). In this area houses are in close proximity to the river when it reaches its highest level; also, when the river recedes at the end of the wet season there is a greater number of residual water bodies (compared with other sectors of the village) (Fig. 8). In contrast, the highest abundance of An. near costai G1 is in the southwestern sector, which is more forested (Supplementary Figure S3).
Spatial distribution of malaria infections and Ny. darlingi abundance by season in Santa Rita, Peru, 2021–2022. Triangles represent the number of Ny. darlingi mosquitoes captured, with the darkest blue being the highest number, and circles represent the number of human P. vivax infections identified by PCR, red circles representing the highest numbers.