Study design, study population and definitions
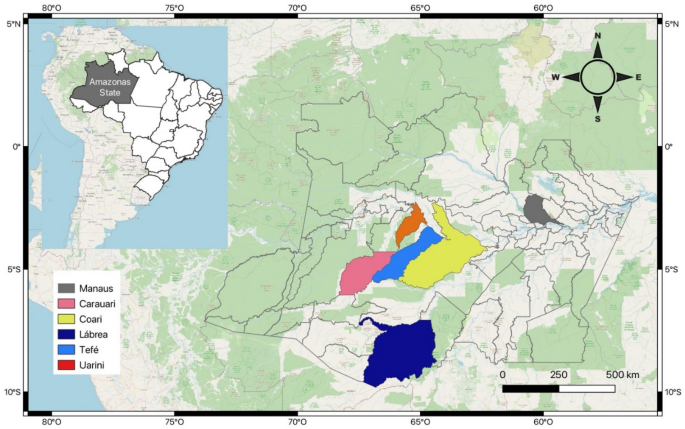
This serological survey was conducted between July 2022 and June 2023 to identify individuals affected by CD outbreaks linked to oral transmission through the consumption of açaí juice (Euterpe spp.). Active case finding was employed for comprehensive participant identification. These outbreaks occurred in five municipalities in the state of Amazonas (Carauari, Coari, Lábrea, Tefé, and Uarini) between 2004 and 2022 (Fig. 4). All individuals exposed/infected during these outbreaks received medical care from the CD service team at FMT-HVD, a national tertiary reference center for tropical and infectious diseases. During active case-finding visits, a structured questionnaire was administered to collect sociodemographic data (sex, age), clinical history (previous leishmaniasis diagnosis, history of blood transfusion), and epidemiological/behavioral information (forest exposure, ability to identify triatomines at home, consumption of palm fruits and bushmeat).
Map of the state of Amazonas highlighting the five municipalities where the study was conducted. The ACD outbreaks peer municipality resulting from açaí ingestion occured between 2004 and 2021: Tefé (outbreak in 2004), Coari (outbreak in 2007), Carauari (outbreaks in 2001 and 2015), Lábrea (outbreaks in 2018 and 2022), and Uarini (outbreaks in 2019 and 2021). The map was created using QGIS 3.32 software.
Based on retrospective medical record analysis, participants were categorized into two groups according to the inclusion criteria:
Group 1 (G1): Individuals who, during the outbreak of oral transmission, were diagnosed with T. cruzi infection. Diagnosis was confirmed through thick smear examination, positive IgM serology (or IgG seroconversion), along with symptoms consistent with ACD. All participants in this group were treated with benznidazole, experiencing clinical improvement post-treatment.
Group 2 (G2): Individuals who were exposed, took the same contaminated food responsible for the outbreak cases, but did not present any clinical or laboratory indications of a T. cruzi infection.
The non-inclusion criteria for the study included: individuals diagnosed and treated during outbreak events, and unwilling participate in the serological survey.
Participants in G1 were assessed for serological cure following treatment. Serological cure was defined as having non-reactive results in all conventional serological tests (IIF, ELISA-B and ELISA-W) and in the in-house species-specific tests (ELISA-TcI and ELISA-TcIV) using serum samples collected during the active search.
For G2, participants were classified based on seroconversion. Seroconversion was defined as reactive serological results in at least two tests with different principles (ELISA + IIF), using serum samples from the active search, indicating presumed CCD. All participants were also asked whether they were receiving medical follow-up for CD.
Sample collection
During the active search, 10 mL blood were collected from each participant into vacutainer tubes without anticoagulant. The samples were then transported to the Nelson Fé Entomology Unit at Fundação de Medicina Tropical Doutor Heitor Vieira Dourado (FMT-HVD) in Manaus, AM. Here, the coagulated blood was centrifuged at 1200 g for 10 min at room temperature. The resulting serum sample was separated, aliquoted, and stored at −20 °C until when needed.
Commercial serological tests
All commercial tests were performed according to the manufacturers’ recommendations. The serological procedures for screening the samples were initially performed at the Central Laboratory of Amazonas (LACEN), where serum samples were tested using two commercial kits: the ELISA Kit Chagas Bioclin (ELISA-B), that use recombinant antigens, with a sensitivity of 99.9% and specificity of 99.3%; and the Indirect Immunofluorescence (IIF) CHAGAS kit – Biomanguinhos, the manufacturer did not provide the specific DTU used in this test, with sensitivity and specificity of ≥ 90%. Both assays provided only qualitative results (reactive or non-reactive). In parallel, at FMT-HVD, the same samples were tested using an additional commercial assay ELISA kit Chagatest v.4.0, Wiener lab (ELISA-W), that use recombinant antigens (SAPA, 1, 2, 13, 30 and 36), with sensitivity range of 99.1–99.9% and specificity of 98.3–99.7%. This test provided quantitative results (reactivity index – RI).
Antigen preparation for in-house assays
The T. cruzi TcI or TcIV antigens used here were produced as described by Cervantes-Landín et al. (2014)40 with some modifications. The T. cruzi TcI or TcIV strains isolated from blood samples of patients diagnosed with acute Chagas disease (ACD) native to the Amazon were used to prepare the parasite mass.
Briefly, cryopreserved T. cruzi isolates were activated in Novy-MacNeal-Nicolle (NNN) medium thereafter cultured in Schneider medium supplemented with 20% fetal bovine serum (FBS) for the multiplication of epimastigote forms for 3 days (TcI) or 2 days (TcIV), at 28 °C. After growth of the isolates, the parasite suspension was centrifuged at 3,500 g for 20 min, discarding the supernatant. The precipitate containing the parasite biomass was washed thrice with Phosphate Buffer Solution (PBS) under the same centrifugation conditions. Approximately 6 × 105 parasitic forms were suspended in 500 µl of 10 mM TRIS-HCl buffer, pH 8.2 with protease inhibitor (12 mM EDTA, 0.1 mM leupeptin) and sonicated for 30 min (UltraCleaner 750 A). The resulting lysate was centrifuged at 12,000 g for 20 min, at 4 °C, and then the supernatant, containing soluble antigens, aliquoted and stored at −20 °C. Protein concentration was determined by the Pierce BCA Protein Assay Kit (Thermo Scientific) following the manufacturer’s instructions. The generated parasite antigens were subsequently used in ELISA-TcI, ELISA-TcIV, WB-TcI, and WB-TcIV assays.
In-house ELISA
Two non-commercial ELISA, developed and standardized at FMT-HVD, (ELISA-TcI and ELISA-TcIV) were used to evaluate the capacity to detect species/lineage-specific antibodies from the serological surveillance. For this purpose, 96-well polystyrene plates (Thermo Fisher Scientific) were sensitized with 10 µg/ml of antigen (TcI or TcIV), incubated overnight at 4 °C followed by washing with PBS containing 0.05% Tween 20 (PBS-T). Nonspecific sites were then blocked with PBS containing 10% FBS with a 2 h incubation step. After washing the wells again, 50 µl/well of serum samples diluted at 1:100 was added and incubated overnight at 4 °C. After six washing cycles, the plates were incubated for 1 h with the anti-human IgG conjugate (INVITROGEN) at a dilution of 1:500. After another wash step, the OPD (O-Phenylenediamine) substrate was added before incubating in the dark for 10 min. The reactions were stopped using 2 N HCl and absorbance readings obtained using a microplate reader (ThermoPlate) at a wavelength of 492 nm. The results were expressed as a reactivity index. Receiver Operating Characteristic (ROC) curve generated using known negative and positive control samples was used to establish cut-off for distinguishing between reactive and non-reactive field samples. Sensitivities of 92% and specificities of 91.6% were observed in both assays.
In-house Western blot
To identify the subfractions of soluble antigens recognized by ELISA-TcI and ELISA-TcIV, a representative sample from G1 (n = 22) and G2 (n = 7) was evaluated using customized Western Blot tests (WB-TcI and WB-TcIV). Here, soluble antigens were separated using 8% SDS-polyacrylamide gel electrophoresis (SDS-PAGE) and transferred to PVDF membrane in a semi-dry Power Blotter System. The membrane was then cut into strips (approximately 20 µg of antigen/strip) which were subsequently placed into individual channels for non-specific binding site blocking. The blocking was done incubating in PBS-Tween 0.05% (PBS-T) containing 5% skimmed milk for 2 h under rocking motion. The strips were the washed with PBS-T solution, before adding diluted serum samples (1:100) and incubated with agitation overnight at 4 °C. Individual strips were six times using PBS-T solution, before adding anti-human IgG conjugate (INVITROGEN) diluted in 1:500. After a 2-hour incubation under agitation, the strips underwent another wash cycle before adding the DAB substrate (Pierce™) until the bands appeared. The reaction was stopped using distilled water. The WB reactivity results were presented based on the frequency of detected band profile among participants.
Statistical analysis
Sociodemographic, epidemiological and clinical variables, as well as qualitative results of serological tests, were assessed regarding the frequency of occurrence in each of the groups G1 and G2. The age variable was categorized into age groups. Contingency analyses (Fisher’s exact test or Chi-square test) were performed to identify differences in frequencies between the groups. For each group, numerical variables such as reactivity indexes (RI; defined by the sample absorbance value/test cut-off value) were represented by median values and interquartile ranges (25% and 75%). Statistical differences between the groups were assessed by analysis of variance (ANOVA). Spearman correlations were performed between the time of outbreak occurrence and the IR for each ELISA test. A 95% confidence interval (p
Ethical aspects
The study was submitted to and approved by the Human Research Ethics Committee – CEP of the Fundação de Medicina Tropical Doutor Heitor Vieira Dourado (FMT-HVD), under certification CAAE: 97505318.7.0000.0005. All participants willingly provided informed and signed consent before participating in study-related procedures after receiving a detailed explanation of the study protocol. All methods were performed in accordance with the relevant guidelines and regulations, including the Helsinki Declaration.