Trial design and participant’s baseline characteristics
Among 285 participants screened for eligibility, 120 patients were randomized equally (1:1) into two groups: the Control group (n = 60), which received physiological saline solution plus standard treatment of care, and the Navax group (n = 60), which received LiveSpo Navax in addition to standard treatment of care. Each group received 3 sprays of either control 0.9% NaCl or LiveSpo Navax in each nostril and throat, totaling 9 sprays per time meaning 27 sprays per day (administered 3 times daily). This dosing regimen was initiated about 5 hours after hospital admission and continued until discharge, typically lasting 7–10 days. By the end of the treatment period, 50 patients in the Control group and 51 patients in the Navax group were included in the final analysis (Fig. 1).
Patients’ demographic characteristics were shown in Table 1. All children with pneumonia due to RSV infection and bacterial co-infection were under 2 years old (1 to 24 months), with the majority being under 6 months old (>58%). Co-infection was defined as a condition where RSV patients were also confirmed with the presence of any type of bacteria based on RT-PCR assay. There were no significant differences between the two groups regarding (i) age (p = 0.9331), (ii) gender distribution (p = 0.9331), (iii) weight (p = 0.3195), and (iv) days of illness before treatment (p = 0.1489). Patients hospitalized with pneumonia exhibited various clinical symptoms, such as cough, wheeze, fever, rhinitis, retractions, dry and moist rales, diarrhea, and vomiting. Physical signs included poor feeding (p = 0.4364), excessive crying (p = 0.7152), convulsions (p = 0.6779), and cyanosis (p = 0.1894), alongside abnormal vital signs such as tachycardia (beats/min) (p = 0.4906), tachypnea (breaths/min) (p = 0.625), and low oxygen saturation (SpO2) (p = 0.2575). Before treatment, no significant difference between the two groups was observed.
Additionally, baseline sub-clinical characteristics, including chest X-rays (p = 0.9639), hematology and biochemistry (p = 0.2661, 0.423, and 0.4255), serum C-reactive protein (CRP) levels (p = 0.1339), number of cases positive for RSV detected by rapid test and real-time PCR assay (>0.9999), the median Ct values for RSV detection (24.45 in the Control group and 23.20 in the Navax group), and number of cases of bacterial co-infection determined by real-time PCR assay (p = 0.1258) and microbial culture (p = 0.8309), showed no notable variation between the two groups before treatment (Table 2 and Supplementary Table 1).
It is important to note that real-time PCR detected H. influenzae (HI) and S. pneumoniae (SP) in a relatively high number of cases, while M. pneumoniae (MP) and B. pertussis (BP) were found in fewer cases (Table 2). Microbial culture, used exclusively for guiding antibiotic treatment, detected a few additional cases of M. catarrhalis, S. aureus, and P. aeruginosa infections, which were not among the seven bacterial species detectable by the real-time PCR kit (Supplementary Table 1). Therefore, subsequent experiments focused on evaluating changes in H. influenzae (HI) and S. pneumoniae (SP) in nasopharyngeal samples.
Safety and symptom relief: shortening treatment duration for symptoms and antibiotic use for pneumonia in children by nasal-spraying Bacillus spores
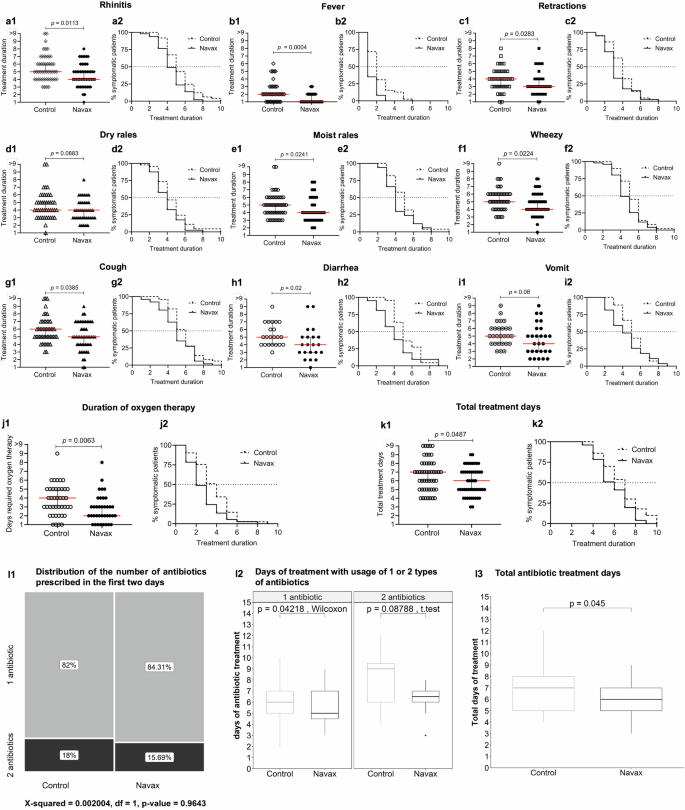
During the treatment period, all participants in both groups showed no symptoms of focal bacterial infection, such as the development of new pharyngeal abscesses, severe otitis media, or worsening rhinitis. There were no cases of nasal mucosal irritation, discomfort during administration, or systemic side effects such as nausea or vomiting. Furthermore, there were no abnormalities observed in vital signs, including heart rate, respiratory rate, and SpO2, following 9 consecutive administrations of either probiotic or placebo sprays over 3 follow-up days (Supplementary Fig. 1). Previous studies have shown that the Bacillus probiotic spore nasal spray is safe for children with RSV and influenza virus30,31. This study confirms its safety for pediatric pneumonia patients. The efficacy of Bacillus spores in alleviating typical clinical symptoms of pneumonia at days 3 and 5 was then evaluated and presented in Table 3. Before treatment (day 0), baseline clinical characteristics of the participants, including cough (p > 0.9999), wheezing (p = 0.495), fever (p = 0.5257), rhinitis (p = 0.495), retractions (p = 0.2205), diarrhea (p = 0.7742), vomiting (p = 0.9151), dry rales (p = 0.1114), moist rales (p > 0.9999), tachycardia (p = 0.4906), tachypnea (p = 0.625), and the need for oxygen therapy (p = 0.2575) did not show statistically significant differences between the two groups. At day 3, the Navax group showed a significantly lower percentage of patients with several symptoms and vital signs, including cough (p = 0.0279), fever (p = 0.0125), retractions (p = 0.0034), diarrhea (p = 0.0478), dry rales (p = 0.0346), moist rales (p = 0.0496), and the need for oxygen therapy (p = 0.0074), and a trend toward a lower percentage of patients with rhinitis (p = 0.0692) and vomiting (p = 0.0876) compared to the Control group. By day 5, Navax treatment lowered the percentage of patients with rhinitis (p = 0.0478), and patients trended toward reduced rates of cough (p = 0.0905) and wheezing (p = 0.0506). After 5 days of treatment, we observed a similar proportion of patients showing improvements in the remaining clinical symptoms and vital signs in both the Control and Navax groups (Table 3).
In the trial, the primary outcome was the treatment duration required to alleviate typical clinical symptoms of pneumonia (Fig. 2a1–k1) with the time-course dependent percentage (%) of patients achieving symptom-free status serving as an alternative representation of this outcome (Fig. 2a2–k2) in the two groups. The term treatment duration refers to the number of days required to resolve a particular symptom after the first day of treatment. Notably, the Navax group experienced faster recovery, resolving most symptoms 1 day earlier compared to the Control group, including rhinitis (p = 0.0113; Fig. 2a1), fever (p = 0.0004; Fig. 2b1), retractions (p = 0.0283; Fig. 2c1), moist rales (p = 0.0241; Fig. 2e1), wheezy (p = 0.0224; Fig. 2f1), cough (p = 0.0385; Fig. 2g1), and diarrhea (p = 0.02; Fig. 2h1). Although the Navax group’s vomiting symptom resolved one day earlier than that of the Control group, the difference was not statistically significant (p = 0.08) (Fig. 2i1). These shortened recovery times improved treatment efficacy for each of those symptoms by approximately 1.2- to 2.0-fold compared to standard treatment. Nevertheless, the treatment duration for dry rales (Fig. 2d1), tachycardia, and tachypnea (Table 3) were similar between both groups. Using Kaplan-Meier analysis, we presented the percentage of symptomatic patients over the treatment period. The analyses showed that the Navax group consistently experienced lower percentages of symptomatic patients at most follow-up time points for 10 out of 11 symptoms and vital signs, as shown in Fig. 2a2–2k2. The time at which 50% of patients recovered in the Navax group was generally 1-day earlier compared to the Control group, highlighting the faster recovery facilitated by Navax treatment. For the dry rales, although there was no difference in the time to 50% recovery between the two groups, a clear trend showed a higher percentage of recovered patients in the Navax group at various follow-up time points (Fig. 2d2). Overall, LiveSpo Navax reduced the duration of oxygen therapy by 2 days (2 days in Navax vs. 4 days in Control, p = 0.0063; Fig. 2j1-j2) and shortened the total pneumonia treatment duration by reducing hospitalization by 1 day (6 days in Navax vs. 7 days in Control, p = 0.0487; Fig. 2k1-k2).
Treatment duration (number of days to resolve symptoms) (a1–k1) and Kaplan–Meier analysis of estimated percentage (%) of symptomatic patients (a2–k2) observed over treatment duration for symptoms of pneumonia in the Control (dashed lines) and Navax (solid lines) groups. The distribution of the number of antibiotics prescribed in the first two days (l1), duration of antibiotic treatment with 1- and 2-type antibiotics (l2), and total antibiotic treatment days (l3) prescribed for children with pneumonia in the Control and Navax groups. Statistical significance was evaluated using the Mann–Whitney test. Median values with 95% CI for each group and the median difference between the two groups are shown in panels a–k. Error bars in panels l2–l3 indicate the upper extreme (Q3 + 1.5 × IQR) and the lower extreme (Q1 – 1.5 × IQR). The significance level for all analyses was set at p < 0.05. Sample size: n = 50 biologically independent patients in the Control group and n = 51 in the Navax group.
For children with pneumonia caused by RSV and bacterial co-infections, antibiotic therapy is essential to inhibit pathogenic bacteria. Antibiotics are prescribed based on standard treatment regimens and antibiogram test results, combined with close follow-up on the treatment (Table 1). The distribution of patients using 1- or 2-type antibiotic regimens, including the names and combinations of antibiotics used, was observed to be similar in the Navax and Control groups within the initial 2 days (82% in Control vs. 84.31% in Navax for 1-type antibiotic, 18% in Control vs. 15.69% in Navax for 2-type antibiotic; p = 0.9643) (Fig. 2l1, Supplementary Table 2). The Navax group showed a reduction in the duration of treatment with 1-type antibiotic by 1 day (p = 0.04218) and with 2-type antibiotics by 3 days (p = 0.08788) (Fig. 2l2). Overall, the total antibiotic treatment time in the Navax group was significantly reduced by 1 day (7 days in Control vs. 6 days in Navax, p = 0.045) (Fig. 2l3). This reduction corresponds to a 14.3% decrease in antibiotic usage for treating pneumonia.
Reducing the concentration of RSV and co-infecting bacteria by nasal-spraying Bacillus spores
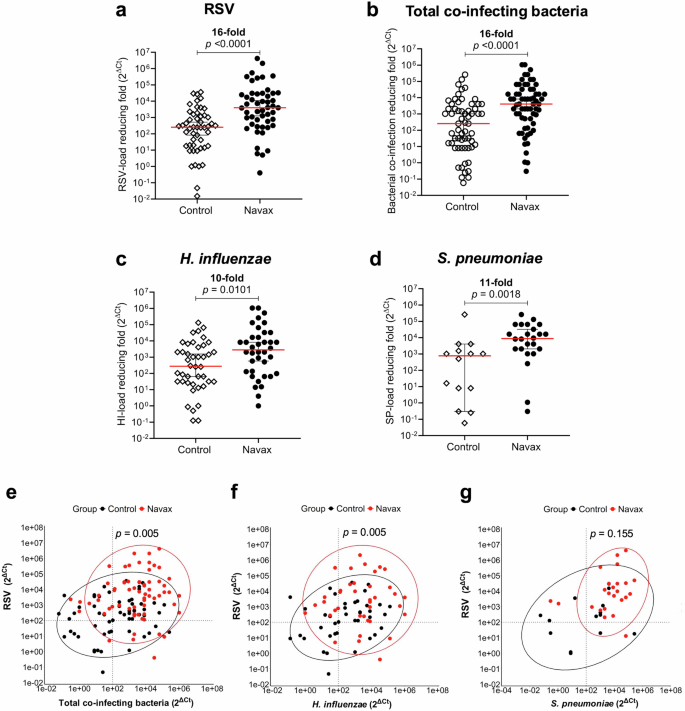
To explore the scientific background of how Bacillus spores alleviate symptoms, we conducted real-time PCR/RT-PCR TaqMan probe assay to analyse fold changes (calculated by 2△Ct, where △Ct = Ct day 3 – Ct day 0) in RSV loads and concentrations of H. influenzae and S. pneumoniae in nasopharyngeal samples, as described in the Methods section. The turnaround time for the assay is approximately 5 hours, making it well-suited for hospital operations with time constraints. Moreover, this semi-quantitative approach, certified under ISO 15189:2022, ensures reliable and standardized results. By detecting both RSV and bacteria in the same clinical sample, we confirmed that a patient was simultaneously infected with RSV and bacteria. Therefore, our analysis semi-quantified reductions in RSV loads and bacterial concentrations to primarily assess the impact of Bacillus spores in enhancing the effectiveness of standard therapy. As shown in Fig. 3a, the 2△Ct data revealed a significant reduction in RSV load in the Navax group, by 3956-fold, which was 16-fold more effective than the 252-fold reduction observed in the Control group (p < 0.0001). Regarding the reduction in a total co-infecting bacterial concentration (including H. influenzae, S. pneumoniae, M. pneumoniae, B. pertussis), the Navax group demonstrated a substantial reduction of 4096-fold, which was 16 folds greater (p < 0.0001) than that in the Control group (256-fold reduction) after 3 days of treatment (Fig. 3b). Notably, among cases infected with H. influenzae and S. pneumoniae, the most common co-infecting bacterial species in respiratory tract, the reduction in H. influenzae and S. pneumoniae load was 2807 and 8801 folds, respectively, in the Navax group, which were 10–11 folds more effective than those in the Control group (p = 0.0101 and 0.0018, respectively; Fig. 3c, d).
Reducing-fold levels (2△Ct) of RSV load (a), total co-infecting bacterial concentration (b), H. influenzae load (c), and S. pneumoniae load (d) in nasopharyngeal samples of Control and Navax groups at day 3 compared to day 0. The differences in the associations between reduced RSV load and reduced total bacterial co-infection concentrations (e), reduced RSV load and reduced H. influenzae (f), and reduced RSV load and reduced S. pneumoniae (g) were confirmed using the energy test. The median differences for these indicators between the two groups were calculated using the Mann–Whitney test. Median values with 95% CI for each group and the median difference between the two groups are shown in panels (a–d). All analyses were considered significant at p < 0.05. Sample size: n = 50 biologically independent samples in the Control group and n = 51 in the Navax group.
Furthermore, we used multiple logistic regression to assess the link between reduced RSV load and reduced co-infecting bacterial concentrations, including H. influenzae and S. pneumoniae (Fig. 3e–g). A reduction threshold of 10² was set, reflecting the reduction levels seen in the Control group following standard hospital treatment. In the Navax group, patients with significant RSV reductions also showed substantial total co-bacterial load reductions, as seen in the red plots clustering in the upper right quadrant (Fig. 3e). This trend differed significantly from the Control group, where patients clustered around the cut-off and lower left quadrant (p = 0.005). Similar correlations were found between RSV reductions and reductions in individual H. influenzae (p = 0.005) and S. pneumoniae (p = 0.115) concentrations (Fig. 3f, g). Bacillus spores (B. subtilis and B. clausii) were detected in the Navax group’s nasopharyngeal samples on day 3, but not in the Control group, confirming proper medication intake. These results indicate that nasal-spraying Bacillus spores enhance the effectiveness of standard therapy by simultaneously reducing RSV and co-bacterial infection loads. It is noted that both groups received inhaled corticosteroids (budesonide) during treatment, with 41 out of 50 patients in the Control group and 41 out of 51 patients in the Navax group. This balanced use of corticosteroids minimizes potential bias in the analysis of RSV load dynamics and mucosal cytokine responses.
Modulating cytokine and IgA responses by nasal-spraying Bacillus spores
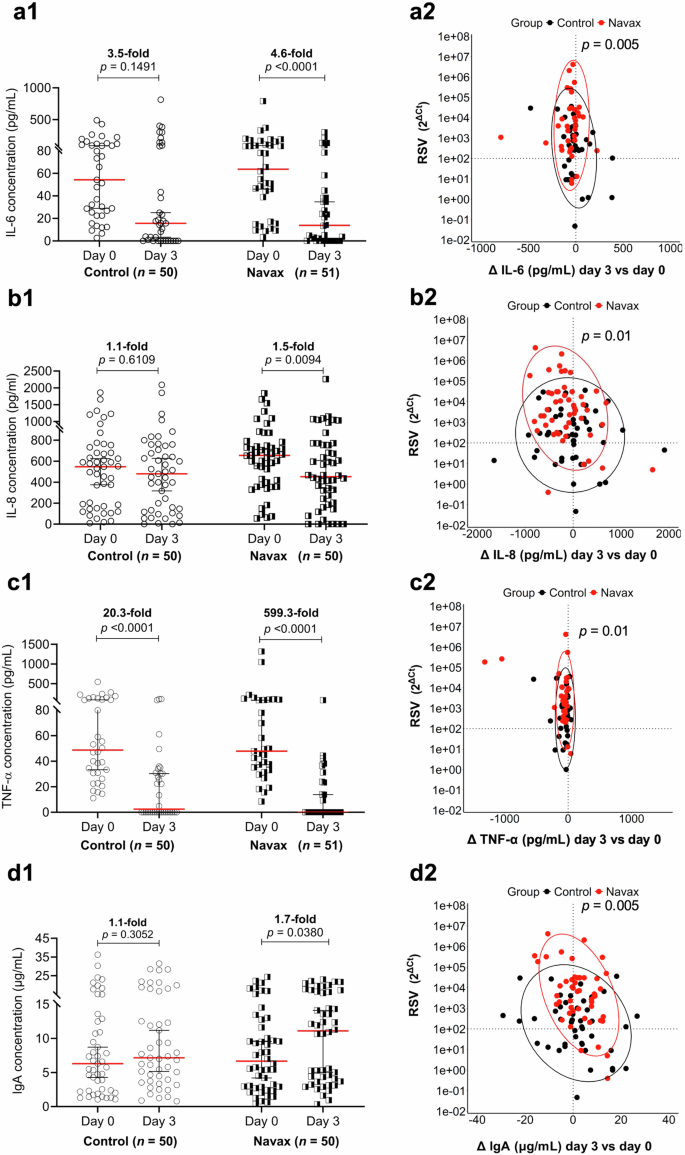
Our previous research shows that Bacillus spores can modulate the immune system by supressing the overexpression of pro-inflammatory cytokines and enhancing mucosal IgA levels in children with ARTIs caused by RSV and influenza virus30,31. Based on this, we predicted that LiveSpo Navax could have similar effects in children with pneumonia from viral and bacterial infections. We examined changes in key pro-inflammatory cytokines (IL-6, IL-8, and TNF-α) and mucosal Immunoglobulin A (IgA) in nasopharyngeal samples between day 0 and day 3 to assess these effects, as symptoms showed the most noticeable improvement during this period. After 3 days of treatment, the Navax group showed significant reductions in IL-6 (4.6-fold, p < 0.0001) and IL-8 (1.5-fold, p = 0.0094), whereas the Control group showed no significant reduction (p = 0.1491 and 0.6109, respectively) (Fig. 4a1, 4b1). Both groups experienced a marked decrease in TNF-α levels after 3 days (p < 0.0001). The Navax group showed an almost 600-fold reduction (from 47.94 to base line of 0.08 pg/mL), nearly 30-fold greater than the Control group (from 48.77 to 2.40 pg/mL) (Fig. 4c1). Additionally, we assessed the effect of Bacillus spore nasal spray on changes in IgA levels (µg/mL) in the nasopharynx, comparing measurements between days 0 and 3. Both groups exhibited a slight increase in IgA levels (Fig. 4d1) from day 0 to day 3, with the Navax group showing a more robust 1.7-fold increase, which was statistically significant (p = 0.038). These findings indicate that nasal-spraying Bacillus spores significantly reduced the overproduction of IL-6, IL-8, and TNF-α cytokines, and moderately increased nasal IgA levels in response to RSV.
Pro-inflammatory cytokine levels (pg/mL) and IgA levels (mg/mL) (a1–d1) in nasopharyngeal samples of Control and Navax groups at day 3 compared to day 0. The association between reduced RSV load and ▵cytokine/IgA levels from day 0 to day 3 (a2–d2) was assessed using multiple logistic regression analysis. The Wilcoxon test was used to calculate the median differences in IL-6, IL-8, TNF-α, and IgA levels at day 0 and day 3 within each group. The differences in the associations between reduced RSV load and changes in IL-6 levels (a2), reduced RSV load and changes in IL-8 levels (b2), reduced RSV load and changes in TNF-α levels (c2), and reduced RSV loads and changes in IgA levels (d2) were validated using the energy test. Comparisons of cytokine concentrations between the two groups were made using the Mann-Whitney test. Only measurable samples at day 0 were included in the statistical analysis. 95% CI for the median in each group and the median difference between the two groups were shown. The significance level for all analyses was set at p < 0.05. Sample size: n = 50 biologically independent samples for all three cytokines and IgA in the Control group; n = 51 for IL-6 and TNF-α, and n = 50 for IL-8 and IgA in the Navax group.
Moreover, we used multiple logistic regression to evaluate the correlation between reduced RSV load and changes in cytokine levels and IgA concentrations (Figs. 4a2–d2). A reduction threshold of 10² was also maintained for RSV load, with cytokines/IgA set at zero. Patients with RSV reductions greater than 10² showed a significant decrease in IL-6 (p = 0.005), IL-8 (p = 0.01), and TNF-α (p = 0.01) levels in Navax group, as indicated by red plots distributing on the upper horizontal center line (Figs. 4a2–c2). Meanwhile, the Control group had more variable plots with some patients even showing increased cytokine levels. Additionally, most patients in the Navax group who experienced substantial RSV reduction also saw an increase in IgA levels, unlike the Control group, which showed no clear trend (p = 0.005, Fig. 4d2). Overall, the Navax group demonstrated a stronger reduction in viral load, which correlated with lower cytokine levels and higher IgA levels compared to the Control group.
Improving nasal microbiota diversity and density at the phyla, genus and species levels by nasal-spraying Bacillus spores
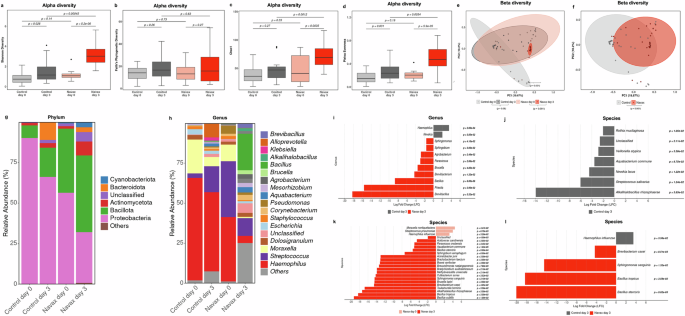
The nasal microbiota plays a crucial role in respiratory health. A balanced nasal microbiota signals good health, while imbalances increase infection risk38,39. To evaluate probiotic effects, we analyzed nasal microbiota changes from 16 representative samples in each group between day 3 and day 0. Initially, both the Navax and Control groups had similar microbial diversity (Fig. 5a-d). After 3 days, diversity increased in both groups, with the Navax group showing more significant increases: 2.8-fold in Shannon (p = 5.2e-06), 1.7-fold in Chao1 (p = 0.0035), and 2.3-fold in Pielou’s evenness indices (p = 5.9e-05). The Control group exhibited smaller increases with 1.8-fold in Shannon (p = 0.026) and 1.7-fold in Pielou’s evenness (p = 0.031), while the increase in Chao1 was not significant (p = 0.27). Notably, the Navax group had a 1.3–1.8-fold higher diversity than the Control group on day 3 (Shannon: p = 0.0045; Chao 1: p = 0.0012; Pielou’s evenness: p = 0.0034). Data obtained from PCoA analysis revealed a more localized distribution in the Navax group on day 3 compared to broader distributions in the Control group (Fig. 5e). Overall, nasal microbiota composition differed significantly between the groups both before and after probiotic treatment (Fig. 5f). Furthermore, we found that the nasal microbiota composition in the analyzed groups was primarily dominated by bacteria from the phyla Proteobacteria, Bacillota (Firmicutes), and Actinobacteria, while Cyanobacteria, Bacteroidetes, and other phyla were present in significantly lower density. Before treatment, both groups had high densities of Proteobacteria. However, by day 3, Proteobacteria significantly decreased, more so in the Navax group (1.80-fold) than in the Control group (1.36-fold). Conversely, Bacillota increased by day 3, with 1.20-fold in the Navax group and 2.30-fold in the Control group (Fig. 5g).
Alpha diversity (Shannon, Chao1, Faith’s PD, Pielou Evenness) (a–d) and beta diversity (PCoA) (e, f) of 16S rRNA microbiota in nasopharyngeal samples from Control and Navax groups at day 3 compared to day 0. The Wilcoxon test and Mann–Whitney test were used for comparisons of alpha diversity within each group and between the two groups. The significance level for all analyses was set at p < 0.05. Distribution of 6 major phyla in the two groups at day 3 compared to day 0 (g). Distribution of the 18 major genera in the Navax and Control groups at day 3 compared to day 0 (h). Log2 fold change (LFC) in the relative abundance of genera in the Navax group compared to the Control group at day 3 (i). LFC in the relative abundance of species in the Control group at day 0 compared to day 3 (j); in the Navax group at day 0 compared to day 3 (k); in the Navax group compared to the Control group at day 3 (l). The Lefse algorithm was used to evaluate the differences between these characteristic markers. Only markers with statistically significant differences (p < 0.05) were included in the analysis. Error bars in panels a–d indicate the upper extreme (Q3 + 1.5 × IQR) and the lower extreme (Q1 – 1.5 × IQR). Sample size: n = 16 biologically independent samples in the Control group and n = 16 in the Navax group.
In the genus-level taxonomic analysis, we evaluated the 18 genera with the highest relative abundances (Fig. 5h). At day 0, the microbiota structure of both the Navax and Control groups was similar, predominantly comprising of genera commonly associated with respiratory pathogens, with Haemophilus accounting for the highest proportion (40–64%), followed by Moraxella (10–21%) and Streptococcus (2–35%). Meanwhile, the relative abundance of beneficial genera such as Bacillus, Brevibacillus and Priestia was low (<0.3%) in both groups. No statistical difference in their abundance was observed between the Control vs. Navax groups. By day 3, in the Control group, Haemophilus and Moraxella decreased by only 1.29- and 3.65-fold, with relative abundances of 49.49% and 5.77%, respectively, while Streptococcus increased 5.44-fold to 16.14%. In contrast, in the Navax group, Haemophilus, Moraxella, and Streptococcus were significantly reduced by 9.08-, 13.33-, and 3.15-fold, reaching relative abundances of 4.42%, 2.12%, and 1.87% at day 3, respectively. Notably, the Navax group exhibited increased diversity of beneficial genera after 3 days, with the emergence of Bacillus (0.26% at day 0 vs. 22.91% at day 3), Alkalihalobacillus (not detectable at day 0 vs. 1.70% at day 3), and Brevibacillus (not detectable at day 0 vs. 2.89% at day 3). In contrast, these genera were almost absent in the Control group at both time points. In summary, the nasal microbiota composition in the Control and Navax groups was comparable on day 0, but showed distinct differences on day 3.
We further conducted a Lefse algorithm analysis36 to identify specific genera and species exhibiting changes in density, either between the two groups or between the two time points of probiotic intervention (days 0 and 3) within each group. Results shown in Fig. 5i indicated 11 genera having significant differences in densities between the two groups at day 3 of the treatment period. Most notable were indicators directly related to the respiratory tract, including Haemophilus, Bacillus, Brevibacillus, and Priestia. Haemophilus showed higher residual presence in the Control group vs. the Navax group with a Log2 Fold Change (LFC) of 3.49 (p = 0.0002). The Navax group retained the presence of Brevibacillus and Priestia, which were absent in the Control group (LFC ranging from −15.61 to −18.23). On the other hand, the Bacillus genus was present at more than an 8.67-fold higher level in the Navax group compared to the Control group.
In terms of species taxonomy, in the Control group, we observed an increase in only two nasal commensal species, Streptococcus salivarius and Veillonella atypica (LFC ranging from −2.84 to −6.01), and no reduction in the harmful species H. influenzae at day 3 compared to day 0 (Fig. 5j). As shown in Fig. 5k, there was a noticeable change in the species composition of the Navax group from day 0 to day 3. At day 0, the dominant co-infection bacterial species related to pneumonia in children, such as M. nonliquefaciens, S. pneumoniae, and H. influenzae, were significantly higher compared to day 3, with LFC (Navax day 0 vs. day 3) of 4.46 (p = 0.0447), 3.97 (p = 0.0067), and 3.18 (p = 0.0328). Conversely, by day 3, the Navax group exhibited a significant increase in beneficial species from the genera Bacillus and Brevibacterium, including B. subtilis (LFC = −19.34, p = 0.01), B. tropicus (LFC = −18.32, p = 0.0358), B. casei (LFC = −14.57, p = 0.0405), and B. stercoris (LFC = −5.47, p = 0.0009), which were almost absent at day 0 (Fig. 5k). Additionally, at day 3, the specific species indicator for the Control group was H. influenzae compared to the Navax group (LFC = 3.49, p = 0.0358). In contrast, the Navax group was distinguished from the Control group by the presence of beneficial species such as B. tropicus (LFC = −18.32, p = 0.0358), B. stercoris (LFC = −20.08, p = 9.02e−5), and B. casei (LFC = −4.29, p = 0.00851), which were nearly absent in the Control group after 3 days of treatment (Fig. 5l).