Dynamics without cure
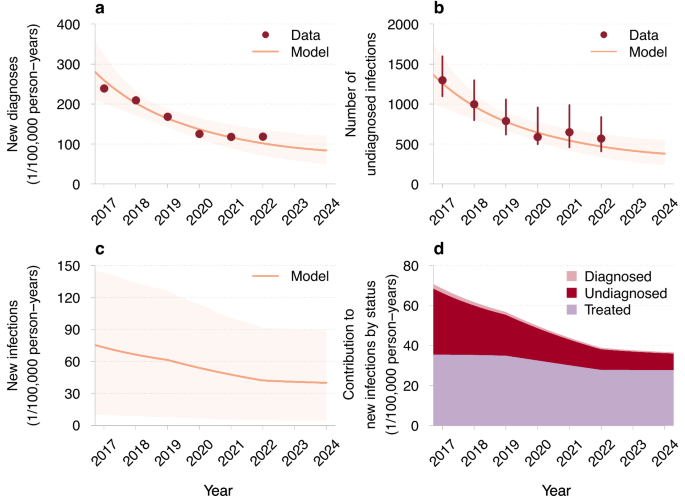
The model was fitted to and reproduced well the annual number of new HIV diagnoses and the estimated number of undiagnosed HIV infections among MSM from 2017 till 2022, reported by the Dutch HIV Monitoring Foundation (Stichting hiv monitoring, SHM) (Fig. 1)2. The number of new HIV infections per 100,000 persons per year acquired in the country was estimated to decrease from 73 (95%CrI 10–143) in 2017 to 42 (95%CrI 5–91) in 2022. The estimated mean time to diagnosis was 28 months (95%CrI 24–32), consistent with the SHM data2. The model predicted that, among all new HIV diagnoses, the proportions of diagnoses within 6 months, between 6 to 12 months, and more than 12 months since HIV infection were 22% (95%CrI 20–24%), 14% (95%CrI 13–16%), and 63% (95%CrI 60–66%), respectively, also aligning with the SHM data2. In 2024, treated individuals accounted for 75% (95%CrI 23–94%) of new infections acquired in the country, undiagnosed individuals for 23% (95%CrI 6%–71%), and diagnosed but untreated individuals for only 2% (95%CrI 0–6%), compared to the respective contributions of these groups of 52% (95%CrI 7–85%), 45% (95%CrI 14–85%), and 3% (95%CrI 1–6%) in 2017. Note that in the model, the category of treated individuals has a very low, but non-negligible infectivity, as it includes not only virally suppressed individuals but also those who are not virally suppressed due to recent treatment initiation, imperfect treatment adherence, treatment failure, or dropout. The estimated HIV prevalence was almost constant, around 6–7% throughout the considered period, which is in the range observed for MSM in Western Europe31. The model was additionally validated using independent data, not included in the model fitting, on the annual number of MSM on PrEP, the annual number of MSM on ART immigrating to the Netherlands from abroad, and the ART coverage among all MSM with HIV from 2017 till 2022 (Fig. S1).
a Incidence of new HIV diagnoses per 100,000 persons per year, b estimated total number of undiagnosed HIV infections, including importations from abroad, c incidence of new HIV infections acquired in the country per 100,000 persons per year, and d contribution to the mean new HIV infections by status of the source of HIV infection. The red dots and the error bars correspond to the mean estimates and the 95% confidence intervals reported by SHM2. The mean trajectories estimated from the model are shown as orange lines. The shaded regions correspond to 95% credible intervals based on 100 samples from the joint posterior parameter distribution.
Cure scenarios
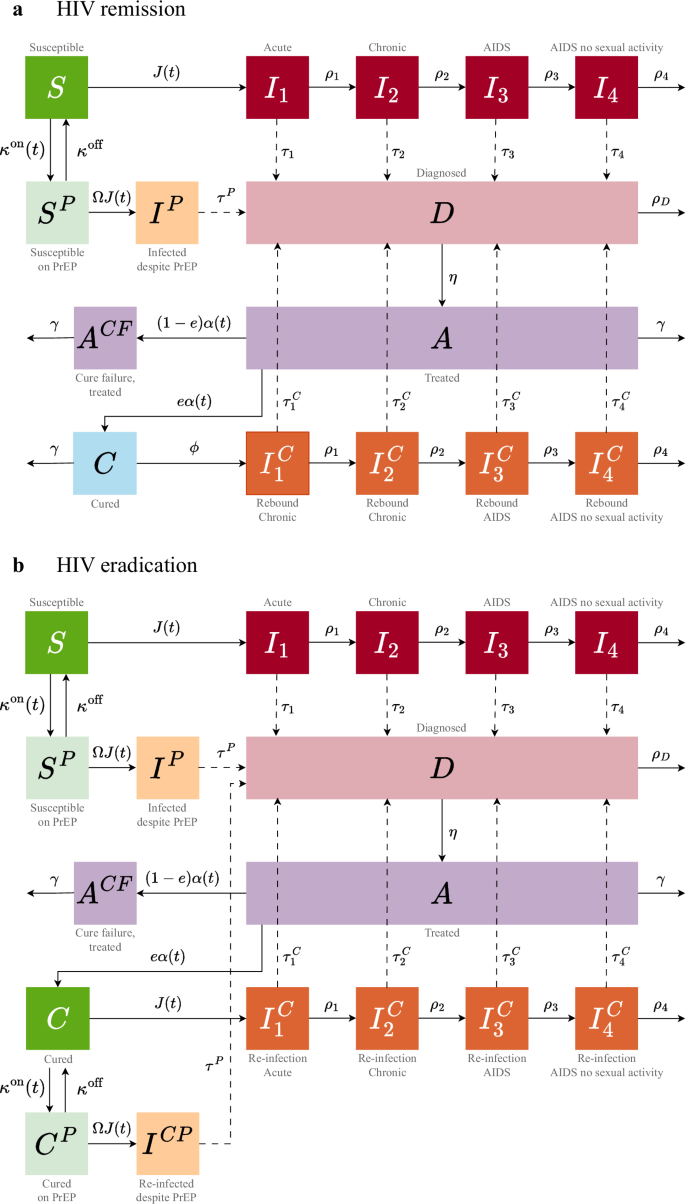
Guided by the TPP9, we developed a transmission model for the HIV remission and HIV eradication scenarios (see Fig. 2 and Methods). The relation of these cure scenarios to the minimum and optimum TPP9 is summarized in Table 1. In both scenarios, the cure was targeted at virologically suppressed individuals on ART (minimum TPP).
Transitions are shown for one risk group. Recruitment into and exit from the sexually active population are not shown. A detailed description of the model equations, parameters, and assumptions for a HIV remission and b HIV eradication is given in the Supplementary Material.
From an HIV dynamics perspective, the key difference between HIV remission and HIV eradication is that, in the remission scenario, the HIV reservoir within an individual is not completely removed. We defined ‘sustained’ HIV remission as a scenario where the virus remains below transmissible levels indefinitely (optimum TPP), whereas ‘transient’ HIV remission allows for possible viral rebound and potential onward transmission after a mean period of virus control of 2 years (minimum TPP). We assumed that individuals achieving HIV remission have full protection against re-infection (optimum TPP). In contrast, in the eradication scenario, all HIV including the intact and rebound-competent reservoir is fully removed. We further assumed that individuals achieving HIV eradication remain susceptible to re-infection (minimum TPP) and could start using PrEP if they have a high risk of HIV acquisition.
In both cure scenarios, we explored the entire range of acceptable values for cure characteristics outlined in the TPP, i.e., annual uptakes (proportion of eligible individuals receiving a cure intervention each year) of 10%, 50%, and 90%, and efficacy (proportion of individuals for whom the intervention was successful) of 20% (minimum TPP) and 90% (optimum TPP). In our model, the cure process is simplified by assuming that cure delivery and ART interruption are instantaneous. Individuals who did not take up a cure intervention or for whom the intervention was unsuccessful remained on ART. Moreover, we considered three monitoring strategies for individuals who may experience a viral rebound after HIV remission or re-infection after HIV eradication: (i) no monitoring (optimum TPP), resulting in a mean (across all HIV stages) diagnostic delay of 28 months, which aligns with the estimated delay of HIV infections under the current standard of care2; (ii) PrEP-like monitoring, with a mean diagnostic delay of 3 months, similar to the testing interval for PrEP users32; and (iii) frequent monitoring (minimum TPP) every two weeks, as used in analytical treatment interruptions studies33.
A cure intervention was assumed to be introduced in 2026, with maximum uptake reached within 3 years. We compared projections of HIV dynamics under the two cure scenarios and the no-cure scenario from 2026 to 2036. Changes in HIV incidence under the cure scenarios were always reported relative to the no-cure scenario. A detailed description of the model equations, parameters, and assumptions is provided in the Methods section, Fig. 2, and the Supplementary Material.
Dynamics for HIV remission scenario
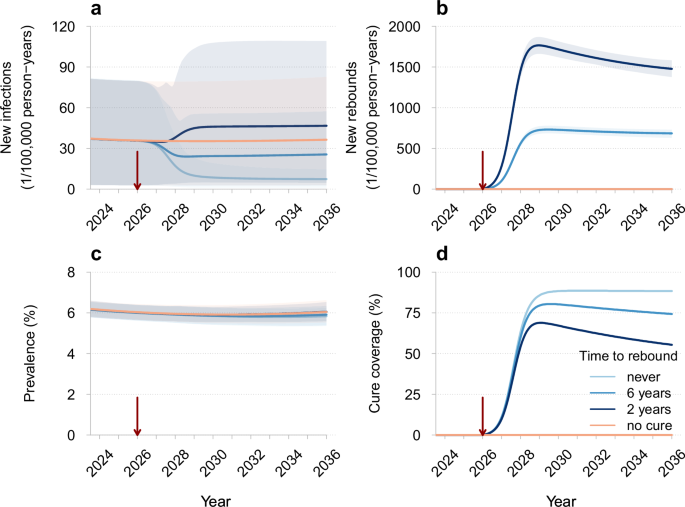
We first explored how the dynamics under the HIV remission scenario might unfold based on different assumptions about the time to viral rebound. We simulated the model with mean rebound times of 2 years and 6 years, as well as the possibility of sustained HIV remission (i.e., no rebound). We assumed 90% intervention efficacy and 90% annual uptake, along with a 3-month diagnostic delay of rebounds. Projections of the epidemic dynamics for these parameters are shown in Fig. 3. Sustained HIV remission, with no risk of rebound, resulted in the fewest new HIV infections compared to the no-cure scenario—7 (95%CrI 1–15) versus 36 (95%CrI 3–83) per 100,000 persons per year in 2036. If the time to viral rebound was 6 years, an estimated 26 (95%CrI 3–57) new HIV infections and 685 (95%CrI 630–730) new rebounds per 100,000 persons per year would occur in 2036. For a shorter rebound time of 2 years, new HIV infections and new rebounds increased to 47 (95%CrI 8–109) and 1,477 (95%CrI 1377–1582) per 100,000 persons per year in 2036. The HIV prevalence, including individuals in whom HIV remission was achieved but the viral reservoir was not fully removed, remained nearly constant regardless of the rebound time, while cure coverage (proportion of eligible individuals achieving HIV remission) was lower for shorter rebound times.
a New HIV infections, b new rebounds in individuals achieving HIV remission, c HIV prevalence (proportion of individuals with HIV), and d cure coverage (proportion of eligible individuals achieving HIV remission) for different times until viral rebound. The legend for different curves shown in d corresponds to all panels. The red vertical arrows indicate the cure introduction. The mean trajectories from the model are shown as solid lines. The shaded regions correspond to 95% credible intervals based on 100 samples from the joint posterior parameter distribution. Different shades of blue correspond to different times until viral rebound. The projections of the model without a cure are shown in orange. Parameters: efficacy of 90% (proportion of individuals for whom the intervention was successful), annual uptake of 90% (proportion of eligible individuals receiving the intervention each year), and a 3-month diagnostic delay of rebounds in individuals who achieved HIV remission.
The contribution of different groups to new HIV infections occurring in the country varied depending on the time to rebound (Fig. S2). For sustained HIV remission, undiagnosed and treated individuals remained the primary sources of new infections. Specifically, in 2036, these groups accounted for 51% (95%CrI 10%–88%) and 45% (95%CrI 5%–89%) of new infections, respectively, compared to 20% (95%CrI 5–67%) and 79% (95%CrI 27–95%) in the no-cure scenario. This suggests that the main effect of sustained HIV remission is a reduction in the contribution to HIV incidence of the population on ART, associated with its reduced relative weight. In the case of transient HIV remission, the contribution of the population on ART to incidence decreased too; however, individuals who were diagnosed but not yet treated, as well as those who experienced viral rebound, became substantial sources of new HIV infections. In 2036, diagnosed but untreated individuals and those with viral rebound accounted for 20% (95%CrI 11–28%) and 27% (95%CrI 14–39%) of new infections for a mean rebound time of 6 years, with even higher proportions, 23% (95%CrI 13–32%) and 32% (95%CrI 18–44%), for a rebound time of 2 years.
The cumulative person-years on ART averted over ten years, relative to the no-cure scenario, also depended on the time to rebound (Fig. S3a). In the sustained HIV remission scenario, ART use was reduced by 73% (95%CrI 72–74%) compared to the no-cure scenario. When the rebound time was 6 years and 2 years, ART use was reduced by 66% (95%CrI 65%–67%) and 56% (95%CrI 54–58%), respectively.
Impact of HIV remission under varied intervention characteristics
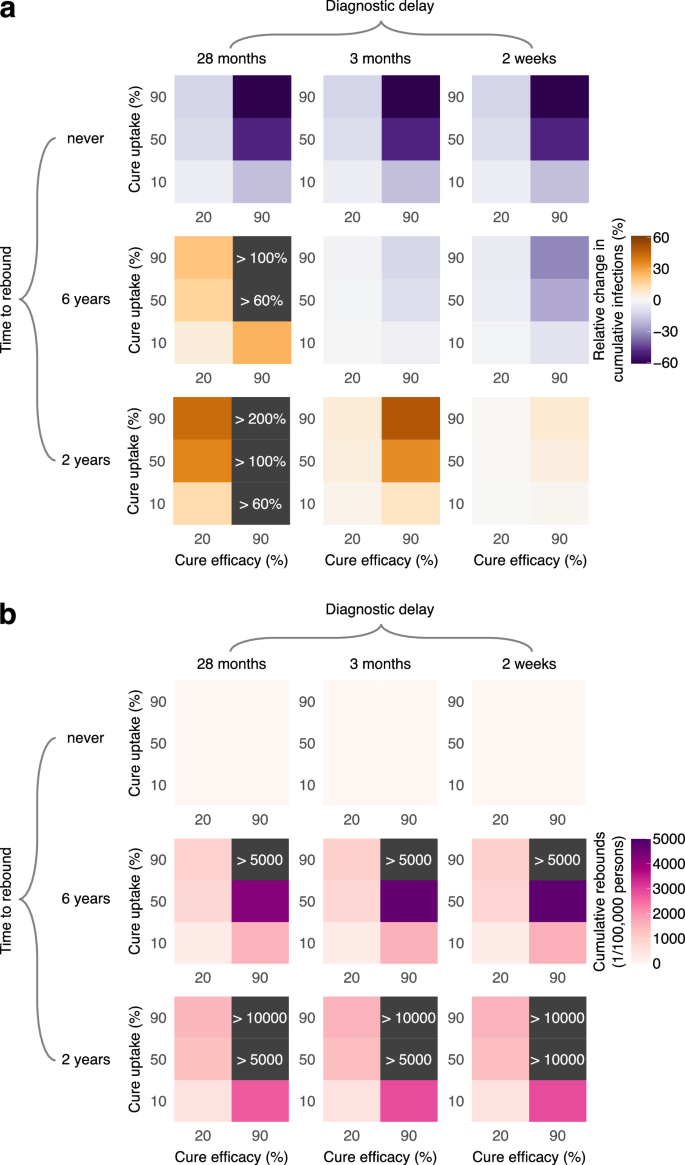
We further systematically compared the impact of HIV remission under varied efficacy, uptake, time to rebound, and diagnostic delay from its introduction in 2026 to 2036 (Fig. 4). Sustained HIV remission consistently resulted in fewer HIV infections, regardless of diagnostic delay (Fig. 4, top rows). Mean reductions in cumulative HIV infections over this period, compared to the no-cure scenario, ranged from 60% (95%CrI 21–72%) for 90% uptake and 90% efficacy to 4% (95%CrI 1–6%) for 10% uptake and 20% efficacy. Intermediate reductions in HIV infections were predicted for all other parameter combinations.
The impact of transient HIV remission on cumulative HIV infections from 2026 to 2036, compared to the no-cure scenario, depended on diagnostic delay and rebound time (Fig. 4, middle and bottom rows). Without a dedicated monitoring strategy for diagnosing rebounds—resulting in a 28-month diagnostic delay, as observed under the current standard of care—this intervention consistently led to an increase in HIV infections. The extent of this increase was greater for shorter rebound times and for higher cure uptake and efficacy. This rise can be attributed to individuals experiencing viral rebound, whose contribution to incidence grows over time, reaching up to 72% (95%CrI 65–84%) in 2036 in the worst-case scenario of a 2-year rebound time, no additional monitoring, and 90% uptake and efficacy (Fig. S4).
In contrast, frequent monitoring of individuals in HIV remission (i.e., a 2-week diagnostic delay of rebounds) led to a reduction in cumulative HIV infections compared to the no-cure scenario for a 6-year rebound time. However, this came with an estimated maximum of 6132 (95%CrI 5770–6484) cumulative rebounds per 100,000 persons. For a shorter 2-year rebound time, even with frequent monitoring, cumulative HIV infections slightly exceeded those in the no-cure scenario, with an estimated maximum of 14,518 (95%CrI 13,611–15,430) cumulative rebounds per 100,000 persons.
We also computed the minimum monitoring frequency required for the HIV remission cure intervention to provide a net benefit in terms of cumulative new infections, using the values of efficacy, uptake, and time to rebound explored in Fig. 4 (see Table S1). The minimum monitoring frequency is approximately every 4.5 to 5 months for all combinations of cure uptake and efficacy, if the mean time to rebound is 6 years. However, if the mean time to rebound is only 2 years, even monitoring as frequently as every two weeks would be insufficient to mitigate the increase in new infections.
Dynamics for HIV eradication scenario
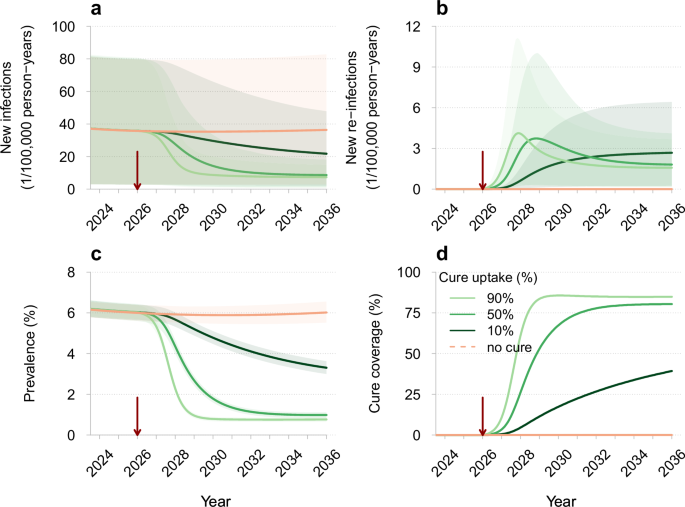
Similar to HIV remission, we first assessed transmission dynamics under the HIV eradication scenario, considering 90% efficacy and a 3-month diagnostic delay for re-infections. The introduction of HIV eradication led to a consistent reduction in the number of new HIV infections in naive individuals, with values varying based on cure uptake (Fig. 5). For 90% uptake, the estimated number of new HIV infections decreased to 7 (95%CrI 1–15) per 100,000 persons per year in 2036, which is comparable to the outcome of sustained HIV remission. For lower uptakes, new HIV infections also declined, albeit less markedly, reaching 9 (95%CrI 1–18) and 22 (95%CrI 2–48) per 100,000 persons per year for 50% and 10% uptake, respectively. Notably, across the entire range of uptakes considered, the estimated number of re-infections in cured individuals remained low, approaching only about 2 re-infections per 100,000 persons per year in 2036. Furthermore, unlike in the HIV remission scenario, HIV prevalence markedly dropped after the introduction of HIV eradication, reaching 3.31% (95%CrI 3.00–3.63%) for 10% uptake and falling to less than 1% for uptakes above 50%. The model also indicated that for lower uptakes, equilibrium in HIV dynamics had not yet been reached, with new HIV infections projected to decline further after 2036 (e.g., falling below 22 per 100,000 persons for 10% uptake).
a New HIV infections (primary infections in naive individuals), b new HIV re-infections (secondary infections in cured individuals), c HIV prevalence (proportion of individuals with HIV), and d cure coverage (proportion of cured individuals among all eligible) for different cure uptakes. The legend for different curves shown in d corresponds to all panels. The red vertical arrows indicate the cure introduction. The mean trajectories from the model are shown as solid lines. The shaded regions correspond to 95% credible intervals based on 100 samples from the joint posterior parameter distribution. Different shades of green correspond to different cure uptakes. The projections of the model without a cure are shown in orange. Parameters: efficacy of 90% (proportion of individuals for whom the intervention was successful) and a 3-month diagnostic delay of re-infections in individuals who achieved HIV eradication.
Similarly to sustained HIV remission, the HIV eradication intervention substantially reduced the contribution of individuals on ART to new HIV infections (Fig. S5). In 2036, this contribution declined from 79% (95%CrI 27–95%) in the no-cure scenario to 44% (95%CrI 5–89%) with a cure uptake of 90%. The contribution of re-infections was negligible. HIV eradication reduced ART use by 73% (95%CrI 72–75%) for a cure uptake of 90% (Fig. S3b), mirroring the effect of sustained HIV remission.
Impact of HIV eradication under varied intervention characteristics
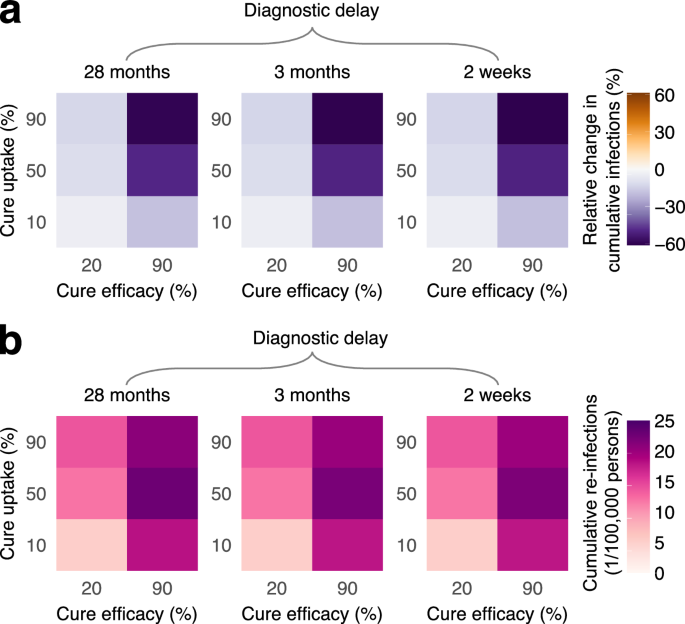
A systematic comparison of the impact of HIV eradication under varied efficacy, uptake, and diagnostic delay of re-infections is shown in Fig. 6. Similar to sustained HIV remission, HIV eradication consistently reduced HIV infections in naive individuals, regardless of diagnostic delay. Like before, the largest and smallest reductions in cumulative HIV infections over the 2026–2036 period were observed at the extreme values of uptakes and efficacy, with the reduction ranging from 4% (95%CrI 1–6%) to 60% (95%CrI 21–72%) for all parameter combinations explored. For all diagnostic delays, the estimated number of cumulative re-infections remained low, not exceeding 23 per 100,000 persons over 10 years.
The color bar corresponds to a mean change in cumulative HIV infections (primary infections in naive individuals) relative to the no-cure scenario and b mean cumulative HIV re-infections (secondary infections in cured individuals) from the introduction of HIV eradication scenario in 2026 to the end of the simulation in 2036. The color bar scale in (a) is the same as that in Fig. 4a for direct comparison.
Robustness and sensitivity analyses
The qualitative trends in projected HIV dynamics from the main analysis hold under alternative assumptions about importation of infected individuals, including a scenario without importation and one where the proportion of undiagnosed imported cases matches that of undiagnosed individuals in the infected population within the country (Figs. S6–S11). Furthermore, our results remained robust across different years of cure introduction (Figs. S12–S13) and when assessed over longer post-cure time frames (Figs. S14–S15). A potential increase in sexual risk behavior among MSM in response to a cure based on survey data did not qualitatively affect the overall outcomes (Fig. S16). Specifically, ten years post-cure, the number of new HIV infections was always lower for HIV eradication compared to the no-cure scenario, but this was not the case for transient HIV remission. Sensitivity analyses regarding the infectivity of individuals after viral rebound indicated that our projections for HIV remission were the most optimistic (Fig. S17). In contrast, the results for HIV eradication were consistent across varying assumptions about the infectivity of re-infections (Fig. S18). In an HIV remission scenario including both rebounds and re-infections (Fig.S19), the results remained consistent with those in Fig. 3, showing an increase in new HIV infections when the time to rebound is two years. The findings in Fig. 3 also hold for a scenario in which HIV remission is modeled with a rebound time following an Erlang distribution instead of an exponential distribution (Fig. S20). Whether an HIV remission cure prolonged survival compared to ART had minimal impact on our results. The qualitative insights into the impact of cure scenarios relative to the no-cure scenario were also robust to potential changes in the underlying trend in HIV incidence, particularly when new HIV infections started to increase from 2022 onward (Fig. S21). Further details of these and additional sensitivity analyses are given in the Supplementary Material.