PCSs impact glucose utilization in monocytes during the induction of trained immunity
Traditional cell culture mediums, such as the Roswell Park Memorial Institute (RPMI) 1640 medium, are designed to promote cell growth and survival ex vivo25. Thus, the concentration of some nutrients in traditional cell culture medium is supraphysiological (Supplementary Fig. 1a). In addition, the conventional medium also lacks most standard culture media, such as lactate, citrate, acetate, β-hydroxybutyrate (βOHB), and pyruvate25. In this study, we referred to those metabolites as PCSs (Supplementary Fig. 1a). Previous studies show that these carbon sources are oxidizable substrates in vivo26,27. Notably, in some situations, these carbon sources exhibit circulatory turnover fluxes exceeding traditional sources, such as glucose and glutamine26,27. Thus, we designed the modified RPMI medium (MM) with approximate polar metabolite concentrations in mouse serum (Supplementary Fig. 1a).
To obtain trained immune cells, we treated purified monocytes with β-glucan for 24 hrs, followed by washing and resting for five days in a culture medium. Then, cells were restimulated with β-glucan or LPS for 24 hrs as previously described24 (Supplementary Fig. 1b). 13C-glucose labeling into lactate and intracellular TCA cycle intermediates, including citrate and fumarate, within 0-8 hrs in β-glucan treated monocytes (Supplementary Figs. 1c). Analysis of metabolite levels in culture medium over time revealed several trends (Supplementary Figs. 1d–1f). As expected, glucose and glutamine were consumed within 12-24 hrs during the trained immunity process, and PCS treatment mildly affected glucose and glutamine consumption in β-glucan treated monocytes (Supplementary Figs. 1d and 1e). A slower decrease of other carbon sources, including OHB, citrate, and pyruvate, was observed in the PCS-supplemented medium compared to the glucose-supplemented medium (Supplementary Fig. 1f). Interestingly, we observed accumulation of several TCA cycle intermediates (αKG and fumarate) in extracellular medium over time, indicated the export of these metabolites (Supplementary Fig. 1f).
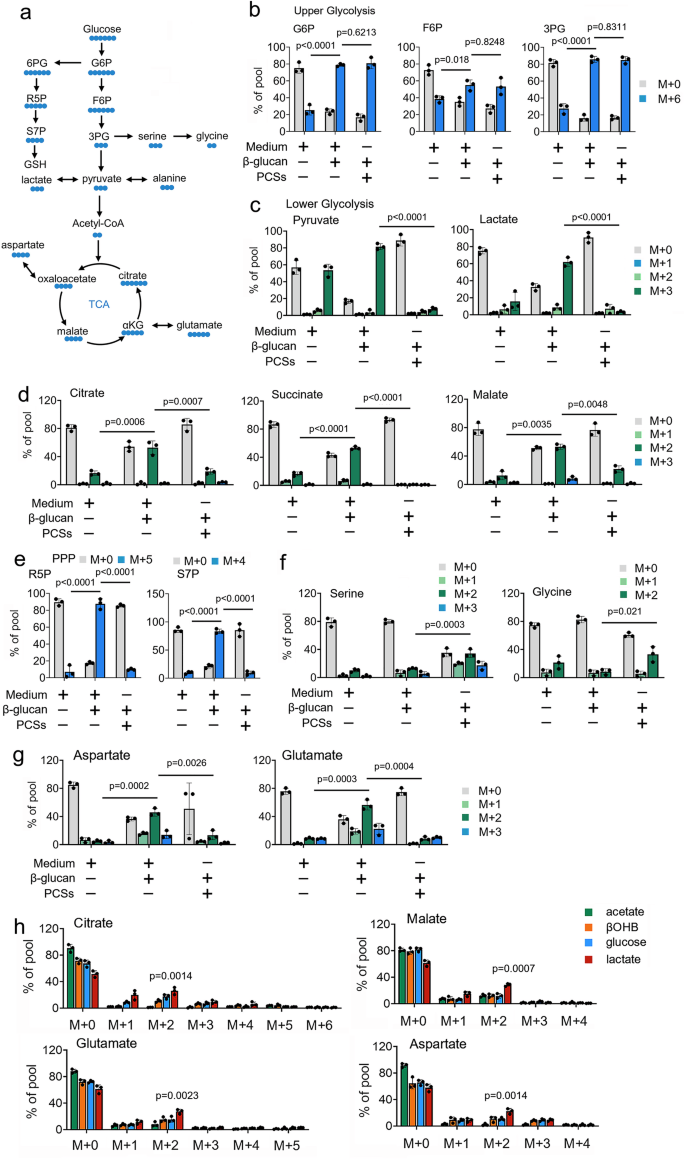
Using 13C-based metabolic tracers, we investigate the role of PCSs on glucose metabolism in trained immunity (Fig. 1a). Mass spectrometry (MS) assay shows that 13C-glucose extensively labels intermediates of glycolysis and the TCA cycle in β-glucan treated monocytes (Figs. 1b-1d). By contrast, in the presence of PCSs at a physiologic concentration in β-glucan treated monocytes, 13C-glucose contribution into TCA cycle intermediates was significantly inhibited (Figs. 1b-1d). Intriguingly, the presence of PCSs at physiologic concentration directs glucose flux to the PPP and the serine synthesis pathway (SSP) (Figs. 1e and 1d). Similar results were obtained in metabolites arising from TCA cataplerosis, such as glutamate and aspartate (Fig. 1g). We observe similar 13C labeling patterns in β-glucan treated monocytes cultured PCSs and 13C-glucose for long (6 days) (Supplementary Fig. 1g). To investigate the contribution of PCSs to TCA cycle metabolism, β-glucan treated monocytes were cultured in MM supplemented with separated 13C-labeled PCSs at physiologic concentrations. We observe that 13C-glucose and 13C-glutamine contributed carbon to the TCA cycle intermediates (Supplementary Fig. 1h). Intriguingly, TCA cycle intermediates were also labeled from some PCSs, including βOHB and lactate, in β-glucan treated monocytes (Supplementary Fig. 1h). Results showed that βOHB and lactate contribute carbon to the TCA cycle by 2-carbon entry via citrate synthase (M + 2 citrate) and carry through the entire cycle in β-glucan treated monocytes (Fig. 1h). Of note, when considering the lower concentration of lactate (1 mM) compared with the concentration of glucose (5 mM), trained monocytes prefer to use PCSs over glucose for TCA cycle metabolism (Fig. 1h). Because it has been reported that alanine did not contribute to the TCA cycle28, the presence of 13C-alanine was included as a negative control for comparison (Supplementary Fig. 1h). Taken together, these findings suggest that PCSs reduced the contribution of 13C-glucose to the TCA cycle, and drives 13C-glucose switch from glycolysis to PPP and SSP, in trained immunity.
a Schematic of 13C-glucose carbon labeling through the TCA cycle, the pentose phosphate pathway (PPP), and the serine synthesis pathway (SSP). b–g Monocytes were cultured in MM containing 13C-glucose (±PCSs) and treated with β-glucan (5 μg/ml) for 4 hrs, followed by mass spectrometry analyzes. h Monocytes were cultured in MM containing the indicated 13C-labeled carbon source and treated with β-glucan (5 μg/ml) for 4 hrs. The relative contributions of 13C from indicated 13C-labeled substrates into metabolites of central carbon metabolism are shown. Data in (b-h) are expressed as means ± SEMs, n = 3 mice per condition, two-way ANOVA. Medium: Phosphate buffered saline. See also Supplementary Fig. 1.
Lactate activates trained immunity
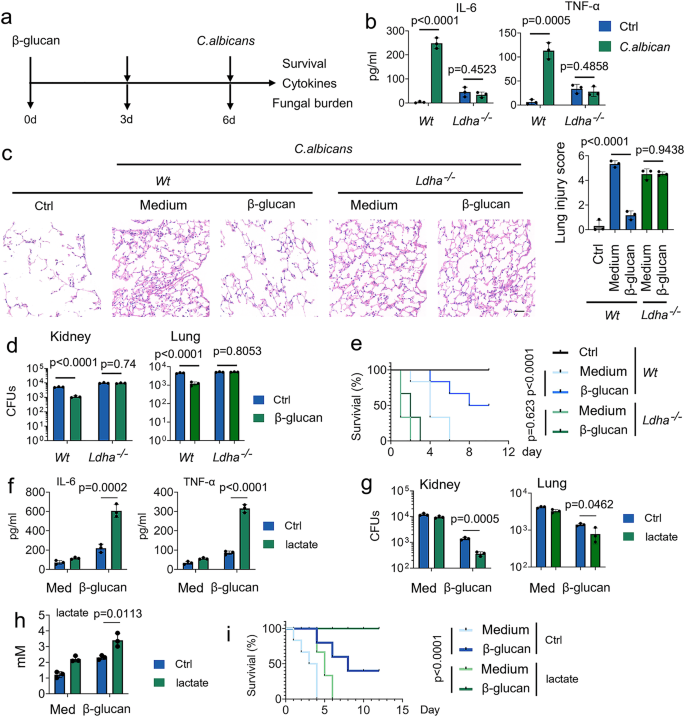
Because among these above PCSs, lactate has the most significant regulation on the TCA cycle (Fig. 1h), we next investigated the role of lactate on trained immunity in response to C. albicans infection (Fig. 2a). In line with a previous study29, C. albicans infection enhances the production of lactate in a time- and dose-dependent manner (Supplementary Fig. 2a). Since lactate dehydrogenase (Ldh) interconverts lactate and pyruvate by using NAD+/NADH as a co-factor30, we asked whether LDHA plays a role in trained immunity. Firstly, we employed the tamoxifen-inducible LDHA knockout mice (LDHA-/-), and LDHA protein expression in diverse organs of LDHA-/- mice was abrogated upon tamoxifen treatment (Supplementary Figs. 2b and 2c). To investigate the role of lactate on trained monocyte function, we trained mice with β-glucan followed by a challenge with C. albicans (Fig. 2a). As expected, IL-6 and TNF-α production was induced in β-glucan-trained Wt mice, but not in LDHA-/- mice (Fig. 2b). Histological analysis of the lung tissue showed that prior β-glucan treatment limits lung damage during subsequent infection (Fig. 2c). Compared with β-glucan-trained Wt mice, more severe lung injury was demonstrated in LDHA-/- mice (Fig. 2c). In this vein, LDHA-/- mice significantly susceptible to C. albicans-induced lethality, accompanied by higher bacterial burden in the lung and kidney tissue (Figs. 2d and 2e). A previous study reported that trained immunity is a protective host against secondary C. albicans infection that relies on monocytes induced by the same pathogen31. Similar results were obtained in C. albicans-trained Wt and LDHA-/- mice (Supplementary Figs. 2d–2g). To determine whether LDHA regulates trained immunity in vitro, we employed two specific small interfering RNAs (siRNAs) for LDHA. We confirmed siRNA efficiency (Supplementary Fig. 2h). SiRNA-LDHA #2 was selected for the following experiments. As shown in Supplementary Fig. 2i, LDHA knockdown inhibits β-glucan-induced expression of IL-6 and TNF-α in human peripheral blood mononuclear cells (PBMCs).
a Schematic representation of in vivo trained immunity experimental setup. b–d Wt and Ldha-/- mice were trained with or without β-glucan. Six days later, mice were infected with or without a secondary C. albicans lethal dose (3 × 106) for 24 hrs, followed by measuring IL-6 and TNF-α levels (b), lung injury(c), and kidney and lung fungal burden (d). e Wt and Ldha-/- mice were trained with or without β-glucan. Six days later, mice were infected with or without a secondary C. albicans lethal dose (3 × 106) for indicated times. Survival curves show data collected until day ten after infection. Statistical analysis was performed using the log-rank test (n = 8 for each group). f–h C57BL/6 mice were trained with or without β-glucan. Six days later, mice were secondarily infected without or with C. albicans (3 × 106) and infused with or without lactate (1 mM) for 24 hrs, followed by measuring IL-6 and TNF-α levels (f), kidney fungal burden (g), and lactate levels in serum (h). i C57BL/6 mice were trained with or without β-glucan. Six days later, mice were infected with or without a secondary C. albicans lethal dose (3 × 106) and infused with or without lactate (1 mM) for indicated times. Survival curves show data collected until day 12 after infection. Statistical analysis was performed using the log-rank test (n = 8 for each group). Data in (b–d, and f–h) are expressed as means ± SEMs, n = 3 mice per condition, two-way ANOVA. See also Supplementary Figs. 2.
Next, we investigated the role of lactate on trained immunity in response to C. albicans infection. As shown in Supplementary Fig. 2j, lactate enhances β-glucan-induced expression of IL-6 and TNF-α in a dose-dependent manner in human PBMCs. Of note, to rule out the possibility that lactate affects trained immunity by changing the acidity13, the pH of the medium was adjusted to an equal level in all in vitro experiments of this manuscript. The presence of lactate increased C. albicans-induced levels of IL-6 and TNF-α, accompanied by lower fungal burden in the kidney and lung tissue (Figs. 2f-2h). Strikingly, lactate increased the protective response of trained monocytes against lethal systemic C. albicans infection (Fig. 2i). These findings suggest that lactate is indispensable for the protective reaction of trained immunity against C. albicans infection.
Lactate is a physiologic fuel during the induction of trained immunity
Next, we investigate the mechanisms by which lactate regulates trained immunity. Firstly, we measured the timeline of 13C-glucose incorporation during β-glucan treatment. β-glucan treated monocytes displayed higher 13C-glucose incorporation into glucose-6-phosphate (G6P), fructose-6-phosphate (F6P), 3-phosphoglycerate (3PG), pyruvate, lactate, citrate, succinate, malate, glutamate and aspartate within 0-4 hrs (Supplementary Figs. 3a–3f), indicates increased glycolytic flow to the TCA cycle. 13C-glucose incorporation flow from oxidative phosphorylation (OxPhos) to SSP and PPP, as well as ATP synthesis and S-adenosylmethionine (SAM) generation, after 4 hrs of β-glucan treatment (Supplementary Figs. 3a–3g). However, 13C-glucose incorporation into glycolysis continued to increase throughout the experiment (Supplementary Fig. 3b). In addition, β-glucan also induced hexokinase 2 (HK2) enzymes and LDHA enzyme activity in a time- and dose-dependent manner (Supplementary Figs. 3h and 3i). Those data suggest β-glucan treated monocytes prefer to use glucose as physiologic fuel early. Still, β-glucan treated monocytes prefer to use glucose for biosynthesis later.
To evaluate the competition between glucose and lactate for TCA cycle metabolism, treated monocytes were cultured with 13C-glucose and unlabeled lactate with gradually increased concentration. Even in the presence of physiologic glucose concentrations (5 mM), β-glucan treated monocytes use low concentrations of lactate (1-2 mM) as a carbon source for intermediates in the TCA cycle (Figs. 3a-3d). By contrast, a low concentration of lactate (1 mM) directs glucose (5 mM) flux from OxPhos to SSP and PPP (Figs. 3a-3f). A substantial fraction of glucose-derived labeled SAM, including m + 5 (via the PPP) and m + 6-9 (via both the PPP and SSP), significantly increased in lactate-treated monocytes (Figs. 3g and 3h), indicates glucose is used for biosynthesis-in the presence lactate. Because lactate contributes to TCA cycle metabolism, we next investigated the role of lactate on trained monocytes bioenergetics. By using Forster resonance energy transfer (FRET)-based ATP sensors targeted to mitochondria (Mito, mtAT1.03), we found that the presence of lactate or β-glucan treatment enhances ATP production (Fig. 3i). The combination of β-glucan and lactate treatment resulted in a significant synergistic induction of ATP production (Fig. 3i). Similarly, the presence of lactate enhances their basal oxygen consumption rate (OCR) and ATP production from OxPhos (Supplementary Fig. 3j). Consistent with 13C metabolite data, lactate increases β-glucan-induced levels of acetyl-CoA, citrate, and malate (Fig. 3j). Finally, we investigate the role of glucose on the lactate contributed TCA cycle metabolism during β-glucan treatment. As shown in Fig. 3k, a low lactate concentration (1 mM) still contributes to TCA cycle intermediates in glucose (1-10 mM) contained culture medium during β-glucan treatment. When the glucose concentration is 25-fold higher than lactate levels, 13C-lactate could not contribute carbon to the TCA cycle (Fig. 3k). These findings suggest lactate is a TCA cycle substrate for trained monocytes under physiologic conditions, even when glucose is available.
a–f Monocytes were cultured in MM containing 13C-glucose (5 mM), indicating unlabeled lactate concentration, and treated with β-glucan (5 μg/ml) for 4 hrs, followed by mass spectrometry analyzes. g Schematic of derivation and contribution of carbon atoms in SAM synthesis. h Experiments were performed as described in (a). 13C-glucose labeling into ATP or SAM is shown. i Monocytes were treated with β-glucan and lactate (1 mM) for 30 min; mitochondrial ATP was measured by the mtAT 1.03 probe. j Monocytes were treated with or without β-glucan and lactate (1 mM) for 12 hrs, then acetyl-CoA, citrate, and malate levels were measured. k Monocytes were cultured in MM containing 13C-lactate (1 mM), indicating unlabeled glucose concentration, and treated with β-glucan (5 μg/ml) for 4 hrs, followed by mass spectrometry analyzes. Data in (a–f and h–k) are expressed as means ± SEMs, n = 3 mice per condition, two-way ANOVA. See also Supplementary Figs. 3.
Lactate fuels the trained immunity via LDHA
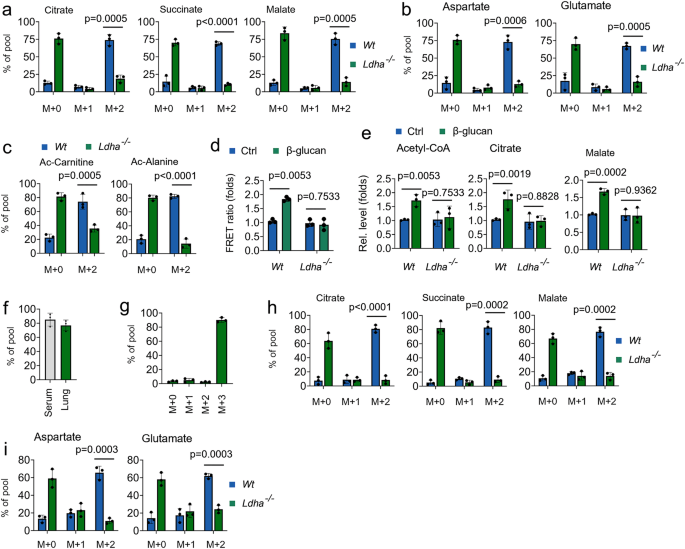
Next, we investigate the role of LDHA on the bioenergetic function of lactate in trained immunity. As shown in Figs. 4a and 4b, decreased abundance of 13C-lactate-derived citrate, succinate, malate, glutamate, and aspartate was observed in LDHA-/- monocytes in response to β-glucan treatment. 13C-lactate also contributed to acetyl-CoA-related metabolites, including Ac-carnitine and Ac-alanine, in β-glucan treated Wt monocytes, but not in β-glucan treated LDHA-/- monocytes (Fig. 4c). By using the mitochondrial ATP probe mtAT, β-glucan treatment enhances and ATP production in Wt monocytes, but not in β-glucan treated LDHA-/- monocytes (Fig. 4d). Consistently, LDHA knockout inhibited β-glucan-induced levels of acetyl-CoA, citrate, malate (Fig. 4e). Sodium oxamate (Oxa) is widely used to inhibit lactate production by regulating the activities of LDHA32. Consistently, β-glucan-induced levels of acetyl-CoA, citrate, and malate were inhibited in Oxa-treated trained monocytes (Supplementary Fig. 4a). We next explored whether the host used lactate as a fuel in trained immunity in vivo by infusing 13C metabolite. In our in vivo system, ~80% enrichment of fully labeled (M + 3) 13C-lactate in serum and lung (Figs. 4f and 4g). Consistent with ex vivo data, infused 13C-lactate readily labeled TCA cycle-derived metabolites in vivo in response to β-glucan treated Wt mice, but not in LDHA-/- mice (Figs. 4h and 4i). We further investigated β-glucan-regulated glucose metabolism in lactate metabolism inhibited system. Intriguingly, β-glucan treatment directs glucose flux from PPP and SSP back to OxPhos when lactate metabolism is inhibited (Supplementary Figs. 4b–4f), suggesting glucose is an alternative carbon source for the TCA cycle in trained immunity. These findings indicate that lactate processing via LDHA is a critical metabolic node in trained immunity.
a–c Monocytes from Wt and Ldha-/- mice were cultured in MM containing 13C-lactate (1 mM) and treated with β-glucan (5 μg/ml) for 12 hrs, followed by mass spectrometry analyzes. d Monocytes from Wt and Ldha-/- mice were cultured in lactate media (0.5 mM glucose, 0.5 mM glutamine, and 2 mM lactate), and treated with β-glucan for 30 min, mitochondrial ATP was measured by the mtAT 1.03 probe. e Monocytes from Wt and Ldha-/- mice were treated with or without β-glucan and lactate (1 mM) for 12 h, followed by measuring acetyl-CoA, citrate, and malate levels. f, g C57BL/6 mice were trained with or without β-glucan. Six days later, mice were infused with 13C-lactate for 12 hrs, followed by mass spectrometry analyzes. h, i Wt and Ldha-/- mice were trained with or without β-glucan. Six days later, mice were infused with 13C-lactate for 12 hrs; monocytes were isolated and followed by mass spectrometry analyzes. All data are expressed as means ± SEMs, n = 3 mice per condition, two-way ANOVA. See also Supplementary Fig. 4.
Lactate regulates histone lactylation via LDHA in monocytes during the induction of trained immunity
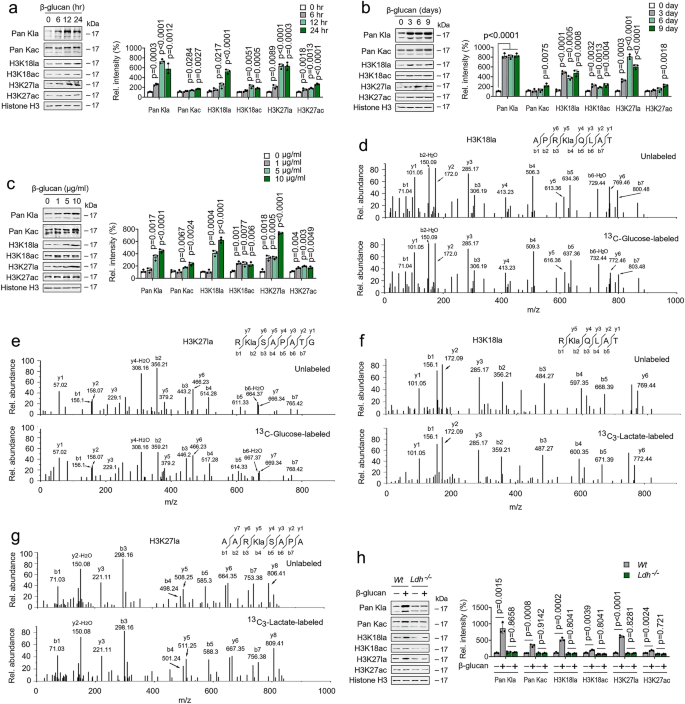
A previous study showed that lysine lactylation (Kla) is a new type of histone mark that can be stimulated by lactate15 (Supplementary Fig. 5a); we suspected that β-glucan regulated high level of lactate impact on histone lactylation in trained immunity. Immunoblots analysis revealed that histone Kla levels were increased; however, histone acetylation (Kac) levels were mildly increased, in a time-dependent fashion in β-glucan treated monocytes (Fig. 5a and Supplementary Fig. 5b). Histone Kla levels elevation was detected as early as 6 hrs after β-glucan treatment (Fig. 5a and Supplementary Fig. 5b). Intriguingly, high histone Kla levels were maintained from days 1 to 9 (Fig. 5b and Supplementary Fig. 5c). As expected, β-glucan challenge induce histone Kla levels, mildly induce Kac levels, in dose-dependent manner (Fig. 5c and Supplementary Fig. 5d). Given that replenishing cells with fresh media did not affect Kla levels, the increase of histone Kla during trained immunity is intrinsic and not due to paracrine effects (Supplementary Fig. 5e). Consistent with previous results15, MS/MS analysis showed that a mass shift of 72.021 Da on lysine residues in the histone was observed in β-glucan treated monocytes (Supplementary Figs. 5f and 5g). In this vein, metabolic labeling experiments using 13C-glucose or 13C-lactate followed by MS/MS analysis demonstrated that β-glucan promotes histone lactylation directly from glucose or exogenous lactate (Figs. 5d-5g). In addition, β-glucan treatment increased histone Kla levels in Wt monocytes but not in LDHA-/- monocytes (Fig. 5h and Supplementary Fig. 5h). These findings suggest high intracellular lactate levels in trained immunity-regulated histone lactylation.
a, b Monocytes were treated with or without β-glucan (5 μg/ml) for the indicated time before western blotting. Quantification of the immunoblots is shown on the right panel. c Monocytes were treated with or without β-glucan at indicated concentrations for 24 hrs before western blotting. Quantification of the immunoblots is shown on the right panel. d, e MS/MS spectra of a 13C-glucose labeled histone Kla peptide and its unlabeled counterpart from β-glucan treated monocytes. f, g Experiments were performed as described in (d,e), except 13C-lactate was used. h Monocytes from Wt and Ldha-/- mice were treated with or without β-glucan (5 μg/ml) for 24 hrs before western blotting. Quantification of the immunoblots is shown on the right panel. All experiments were repeated at least three times. Data in (a-c and h) are expressed as means ± SEMs, n = 3 mice per condition, two-way ANOVA. See also Supplementary Fig. 5.
Lactate maintains chromatin accessibility via histone lactylation in monocytes during the induction of trained immunity
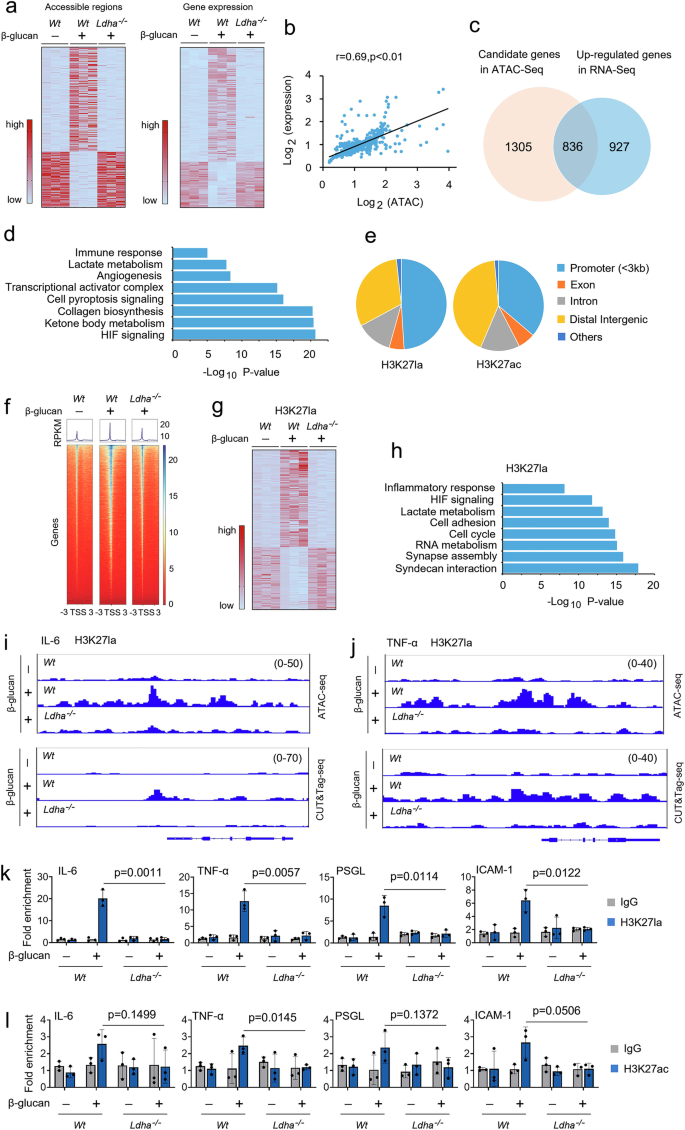
To investigate the mechanical effect of lactate production on trained immunity, we performed the Assay for Transposase-Accessible Chromatin with high throughput sequencing (ATAC-seq) analysis to compare chromatin region accessibility changes between Wt monocytes, Wt, and LDHA-/- trained monocytes. We identified 2,848 chromatin regions exhibiting accessibility changes (Fig. 6a). To evaluate the functional impact of lactate on transcription, we performed RNA-seq in Wt monocytes, Wt, and LDHA-/- trained monocytes in biological duplicates (Fig. 6a). We first explored the relationship between changes in chromatin accessibility and gene expression. We assigned each differentially accessible regions (DARs) to its nearest transcription start site and examined the gene expression differences. We observed significant correlations between changes in chromatin accessibility and expression of the neighboring gene (Fig. 6b). Next, we identified 836 genes that were upregulated in Wt monocytes, Wt, and LDHA-/- trained monocytes, which were also marked with chromatin accessibility alterations (Fig. 6c). Gene ontology (GO) biological process analysis of these genes revealed significant enrichment of biological pathways, including immune response, lactate metabolism, and HIF signaling (Fig. 6d). We choose H3 lysine 27 (H3K27la), this lysine residue is also acetylated (H3K27ac), to elucidate the potential functional significance of histone lactylation in trained immunity by using CUT&Tag, a novel genome-wide immunotethering assay. Genome-wide distribution analysis reveals that H3K27la and H3K27ac histone modifications were located mainly within the promoter regions (Fig. 6e). As anticipated, H3K27la peaks were increased near transcription start sites (TSSs) in Wt-trained monocytes, but not LDHA-/- trained monocytes (Fig. 6f). However, H3K27ac peaks were slightly increased near TSSs in Wt-trained monocytes compare with Wt non-trained monocytes (Supplementary Fig. 6a). We compared the expression of H3K27la-marked genes in Wt non-trained monocytes, Wt and LDHA-/- trained monocytes. Among the total 7,547 shared peaks between the three groups, we obtained 248 peaks that were significantly changed (Fig. 6g). GO pathway analysis revealed that biological processes such as inflammatory response, lactate metabolism, and cell adhesion, which is consistent with ATAC-seq data (Fig. 6h). To test the role of H3K27la and H3K27ac in the function of trained monocytes, we integrated our ATAC-seq datasets and CUT&Tag datasets into the Integrative Genomics Viewer to examine their binding profiles on the genes. As shown in Supplementary Fig. 6b, H3K27ac peaks were slightly enhanced at the promoters of IL-6 and TNF-α in Wt-trained monocytes compared with Wt non-trained monocytes. However, H3K27la peaks were highly enriched at the promoters of critical genes of inflammatory factors and cell adhesion molecules, including IL-6, TNF-α, PSGL, and ICAM-1, in Wt-trained monocytes, but not in LDHA-/- trained monocytes (Figs. 6i, 6j and Supplementary Figs. 6c and 6d). We then used a chromatin immunoprecipitation (ChIP)-qPCR assay to assess the effects of H3K27la and H3K27ac on the stabilized effector gene expression. Indeed, H3K27la recruitment to the promoter regions of IL-6, TNF-α, PSGL, and ICAM-1 was increased in Wt-trained monocytes but not in LDHA-/- trained monocytes (Fig. 6k). Only minor differences existed between the from the H3K27ac recruitment to the promoter regions of IL-6, TNF-α, PSGL, and ICAM-1 in Wt-trained monocytes compare with the Wt non-trained monocytes (Fig. 6l). The role of histone modifications on the activation of the promoter is not restricted to one lysine, as similar results were obtained by H3K18la and H3K18ac (Supplementary Figs. 6e and 6f). In line with these observations, blocking CBP/p300 histone acetyltransferase (HAT) activity with HAT inhibitor (HATi) also decreased H3K27la and H3K18la interaction with the promoter of IL-6, TNF-α, PSGL, and ICAM-1 (Supplementary Figs. 6g and 6h). These findings suggest that intrinsic lactate production alters acetylation-dependent epigenetic programming at the promoter of inflammatory factors and cell adhesion molecules in trained monocytes.
a Heatmap representing the differentially accessible regions (DARs) of ATAC-seq (left panel) and differential gene expression of RNA-seq (right panel) in monocytes from Wt monocytes, Wt trained monocytes, and Ldha-/- trained monocytes trained as described in Fig S1b. b DARs of ATAC-seq and differential gene expression of RNA-seq subjected to Pearson’s correlation analysis. Correlation coefficient (r) and P values were calculated by Pearson’s correlation analysis. c Venn diagram depicting the number of candidate genes in ATAC-seq and upregulated genes in RNA-seq of Wt monocytes, Wt trained monocytes, and Ldha-/- trained monocytes. d GO analysis of significantly different genes in (c). The most significant and nonredundant biological processes are shown, with respective gene numbers and P values. P values by two-tailed Student’s t-test. e Pie chart showing the distribution of H3K27la and H3K27ac at annotated genomic regions in trained monocytes. f Heatmap showing the genomic occupancy of H3K27la ± 3 kb flanking TSSs in trained monocytes from Wt and Ldha-/- mice. The genes shown in rows are sorted in descending order by signal strength. g Heatmap of chromatin peak accessibility for significantly different peaks in trained monocytes from Wt and Ldha-/- mice. Each row represents a Z score of log2-transferred normalized read counts within each sample using CUT&Tag-seq. h GO analysis of significantly different genes in (g). Most significant and nonredundant biological processes with respective gene numbers and P values are shown. P values two-tailed Student’s t-test. i, j Representative ATAC-seq and CUT&Tag-seq genome browser tracks for H3K27la peak enrichment at IL-6 and TNF-α gene loci for trained monocytes from Wt and Ldha-/- mice. k, l Monocytes from Wt and Ldha-/- mice were trained as described in Fig S1b, followed by ChIP analysis. Data in (k and l) are expressed as means ± SEMs, n = 3 mice per condition, two-way ANOVA. See also Supplementary Fig. 6.
Exogenous lactate supports trained immunity through contribution to the TCA cycle and histone lactylation
Intracellular lactate can be produced by LDHA or imported by monocarboxylate transporter 1 (MCT1; also known as SLC16A1)12. Thus, we sought to explore the effect of MCT1 on the trained immunity. As shown in Supplementary Fig. 7a, IL-6, and TNF-α production was induced in β-glucan-trained Wt mice, but not in MCT1 inhibitor AZD3965 (AZD) treated mice. In this vein, AZD–treated mice were significantly susceptible to C. albicans-induced lethality, with higher bacterial burden in the lung and kidney tissue (Supplementary Figs. 7b and 7c). Next, we evaluate the role of MCT1 on the lactate-fueled TCA cycle by treating trained monocytes with 13C-lactate. As shown in Supplementary Figs. 7d–7f, AZD treatment abolished lactate entry into the TCA cycle. As expected, β-glucan treatment increased histone Kla levels in Wt monocytes but not in AZD-treated monocytes (Supplementary Fig. 7g). ChIP-qPCR assay indicated that AZD treatment inhibit H3K27la and H3K18la recruitment to the promoter regions of IL-6, TNF-α, PSGL, and ICAM-1 (Supplementary Figs. 7h and 7i). Hence, MCT1-mediated lactate uptake service as a TCA cycle substrate regulated cytokine production via histone lactylation.
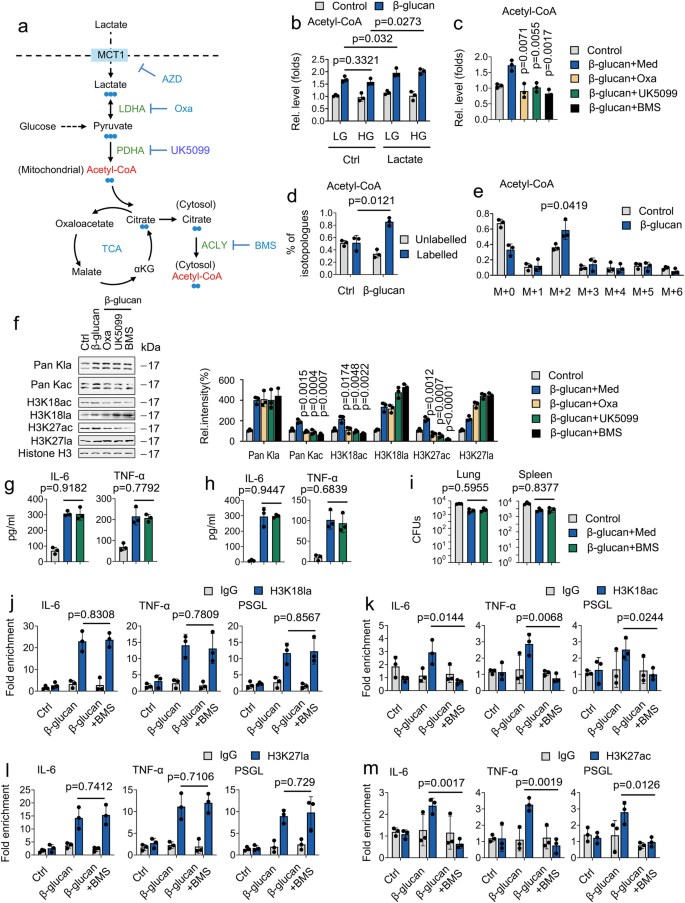
Our data revealed that β-glucan mildly induced histone acetylation. We hypothesized that lactate is taken up and readily converted to pyruvic acid through LDHA and subsequently converted to cytosolic acetyl-CoA in two enzymatic catalyzed reactions by pyruvate dehydrogenase (PDHA) and ATP-citrate lyase (ACLY), respectively33 (Fig. 7a). To this purpose; we compare the role of glucose and lactate on acetyl-CoA generation. As shown in Fig. 7b, neither high glucose (HG) nor low glucose (LG) affect β-glucan-regulated acetyl-CoA generation-however, lactate treatment induced β-glucan regulated acetyl-CoA generation, indicating that β-glucan promotes acetyl-CoA generation via lactate. Still, not glucose (Fig. 7b). To dissect the key step of lactate metabolism involved in acetyl-CoA generation, we employed a series of pharmacologically targeting LDHA, PDHA and ACLY by the LDHA inhibitor (sodium oxamate, Oxa), pyruvate transporter inhibitor (UK5099), and ACLY inhibitor (BMS303141, BMS), respectively (Fig. 7a). Interesting, treatment of Oxa, UK5099, and BMS inhibit β-glucan induced acetyl-CoA generation (Fig. 7c). LC/MS analysis focused on isotopologues of acetyl-CoA demonstrate that lactate carbons labeled the majority of acetyl-CoA. The m + 2 acetyl-CoA isotopologue revealed the most substantial labeling pattern derived from lactate carbons (Figs. 7d and 7e). To pinpoint the pathway in how lactate facilitates histone acetylation, we hypothesized that this involves oxidative metabolism, particularly ACLY. Strikingly, BMS counteracted β-glucan-mediated histone acetylation but not histone lactylation (Fig. 7f). Surprising, the ACLY inhibitor did not affect β-glucan-mediated IL-6 and TNF-α production, and bacterial burden in the lung and kidney tissue in vitro and in vivo (Figs. 7g-7i). In addition, BMS treatment inhibits H3K27ac and H3K18ac, but not H3K27la and H3K18la, recruitment to the promoter regions of IL-6, TNF-α, and PSGL (Figs. 7j-7m). These results suggest that lactate is a significant substrate for acetyl-CoA production. Compared with lactate-mediated histone acetylation, lactate-mediated histone lactylation plays a dominant role in trained immunity.
a Schematic of 13C-lactate carbon labeling through the TCA cycle. b Monocytes were cultured with high glucose (HG) (15 mM), low glucose (LG) (3 mM), and/or lactate (1 mM), and treated with or without β-glucan (5 μg/ml) for 24 hrs, followed by measuring acetyl-CoA levels. c Monocytes were cultured in lactate media (0.5 mM glucose, 0.5 mM glutamine, and 2 mM lactate) and treated with or without β-glucan (5 μg/ml), sodium oxamate (20 mM), UK5099 (10 μM), BMS303141 (BMS) (10 μM) for 24 hrs, followed by measuring acetyl-CoA levels. d, e Monocytes were cultured in MM containing 13C-lactate (1 mM) and treated with β-glucan (5 μg/ml) for 4 hrs, followed by LC/MS analysis. Shown are labeled and unlabeled fractions of acetyl-CoA. f Monocytes were cultured in lactate media (0.5 mM glucose, 0.5 mM glutamine, and 2 mM lactate) and treated with or without β-glucan (5 μg/ml), sodium oxamate (20 mM), UK5099 (10 μM), BMS (10 μM) for 24 hrs before western blotting. Quantification of the immunoblots is shown on the right panel. Experiments were repeated at least three times. g Monocytes were cultured in lactate media (0.5 mM glucose, 0.5 mM glutamine, and 2 mM lactate), treated with or without β-glucan (5 μg/ml), BMS (10 μM) for 24 hrs, and then IL-6 and TNF-α levels were measured. h, i Wt mice were trained with or without β-glucan. Six days later, mice were infected with or without a secondary C. albicans lethal dose (3 × 106) and/or BMS (50 mg/kg) for 24 hrs, followed by measuring IL-6 and TNF-α levels (h) and fungal burden (i). j–m Monocytes from Wt mice were treated as described in (g), followed by ChIP analysis. Data are expressed as means ± SEMs, n = 3 mice per condition, two-way ANOVA. See also Supplementary Fig. 7.