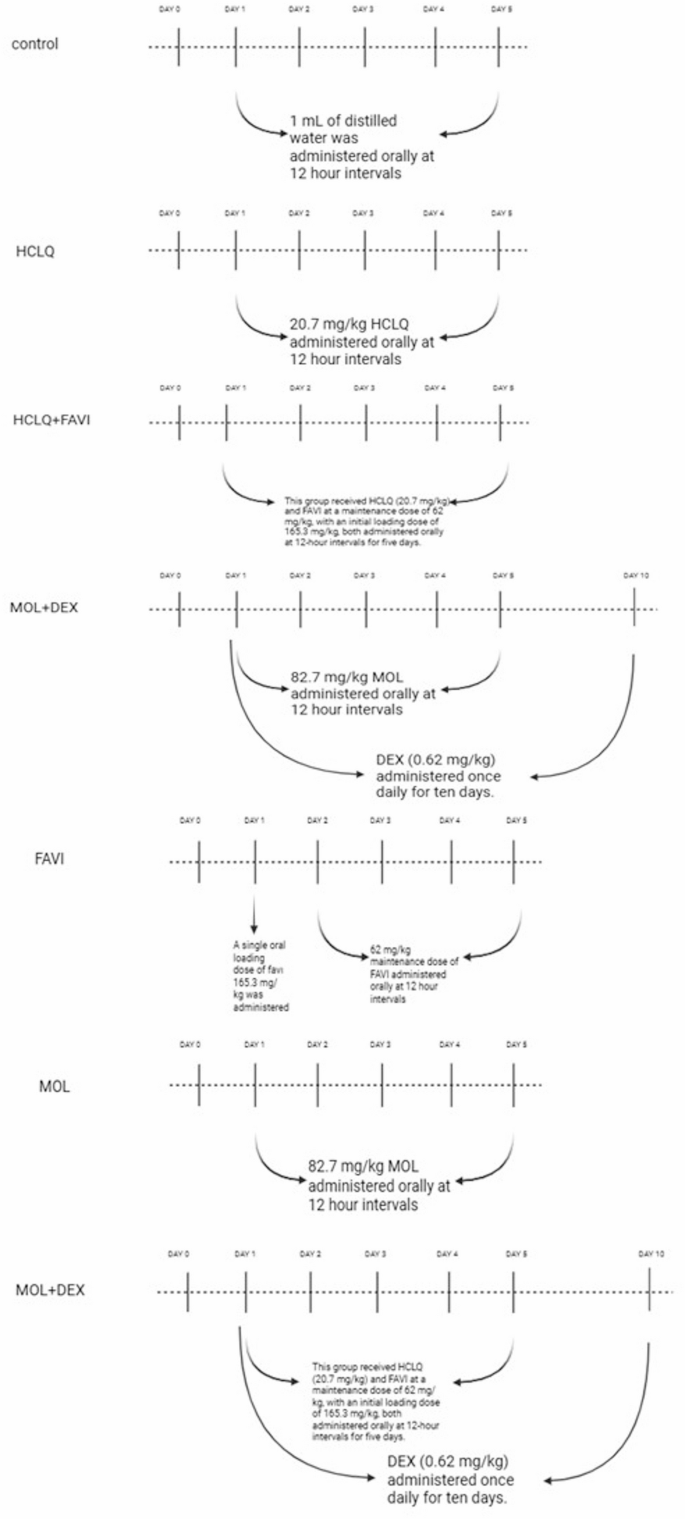
Animals and experimental procedure
This study was conducted on male Wistar albino rats that were three months old, with body weights ranging from 250 to 350 g. The animals were obtained from the Inonu University Laboratory Animals Research Center. All procedures related to animal care and experimentation accordance with the National Institutes of Health Animal Research Guidelines and the ARRIVE Guidelines19. Ethical approval for the study was granted by the Animal Research Ethics Committee of Inonu University, Faculty of Medicine (Protocol: 2021/1–6).
The rats were kept under controlled environmental conditions, including a 12-hour light/dark cycle, a temperature of 22 ± 2 °C, and humidity levels maintained at 60 ± 5%. They were fed a standard pellet diet and provided with unlimited access to tap water. A priori power analysis was conducted using G*Power software (version 3.1.9.7) to determine the minimum required sample size for comparing seven independent experimental groups using one-way ANOVA. The analysis was based on the following assumptions:
Effect size (f): 0.55 (considered a large effect according to Cohen’s criteria).
Alpha error probability (α): 0.05.
Statistical power (1–β): 0.80.
Number of groups: 7.
Based on these parameters, the total required sample size was calculated to be 56 animals, indicating that each group should include at least 8 rats. Accordingly, sample sizes were determined to meet this minimum requirement. After a one-week adaptation period, the animals were randomly divided into seven experimental groups as follows (Fig. 1):
Control Group (n = 8): Rats received 1 mL of distilled water orally as a vehicle solution every 12 h for five consecutive days.
HCLQ Group (n = 10): Rats were administered HCLQ at a dose of 20.7 mg/kg (Plaquenil® 200 mg tablet, SANOFİ Sağlık Ürünleri Ltd. Şti, İstanbul, Turkey) by oral gavage every 12 h for five days.
FAVI Group (n = 10): Rats received FAVI (Favimol® 200 mg film-coated tablet, Neutec İlaç Sanayi Ticaret A.Ş., Sakarya, Turkey) orally at 12-hour intervals for five days. The first dose was a loading dose of 165.3 mg/kg, followed by a maintenance dose of 62 mg/kg.
MOL Group (n = 10): Rats were given MOL at a dose of 82.7 mg/kg (Covunavir® 200 mg capsule, Abdi İbrahim İlaç Sanayi ve Tic. A.Ş., İstanbul, Turkey) orally every 12 h for five days.
HCLQ + FAVI Group (n = 10): This group received HCLQ (20.7 mg/kg) and FAVI at a maintenance dose of 62 mg/kg, with an initial loading dose of 165.3 mg/kg, both administered orally at 12-hour intervals for five days.
MOL + DEX Group (n = 10): Rats were treated with MOL (82.7 mg/kg) every 12 h for five days, along with DEX (0.62 mg/kg) administered intramuscularly once daily for ten days.
HCLQ + FAVI + DEX Group (n = 10): Rats in this group received HCLQ (20.7 mg/kg) and FAVI at a loading dose of 165.3 mg/kg, followed by a maintenance dose of 62 mg/kg, both given orally every 12 h for five days. Additionally, DEX (0.62 mg/kg) was administered intramuscularly once daily for ten days (Dekort® 8 mg/2 mL injectable solution, Deva Holding A.Ş., İstanbul, Türkiye).
The human-equivalent doses of each drug were selected based on reported clinical protocols used during the COVID-19 pandemic. HCLQ was administered at a dose adapted from the 400 mg/day treatment commonly employed early in the pandemic15. FAVI dosage was based on the protocol using an initial loading dose of 1600 mg twice daily, followed by 600 mg twice daily11. MOL dosing was adapted from the clinical dose of 800 mg twice daily for 5 days4. DEX was administered based on the RECOVERY trial dosing of 6 mg/day18. These human doses were converted to equivalent rat doses using the body surface area normalization method as described by Nair and Jacob20. On the 14th day following drug administration and completion of the experimental procedure, one of the carotid arteries was cannulated to measure systolic, diastolic, and mean blood pressure (BP) as well as heart rate (HR) under anesthesia with ethyl carbamate (Urethane®, Sigma-Aldrich, Inc., St. Louis, MO, United States) at a dose of 1.2 g/kg administered intraperitoneally. To monitor electrocardiographic (ECG) changes, three-lead ECG electrodes were utilized. BP, HR, and ECG data were recorded using the Biopac MP-100 Data Acquisition System and its computer-based recording software. This method is consistent with our previously published approach in experimental cardiovascular pharmacology studies21,22. The type of arrhythmia was identified, and PR, QRS, and QT intervals were analyzed from the recorded data in accordance with the Lambeth Convention criteria23.
After the completion of the experimental protocol, all animals were euthanized under ethyl carbamate anesthesia by surgical exsanguination. The body, heart and lung weights of rats were weighed and autopsies were performed on the heart, descending aorta, lung and kidney tissues. To maintain anatomical consistency and minimize regional morphological variability, tissue samples were always collected from the same predefined regions of each organ. For the heart, samples used for biochemical analysis were taken from the free wall of the right ventricle, whereas samples used for histopathological and immunohistochemical examinations were obtained from the anterior wall of the left ventricle. These regions were selected because they are standard reference sites widely used in experimental cardiology and provide reproducible tissue characteristics. Similarly, for the lung and aorta, all animals were sampled from equivalent lobes and anatomical segments to ensure uniformity across groups. This standardized sampling approach ensured that inter-group differences reflected treatment effects rather than intrinsic anatomical variations. Histopathologic and immunohistochemical findings related to renal tissue were presented in the study published by Yildiz et al.24. A piece of heart, descending aorta and lung tissues were evaluated under a light microscope for biochemical, histopathological and immunohistochemical examinations after appropriate staining and follow-up.
Histopathological analysis
At the end of the experiment, heart, vascular and lung tissues were fixed in 10% formaldehyde. After tissue tracing, 4 μm thick sections were taken from the prepared paraffin blocks. The tissue sections were stained with the hematoxylin-eosin (H-E) staining method to determine the general morphologic structure. Heart sections were evaluated for congestion-hemorrhage, infiltration, interstitial edema and cardiomyocyte degeneration (dense eosinophilic cytoplasm and pyknotic nuclei). Ten randomly selected areas were examined and the areas were scored according to the severity of histologic changes as 0: no change, 1: mild, 2: moderate, 3: severe. In the evaluation for the aorta, tunica intima-media thickness was measured by randomly selecting 5 areas from each section. In addition, the whole area was examined and histologic changes observed in the aorta wall (thinning of elastic lamellae, loss of myofibrils in smooth muscle cells) were scored according to severity as 0: no change, 1: mild, 2: moderate, 3: severe change. Lung sections were evaluated for infiltration, alveolar macrophage density, alveolar septa thickening and interstitial edema. Ten randomly selected areas were examined and the areas were scored according to the severity of histologic changes as 0: no change, 1: mild, 2: moderate, 3: severe change. Analyses were performed with a Leica DFC-280 research microscope using the Leica Q Win Image Analysis System (Leica Micros Imaging Solutions Ltd., Cambridge, UK).
Immunohistochemical analysis
For immunohistochemical analysis, deparaffinized and rehydrated sections were placed in a pressure cooker and boiled in 0.01 M citrate (pH 6.0) for 15–20 min. The sections were treated with 3% hydrogen peroxide for 12 min to block endogenous peroxidase enzyme activity. After the protein block (ultra V block) was applied to the sections washed with PBS for 5 min, the sections were incubated with primary antibody (vimentin, caspase-3, RIPK3: Santa Cruz) for 60 min at 37 ˚C. The tissues were washed with PBS and treated with biotinylated secondary antibody at 37 ˚C for 10 min. After this process, the sections were incubated with streptavidin peroxidase at 37 ˚C for 10 min. The chromogen-treated sections were then stained with hematoxylin and covered with water-based sealer. Staining was scored semiquantitatively based on the extent (0: 0: 0–25%, 1: 26–50%, 2: 51–75%, 3: 76–100%) and severity (0: absent, + 1: mild, + 2: moderate, + 3: severe) of immunoreactivity. The total staining score was obtained by calculating prevalence x severity.
Tissue biochemical analysis
For biochemical analyses, tissue samples were removed immediately after euthanasia, washed with ice-cold phosphate-buffered saline (PBS, pH 7.4, 50 mM; BioShop, Burlington, Canada) to remove excess blood and debris, dried, and weighed. Each tissue sample was then transferred to pre-chilled homogenization tubes containing the appropriate volume of ice-cold homogenization buffer (PBS; pH 7.4, 50 mM) supplemented with a protease inhibitor cocktail (Sigma-Aldrich, St. Louis, MO, USA) to prevent protein degradation. Homogenization was performed on ice using an Ultra Turrax T25 mechanical homogenizer (IKA Labortechnik; IKA-Werke, Staufen, Germany) at 12,000 rpm for 30 s until a homogeneous suspension was obtained. Afterward, the tissues were blended in a homogenizer for 3 × 15 s and sonicated by the Bandelin Sonopuls HD 2070 ultrasonic homogenizer (3 cycles, 15-s pulses for each cycle; 1-min intervals between pulses; Bandelin Electronic GmbH & Co. KG, Berlin, Germany). The homogenates were then centrifuged (Hettich Universal 320R, Germany) at 14,000 rpm for 15 min at 4 °C. The resulting supernatants were carefully collected and stored at −80 °C until further biochemical analyses. Protein concentrations in the supernatants were measured using the Bradford method to ensure equal protein loading in subsequent analyses25. Bovine serum albumin (CAS Number: 9048-46−8, Sigma-Aldrich, St. Louis, MO) was used as the standard protein. All procedures were performed under cold conditions to preserve enzymatic activity and prevent degradation of target molecules. Interleukin-6 (IL-6), tumor necrosis factor (TNF-α), myeloperoxidase (MPO), nitric oxide (NO), total oxidant status (TOS), and total antioxidant status (TAS) were measured in heart, descending aorta, and lung tissues using Enzyme-Linked Immunosorbent Assay (ELISA) kits. IL-6 (Cat No.: E0135Ra), TNF-α (Cat No.: E0082Hu), MPO Cat No.: E0436Mo, and NO (Cat No.: SH0030) kits were purchased from Bioassay Technology Laboratory (BT-Lab; Shanghai, China). TOS and TAS kits were provided by Rel Assay (Rel Assay Diagnostics kit, Mega Tip, Gaziantep, Turkey).
Statistical analysis
All statistical analyses were performed using SPSS for Windows version 26. As the data did not follow a normal distribution, the Kruskal–Wallis test, a non-parametric method, was used to assess overall differences among the groups. When the Kruskal–Wallis test indicated significant differences, pairwise comparisons were conducted using the Mann–Whitney U test with Bonferroni correction to adjust for multiple testing.
Categorical variables were summarized as frequencies and percentages, while continuous variables were expressed as median (minimum–maximum). A p-value of < 0.05 was considered statistically significant. Statistically significant pairwise differences identified in post-hoc analyses were indicated in the figures using letter-based annotations (e.g., a, b, c) to denote statistically distinct groupings.