Study design
We conducted a single-center, open-label, opportunistic pharmacokinetic (PK) study in preterm infants (
Study procedures
Infants received ampicillin per standard of care, typically while awaiting results of evaluation for EOS shortly after birth. The decision to start ampicillin was not a part of the study, and no specific dosing was required; however, during the period of study, preterm infants at our center received ampicillin 200 mg/kg/day divided every 8 h, infused over 15 min. There was no minimum or maximum duration of ampicillin administration required by the study.
Whole blood samples (minimum 200 μL) were collected for ampicillin PK from enrolled infants within a preferred sampling window between 6 and 120 h after the final dose of ampicillin. Sample collection occurred at the time of standard of care lab draws and was collected primarily by venipuncture, with capillary heel-stick collection used when venous access was not available. Samples were processed identically regardless of source (see Sample processing section), and the assay used has been validated for the measurement of ampicillin in human plasma from various blood sources. Up to a maximum of 10 PK samples and 2 mL/kg total whole blood sampled per subject. We collected all samples in human EDTA plasma tubes and centrifuged the samples at 3500 × g for 10 min at 4 °C within 1 h of the blood draw. We then transferred the plasma into a second polypropylene tube and immediately froze specimens at −20 °C. Specimens were transferred to a −80 °C freezer typically within 8 h, but at maximum within 24 h.
Dosing information was recorded from the electronic health record (EHR), including documented start times, and PK sample collection times were recorded by study staff. We collected data from the EHR, including demographics (gestational age, sex, race, ethnicity, birthweight, and postnatal age and current weight closest to dosing and PK sample collection times). Race and ethnicity data were collected from EHR and are recognized as social constructs and not expected to be related to ampicillin clearance. For dosing events, PK sample collection times, and other events of interest, the closest demographic values, including weight and height, and laboratory values, including serum creatinine, plasma albumin, total and direct bilirubin, aspartate aminotransferase, and alanine aminotransferase, were recorded. When these values were not recorded at the same time as an event (dosing event, sample collection event, or simulated time), the nearest last observation was carried forward or the next observation carried backward within ±7 days.
Sample processing
Plasma samples were sent to OpAns Laboratory (Durham, NC) for measurement of ampicillin concentrations. The longest time from sample storage at −80 °C to processing was 6 months, which is below the known stability of ampicillin in human EDTA plasma at 216 days at –80 °C. Sample analysis followed methods previously developed and validated by OpAns. [15, 16] Ampicillin was extracted from 10 μL of human plasma by protein precipitation using acetonitrile fortified with [2H5]-ampicillin as an internal standard. Calibration standards and quality control samples were prepared in drug-free K2EDTA human plasma. Samples underwent high-performance liquid chromatography with tandem mass spectrometric detection using a validated assay. The assay method used has a validated range of 0.05 to 50 μg/mL. Samples with concentrations above the quantitation limit (AQL) were evaluated for evidence of detector saturation (chromatographic peak broadening). If absent, AQL concentrations were reported as is. For concentrations below the quantitation limit (BQL), if the chromatographic peak response could be characterized (i.e., extrapolated peaks had reasonable signal-to-noise ratios), the extrapolated values were reported. Quality controls were nominal concentrations of ampicillin in human plasma at 0.06, 4.0, and 40 μg/mL, and intra-assay accuracy ranged from 99.2-111.2%. Accuracy greater than 100% signifies that the assay overestimated the amount of ampicillin in the sample relative to its nominal concentration; the range reported is within the FDA ± 15% deviation from nominal values, so it was considered acceptable.
Concentration-time plots, comparison of observed and model-predicted values, and Monte Carlo simulations
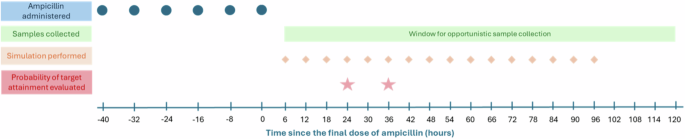
The observed ampicillin concentrations were plotted versus time since ampicillin discontinuation, or the time that the final dose of ampicillin was administered. Using the published ampicillin population PK model by Tremoulet et al., we conducted simulations with our prospectively-collected individual infant characteristics to predict antibiotic exposures after the final dose of ampicillin using NONMEM 7.3 (Icon, Dublin, Ireland). [13] The Tremoulet model was developed from a study of 73 infants (n = 142 ampicillin concentrations), including 21 infants born ≤34 weeks GA and ≤7 days postnatal age. [13] The model was a one-compartment model with linear elimination, and described that ampicillin clearance varies with weight, postmenstrual age, and serum creatinine. Here, we performed 500 simulations per infant to generate concentration-time profiles, inputting infant characteristics and the dosing events. Concentrations were predicted every 6 h starting from 6 h after the final dose of ampicillin (time 0) through 96 h after the final dose of ampicillin. Predicted concentrations were also simulated for each infant at the specific times that PK samples were collected. A timeline of ampicillin administration, PK sample collection, and PK simulations for a typical participant is shown in Fig. 1. The observed sample concentration was compared to the predicted concentrations (n = 500 simulations for each observed concentration) at that time point. With each simulation, NONMEM produces two predicted concentrations: the population predicted concentration, or PRED, based on fixed effects and the mean population parameter estimates, and the individual predicted concentration, or IPRED, predicted incorporating both fixed and random (individual-patient) specific random effects. The model-predicted concentrations were compared to the observed ampicillin concentrations by plotting population-predicted concentrations (PRED) and median individual predicted concentrations (IPRED) versus observed concentrations for within-range samples. For model comparison, AQL and BQL values were excluded to be consistent with the methods followed in developing the Tremoulet model. Notably, these individual predicted values were derived by sampling from the random effects distributions and incorporate unexplained interindividual variability, rather than model fitting of the newly collected observed data.
Example timing of ampicillin administration, opportunistic sample collection window, and timing of pharmacokinetic simulations performed for a typical enrolled infant. Post-discontinuation antibiotic exposure times are measured relative to the time of the final ampicillin dose. For each infant, in addition to the simulations performed every 6 h starting from the final dose of ampicillin through 96 h from the final dose, simulations were also performed to predict ampicillin concentrations at the time of any PK sample collection. The probability of target attainment analysis was conducted using the simulated values at 24 and 36 h after the final dose of ampicillin.
Probability of target attainment analysis
The probability of target attainment (PTA) was the proportion of simulations (n = 27 infants, 500 simulations for each time point) where the individual predicted concentration (IPRED) was greater than various MIC targets of interest: 0.25, 1, 4, and 8 μg/mL. The PTA was calculated at 24 and 36 h after the final dose of ampicillin (see Fig. 1 for timeline). This analysis was conducted for the entire cohort as well as by GA subgroup:
For time-dependent antibiotics such as beta-lactams, the fraction of time spent above MIC (fT > MIC) is the pharmacodynamic endpoint that best relates exposures to efficacy, [17] with optimal bacterial killing occurring when concentrations are maintained above MIC for 40–50% of the dosing interval. We examined four MIC targets in the range of 0.25–8 μg/mL, which represent MIC susceptibility breakpoints for organisms commonly observed in the NICU. While lower MIC targets such as 0.25 and 1 μg/mL provide coverage for Group B Streptococcus and Listeria monocytogenes, respectively, higher MICs (4 and 8 μg/mL) are relevant for organisms such as Enterococcus faecalis and E. coli.
Duration of post-discontinuation antibiotic exposures
To calculate the PDAE, or the number of hours following the final dose of ampicillin that the ampicillin concentration remained above a given MIC target, [14, 18] we used the formula \(C={C_{0}}* {e}^{-{kt}}\), where C was the MIC target of interest, C0 was the minimum concentration at steady state (Cmin,ss) value simulated by NONMEM at the time of the final ampicillin dose, k was the elimination constant, derived from the simulations, and t was the PDAE time, in hours, at which the MIC target concentration was reached. We summarized PDAE in median and 95% confidence intervals (CI, calculated as the 2.5 and 97.5 percentile values for PDAE). We repeated this analysis for the various MIC targets and for each GA subgroup.
Statistical analysis
Descriptive statistics were performed for demographic data and to summarize PDAE durations. Discrete-variable summaries included counts and proportions. For continuous variables, descriptive statistics included the number of observations, mean, median, and percentiles (2.5, 25, 75, 97.5). R Studio version 2022.07.1 was used for statistical analysis and graphical visualization of pharmacokinetic data.