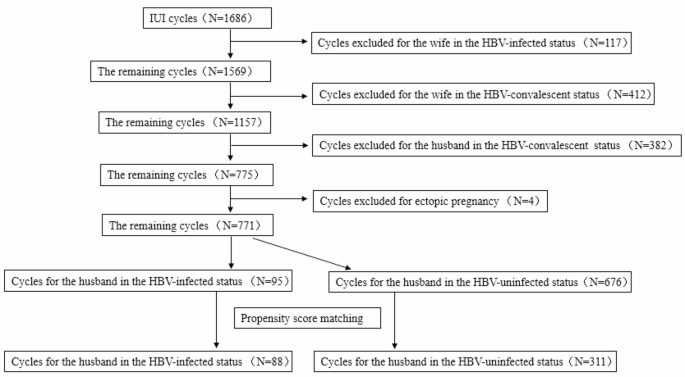
Patient enrollment
A total of 1,686 IUI cycles were performed at the Assisted Reproductive Center of Zhongshan City People’s Hospital from January 2015 to September 2023. Several cycles were excluded from analysis for the following reasons: 117 cycles due to the female partner’s HBV-infected status, 412 cycles due to the female partner’s HBV-convalescent status, 382 cycles due to the male partner’s HBV-convalescent status, and 4 cycles due to ectopic pregnancy. After applying these exclusion criteria, 771 cycles remained eligible for analysis. Within this refined cohort, the study group (HBV-infective group) comprised 95 cycles in which the male partner was HBV-infected and the female partner was HBV-uninfected. The control group (HBV-uninfective group) consisted of 676 cycles in which both partners were HBV-uninfected, as depicted in Fig. 1.
Characteristics of the two groups of couples before and after matching
Before the matching process, there were no significant differences between the two groups in several key characteristics, including D-dimer levels, thyroid-stimulating hormone levels, number of IUI treatments, stimulation protocols, body mass index, basal FSH (bFSH), basal LH (bLH), bFSH/bLH ratio, endometrial thickness on the day of IUI, number of follicles ≥ 14 mm on IUI day, luteal phase support, presence of polycystic ovary syndrome, endometriosis, male infertility factor, sperm concentration, percentage of progressive motile sperm, and total count of progressive motile sperm after semen processing (P > 0.05). However, significant differences were observed in female age, male age, duration of infertility, and type of infertility between the two groups (P
To control for these significant differences and important IUI treatment factors, the two groups were matched for female age, duration of infertility, and type of infertility. After matching, the study group (HBV-infective group) included 88 cycles and the control group (HBV-uninfective group) included 311 cycles, as illustrated in Fig. 1. Following matching, a statistically significant difference was observed in male age between the two groups (P P > 0.05), as shown in Table 1.
Comparison of pregnancy outcomes of IUI between the two groups before and after matching
Before the matching process, there were no significant differences between the two groups in terms of hCG positivity rate, clinical pregnancy rate, early pregnancy loss rate, preterm birth rate, low birth weight rate, mean birth weight, or gender ratio (P > 0.05). These results indicate that the initial outcomes following IUI treatments were comparable, regardless of the male partner’s HBV infection status.
After matching based on relevant characteristics, hCG positivity rate, clinical pregnancy rate, preterm birth rate, low birth weight rate, mean birth weight, and gender ratio continued to show no significant differences between the groups (P > 0.05). However, a significant difference was observed in early pregnancy loss rate, which was notably higher in the HBV-infective group compared to the HBV-uninfective group (43.75% vs. 14.00%, P 2.
GEE analysis of pregnancy outcome of IUI
GEE was used for data analysis before and after propensity score matching. After adjusting for potential confounding factors, including female age, male age, type of infertility, bFSH/bLH ratio, endometrial thickness on IUI day, number of follicles ≥ 14 mm on IUI day, luteal phase support, and total progressive motile sperm count post-semen processing, the male partner’s HBV infection status was not significantly associated with hCG positivity or clinical pregnancy in IUI treatment (P > 0.05), both before and after matching. However, after adjusting for female age, male age, type of infertility, bFSH/bLH ratio, and luteal phase support, male HBV-infection status was significantly associated with an increased risk of early pregnancy loss in IUI treatment both before and after matching (P P 3.
Discussion
China is recognized as a high-prevalence region for HBV, with approximately 5–7% of the population testing positive for HBsAg4. Studies conducted across multiple reproductive centers in China have revealed that the prevalence of HBsAg among male patients ranges from 7.34% to 10%5,6. At our center, the prevalence rate of HBsAg in male patients undergoing IUI treatment is 11.3%, which is marginally higher than that in the general population.
HBV is known not only for its potential to cause liver damage but also for its ability to affect male genitourinary tract tissues, thereby reducing male fertility. Moreover, studies have demonstrated that HBV can be detected in the testes, sperm, and semen7,8, and it may even be transmitted vertically via sperm9. Given these potential effects on male fertility, many infected individuals seek ART to achieve conception. Considerable research has examined how male HBV infection influences the outcomes of ART, such as in vitro fertilization (IVF) and intracytoplasmic sperm injection (ICSI)10,11,12,13. In most of these studies, patients were divided into two groups based on HBsAg status (positive vs. negative). The results showed that HBV infection is associated with a lower fertilization rate in IVF cycles, although the overall pregnancy rate appears unaffected10,11. In ICSI cycles, however, HBV infection has been linked to reduced normal fertilization rates, lower high-quality embryo formation rates, decreased implantation rates, and lower clinical pregnancy rates13. Despite these findings for IVF and ICSI, there is limited research on the relationship between male HBV infection and the pregnancy outcomes of IUI treatments. Consequently, it remains unclear whether the effects observed in IVF and ICSI also apply to IUI.
The grouping criteria in this study differed from those of previous research by focusing specifically on patients’ HBV infection status, categorizing them as either HBV-infected or HBV-uninfected, while intentionally excluding patients in the HBV-convalescent stage. This decision was based on several considerations: First, HBV can damage hepatic function and impair the oxidative breakdown of estrogen, leading to elevated estrogen levels and reduced androgen levels in the bloodstream14,15. Such a hormonal imbalance may result in testicular atrophy and sexual dysfunction in males16,17. In the context of IUI treatment, the period of HBV convalescence and liver function recovery can influence the levels of these hormones. This is particularly relevant for patients who achieve HBsAg-negative status through anti-HBV therapy, as their hormonal balance during the recovery phase may still affect the success of fertility treatments. Second, HBV infection increases oxidative stress18, including in semen19, while reducing antioxidant levels in the body. Administering appropriate antioxidants can support recovery. However, it is important to carefully assess the potential effects of different dosages and treatment durations on oxidative stress, especially during the HBV-convalescent stage. Elevated oxidative stress may negatively affect sperm motility19. Third, interferon and lamivudine, commonly used in anti-HBV treatments, have been associated with reproductive toxicity, including reduced sperm motility and structural damage to testicular tissue20,21,22. In the HBV-convalescent stage, the duration of drug administration and the time since discontinuation can vary widely between patients, potentially affecting reproductive health. Given these considerations, along with the fact that patients with serum HBsAg-negative status may include those in the HBV-convalescent phase, in whom the effects of HBV on physical function may not be entirely absent, it was crucial to select subjects distinctly categorized as HBV-infected (fully affected by HBV) and HBV-uninfected (completely unaffected by HBV). This approach ensured a more reliable assessment of the impact of HBV infection on fertility treatments.
This study demonstrated that male HBV infection does not affect semen parameters, hCG positive rate, clinical pregnancy rate, preterm birth rate, low birth weight rate, mean birth weight, or infant gender ratio in IUI treatment. However, it significantly increased the risk of early pregnancy loss, with HBV-infected males being 7.02 times more likely to experience early pregnancy loss than HBV-uninfected males. These findings offer important insights for male patients with HBV infection considering IUI treatment, highlighting the need for careful consideration and management of HBV infection in the context of reproductive health.
The impact of HBV on male reproductive health has garnered significant attention in recent research. Evidence shows that HBV can be detected at various stages of spermatogenesis, as well as in the sperm and semen of carriers, in both free HBV-DNA and integrated forms7,8. Specifically, free HBV-DNA has been found in the seminal plasma and the cytoplasm of the sperm head, but not in the acrosome1,7. In contrast, integrated HBV-DNA has been detected within the chromosomes of sperm cells, indicating a deeper level of interaction between the virus and male gametes23,24. Moretti et al. reported that HBV significantly increases the proportion of apoptosis and necrosis in sperm18. Similarly, Huang’s study revealed that HBV-DNA integrates into sperm chromosomes in a non-specific manner through multilocus recombination, resulting in elevated sperm chromosomal aberrations and DNA damage, potentially compromising the fertilization capability of the sperm and the health of the offspring24. Other studies have demonstrated that merely incubating sperm with HBV S protein can trigger apoptosis and DNA damage, with the severity of effects depending on exposure dose and duration25,26. Damage to sperm DNA can negatively affect the development of embryos. In addition, as previously mentioned, male patients with HBV infection may experience reproductive impairment from abnormal sex hormone levels, oxidative stress, and adverse effects of anti-HBV medications, all of which disrupt sperm production and development. In the context of IUI treatment, it is essential to use density gradient centrifugation to remove apoptotic or necrotic sperm while retaining healthy ones. However, research has found that the DNA fragmentation index (DFI) in motile sperm does not significantly decrease after this procedure, and in approximately 60% of patients, it may even increase27,28. Based on these evidence, an elevated DFI in motile sperm could potentially contribute to early pregnancy loss. Nevertheless, such sperm DNA damage does not affect the hCG positive rate, clinical pregnancy rate, preterm birth rate, and low birth weight rate.
Furthermore, it is important to consider two key issues: whether processed sperm retains HBV and whether this could result in vertical transmission of HBV from father to offspring. Semen processing via density gradient centrifugation and washing can effectively remove HBV present on the sperm surface and in the seminal plasma29,30. However, HBV may persist within the plasma of the sperm head and in sperm chromosomes 31,32. Although complete elimination of HBV from the semen of every patient is unlikely, processing markedly reduces the viral load31. After processing, only 0.3%–1.1% of sperm are found to carry HBV32, greatly reducing the likelihood of vertical transmission to the embryo. Despite this low probability, transmission remains possible. HBV can be transferred into the oocyte via sperm, leading to embryonic infection. Research has shown that mRNA transcription of HBV genes occurs in discarded embryos from IVF cycles involving HBsAg-positive male partners and HBsAg-negative female partners33,34, supporting the evidence that HBV can be vertically transmitted from a father to their offspring. The transcription of HBV genes in embryos is regulated by DNA methylation35, microRNA36, and host genes37. Experimental evidence shows that HBV S protein reduces the yield of two-cell embryos from human–hamster fertilization. In contrast, HBV X protein decreases mitochondrial membrane potential in human embryonic stem cells and increases DNA damage34. These effects may hinder embryonic development. Epidemiological surveys of fetal HBV serology showed that the transmission of HBV from the sperm of a carrier father to the fetus is rare38. Furthermore, although high-throughput sequencing of peripheral blood cells is now common in clinical diagnostics, no report has documented integration of HBV-DNA into human chromosomes. This absence of evidence implies that embryos infected via paternal transmission may fail to develop to term, being lost through implantation failure, biochemical pregnancy, or miscarriage. The results of this study support this view, suggesting that early pregnancy loss may serve as a natural mechanism for eliminating embryos infected through paternal vertical transmission.
Another factor that may have influenced this study’s outcome is the effect of male HBV infection on the reproductive system of healthy female partners. HBV is present in both semen and sperm, and its protein antigens can be expressed within sperm35,39. Consequently, when semen or sperm carrying HBV is introduced into the female reproductive tract during sexual intercourse, it may trigger an immune response. Since the 1980 s, China has provided free HBV vaccinations for children40, which has resulted in the majority of individuals of reproductive age possessing an immune defense against HBV. Repeated exposure to HBV antigens through sexual intercourse could therefore provoke a sustained anti-HBV immune reaction. The immune environment of the uterine cavity is thought to lean towards a Th1 state41. We speculate that this specific immune profile may not interfere with fertilization or implantation but could hinder the maintenance of pregnancy. As a result, this study found that while the hCG positive rate and clinical pregnancy rate remained unchanged, there was an increase in the rate of early pregnancy loss. Sperm also engage in complex interactions with the female reproductive tract, influencing early embryo development42. Notably, Lao et al. reported that male HBV infection may increase the risk of fallopian tube damage and infertility in female partners43. Similarly, Zandieh et al. demonstrated that sperm DNA damage can activate the toll-like receptor signaling pathway in human fallopian tubes, thereby resulting in the upregulation of inflammatory cytokines and chemokines44. Based on this evidence, we hypothesize that inflammation in the fallopian tubes and uterine cavity induced by HBV-related sperm DNA damage may be another mechanism that negatively affects pregnancy. Furthermore, sexual intercourse might cause inflammation in the female reproductive tract and alter its microbiota45,46, which plays a critical role in reproductive health and infertility treatment outcomes47,48. Disruption of this microbiota may therefore contribute to the higher rate of early pregnancy loss observed in this study among female partners of HBV-infected males.
Limitations
This study had certain limitations. First, the sample size was modest, which may lead to biased parameter estimates, overfitting, and potential model convergence issues. Second, the study was a retrospective single-center analysis, which may have introduced selection bias and influenced patient characteristics. The retrospective design also posed challenges in sample accessibility and data supplementation, resulting in the absence of certain clinical data, such as sperm DFI, HBV viral load, and specific antiviral treatments. These gaps may have introduced unmeasured confounding variables. Third, some proposed mechanisms underlying early pregnancy loss associated with male HBV infection were speculative, based on literature, rather than directly supported by the study’s data. Therefore, the results should be interpreted with caution.
To obtain more comprehensive insights and confirm the validity of these findings, future research should adopt prospective, multicenter designs with larger sample sizes. Despite these limitations, the results of this study provide important insights and serve as a valuable reference for male patients with HBV infection considering IUI treatment.