Ethics approval and consent to participate
Details of a patient’s course of hospitalization at a study site were collected after obtaining written informed consent from the patient or from the patient’s legal guardian, where applicable.
The study followed the applicable local and national guidelines and regulations for research involving human participants. It was approved by the Indian Government’s Health Ministry Screening Committee (TDR/692/2017-ECD-II, dated 7th July 2017) and institutional review boards of participating sites:
-
Chandigarh—Postgraduate Institute of Medical Education and Research, Chandigarh, Institutional Ethics Committee (reference number PGI/IEC/2021/000612 dated May 3, 2021); Government Medical College and Hospital, Sector 32, Chandigarh Institutional Ethics Committee (reference number GMCH/IEC/2021/633R/368 dated September 24, 2021).
-
Vellore—Christian Medical College, Vellore, Institutional Review Board (minute number 13489 (OBSERVE) dated October 28, 2020).
-
Ludhiana—Christian Medical College and Hospital, Ludhiana, Institutional Ethics Committee (reference number IECCMCL/BMHR-06-362-21/Ren-Apprvl/Microbiology, dated June 24, 2021).
-
Delhi—All India Institute of Medical Sciences, New Delhi, Institute Ethics Committee (reference number IEC-562/03.11.2017, RP-13/2017, dated November 6, 2017, extension on April 5, 2021); Chacha Nehru Bal Chikitsalaya, Delhi (reference number F.1/IEC/CNBC/17/04/2021/Protocol no. 99/13159 dated November 2, 2021).
-
Ahmedabad—Indian Institute of Public Health, Gandhinagar, Institutional Ethics Committee (reference number TRC/2020-21/18, dated November 15, 2021).
-
Kolkata—The Calcutta Medical Research Institute, Kolkata, Institutional Ethics Committee (reference number IEC/02/2022/APRV/16 dated October 27, 2022); Fortis Hospital, Kolkata, Ethics Committee (IEC protocol submission number IEC/2022/OAS/02 dated June 15, 2022); Ruby General Hospital Ltd., Kolkata, Institutional Ethics Committee (format number IEC-RGH/KOL/2021/002/CS dated March 28, 2022).
-
Bengaluru—St. Johns Medical College & Hospital, Bengaluru, Institutional Ethics Committee (reference number IEC/1/1272/2021, dated November 18, 2021)
Study setting
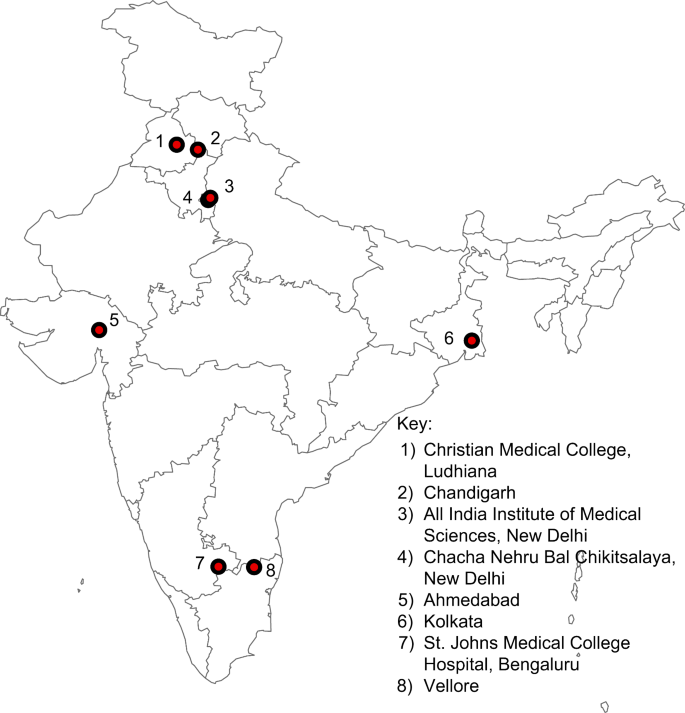
Data were collected from eight sites representing the Northern, Southern, Eastern, and Western regions of India. Of these, the Chandigarh and Vellore sites collected and reported S. Typhi and S. Paratyphi isolates as part of a hybrid surveillance setup (Tier 2) as previously described25. The remaining six were lab-based surveillance setups that involved tertiary care hospitals and regional reference laboratories, similar to the Tier 3 surveillance in SEFI’s first phase26. Study hospitals were tertiary care centers unless otherwise specified in the subsequent section. These centers primarily serve their local populations and offer some referral services to patients from nearby towns and villages. Additionally, the Christian Medical College, Vellore, and the All India Institute of Medical Sciences, New Delhi, have broader areas from which they attract referrals. Patients at the study hospitals are from a variety of age-groups and socio-economic strata.
Northern sites:
-
Chandigarh (CHD): Civil Hospital Sector-45 (secondary care), Government Medical College and Hospital Sector 32 (tertiary care), and Government Multispecialty Hospital Sector 16 (tertiary care) were part of the surveillance network. It should be noted that Civil Hospital Sector-45 was a secondary care center and would primarily serve its nearby wards. The Post-Graduate Institute of Medical Education and Research (PGIMER) Chandigarh was the coordinating center for the site and processed all blood samples collected.
-
All India Institute of Medical Sciences (AIIMS), New Delhi.
-
Chacha Nehru Bal Chikitsalaya (CNBC), New Delhi; this is a pediatric specialty hospital that does not treat those who are 15 years of age or older.
-
Christian Medical College, Ludhiana (CMCL).
Western site:
-
Ahmedabad (AMD): CIMS Hospital, SAL Hospital and Medical Institute, Apollo Hospital International Ltd. Gandhinagar and Neuberg Supratech Reference Laboratories (NSRL) provided laboratory-based surveillance of the site. The three hospitals provided up-to-tertiary care; NSRL acted as a reference laboratory for healthcare facilities in the region and, in collaboration with the Indian Institute of Public Health, Gandhinagar, was the coordinating center for this site.
Eastern site:
-
Kolkata (KLK): The Calcutta Medical Research Institute, Chittaranjan National Cancer Institute, Fortis Hospital, Anandpur, and Ruby General Hospital provided laboratory-based surveillance of the site. All four hospitals provided up-to-tertiary care. The surveillance was coordinated by the ICMR- National Institute for Research in Bacterial Infections (NIRBI, previously the National Institute of Cholera and Enteric Diseases), Kolkata.
Southern sites:
-
Vellore: The Christian Medical College (CMC), Community Health and Development Unit (CHAD), and Low-Cost Effective Care Unit (LCECU) were part of the surveillance network. It should be noted that CHAD and LCECU provide primary and secondary care to their local populations. CMC Vellore processed all the blood samples collected.
-
St. John’s Medical College Hospital (STJ), Bengaluru.
Figure 2 shows the geographical locations of the sites.
Geographical locations of sites participating in the second phase of the Surveillance for Enteric Fever in India study. The map was created using ArcGIS ver. 10.8 (https://www.arcgis.com/index.html).
Inclusion and exclusion criteria
At Tier 2 sites, consenting patients who were ≥ 6 months of age, from the site’s catchment areas, and admitted with a febrile illness at a study hospital were recruited and had blood samples collected, which were subsequently cultured. Patients with known diagnoses of malignancy or those who were not Indian citizens were excluded. At Tier 3 sites, cultures of blood or bone marrow samples collected from patients were monitored via routinely maintained laboratory registers at each participating center. The decision to conduct either a blood or bone marrow culture was taken by a patient’s treating physician. There were no exclusion criteria for Tier 3. The surveillance periods for each site are listed in Table 6.
Lab processes
Ahmedabad, Chandigarh, and Vellore used automated BACTEC systems for blood and bone marrow cultures, whereas St. Johns, Bengaluru, and Kolkata used automated BacT/Alert systems. CNBC Delhi and CMC Ludhiana had automated BACTEC systems, but conducted either automated or conventional blood cultures. AIIMS Delhi primarily conducts conventional cultures, with approximately 5% conducted using an automated BACTEC machine. Each site stored S. Typhi and S. Paratyphi isolates at − 20 °C or less. Of the isolates collected, 94.1% (n = 1415) were transferred to the CMC Vellore Microbiology Laboratory for re-identification through standard biochemical assays followed by serotyping using commercial antisera (BD Difco, USA) in accordance with the Kauffmann-White classification system, as described in the Manual of Clinical Microbiology27.
Kirby-Bauer disc diffusion (DD) assays were performed for ciprofloxacin (5 µg), pefloxacin (5 µg), ampicillin (10 µg), ceftriaxone (30 µg), co-trimoxazole (1.25/23.75 µg), azithromycin (15 µg) and chloramphenicol (30 µg). MICs were determined for azithromycin (0.12–128 µg/mL) and ceftriaxone (0.008–16 µg/mL) via broth microdilution which were utilized to reconfirm any resistance found via DD tests. Antimicrobial susceptibility test results were interpreted as per CLSI guidelines10. The 50th percentile (MIC 50) and 90th percentile (MIC 90) MIC values were also calculated for ceftriaxone and azithromycin. The reference isolate for DD tests involving ampicillin (10 µg; 15–22 mm), co-trimoxazole (1.25/23.75 µg; 23–29 mm), chloramphenicol (30 µg; 21–27 mm), ceftriaxone (30 µg; 29–35 mm), ciprofloxacin (5 µg; 29–37 mm), and pefloxacin (5 µg; 25–33 mm), was American Type Culture Collection (ATCC) E. coli 25922, while the reference for azithromycin (15 µg; 21–26 mm) DD tests was S. aureus 25923. Broth microdilution testing for azithromycin (0.50–2 µg/mL) used ATCC S. aureus 29213 as the reference, and ATCC E. coli 25922 for ceftriaxone (0.03–0.12 µg/mL).
The S. Paratyphi B isolates, and ceftriaxone resistant S. Typhi isolates were selected for whole-genome sequencing. Genomic DNA was extracted using the QIAamp® Mini Kit (QIAGEN, Hilden, Germany) following the manufacturer’s protocol. DNA purity was evaluated using a Nanodrop One spectrophotometer (Thermo Fisher, Waltham, USA), while precise quantification was performed using a Qubit Fluorometer with the dsDNA HS Assay Kit (Life Technologies, Carlsbad, USA). For short-read sequencing, genomic DNA was fragmented, and paired-end libraries were constructed using the Illumina Nextera DNA Flex Library Kit and Nextera DNA CD Indexes (Illumina, Massachusetts, USA). Equimolar pooled libraries were sequenced on the Illumina NovaSeq 6000 platform (Illumina, San Diego, USA), generating 2× 150 bp paired-end reads.
Short reads generated from the Illumina platform were assessed for quality using FastQC v0.12.1, ensuring that only high-quality reads (Phred score > 30) were retained for downstream analysis. De novo genome assembly was performed using SPAdes28. Assembled genomes were uploaded to the Pathogenwatch platform v23.0.229 for comprehensive genotypic analysis. This included Salmonella 7-gene multilocus sequence typing30 and genotype identification using Genotyphi31. Additionally, antimicrobial resistance (AMR) genes, point mutations, plasmid replicon types, and single nucleotide polymorphisms (SNPs) were detected within the Pathogenwatch framework.