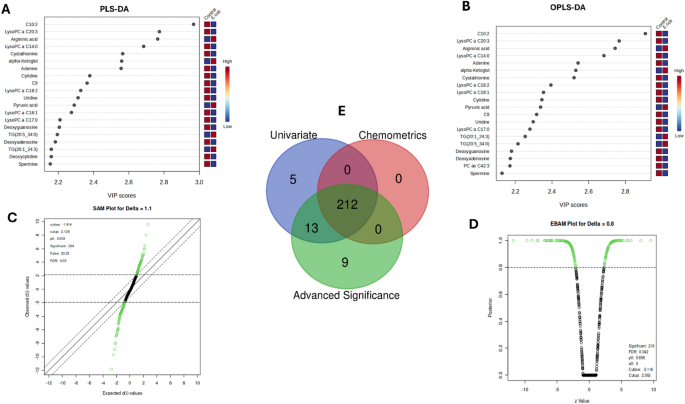
By SAM analysis, 234/598 (39.13%) metabolites were determined to be statistically significant (p = < 0.05) (Table S2E). Of which, 96 (41.02%) metabolites were upregulated and 138 (58.97%) were downregulated according to the d-value scores with the range of 9.5 to 2.12 and − 11.80 to −1.9 respectively in 598 queried metabolites [false discovery rate (FDR) < 0.05] (Fig. 5c). Among these upregulated metabolites, argininic acid displayed the highest increase (d.value = 9.57, p = < 0.05), indicating major alterations in arginine metabolism that are probably related to the physiological stress response due to E. coli infection. Furthermore, alpha-ketoglutaric acid showed a very significant increase with a d.value of 8.0145 and a p = < 0.05, indicating an increased production of energy through the tricarboxylic acid (TCA) cycle. Pyruvic acid also exhibited significantly enhanced glycolytic activity, likely reflecting its role in rapid energy provision, with a d.value of 6.48 and a p = < 0.05. TG (20:5_34:0) showed increased TG mobilization so as to meet the increased energy requirement associated with infection (d.value = 6.04 p = < 0.05).
On the contrary, out of the top three most downregulated metabolites, C10:2 had a z-value of −11.807 with a p = < 0.05, hence, highly diminished fatty acid metabolism could implicate disturbed lipid mobilization due to E. coli infection. Among these, the most significantly downregulated metabolites included LysoPC a C20:3 (z.value = −9.66, p = < 0.05) and LysoPC a C14:0 (z.value = −8.91, p = < 0.05), demonstrating disturbances in lysophosphatidylcholine metabolism and thus may indicate defective membrane remodeling and decreased phospholipid availability under the infection.
Subsequent to the SAM analysis, EBAM also provided strong evidence for the alteration in the levels of metabolites in the E. coli infected group (Fig. 5d) (Table S2F). A total of 210 (35.11%) out 598 metabolites were determined to be statistically significant. These results indicated the overall increased energy metabolism and stress response in the infected birds as evidenced by the up regulation of argininic acid (z-value = 9.576, p = < 0.05), alpha-ketoglutaric acid (z-value = 8.01). In contrast, metabolites such as LysoPC a C20:3 (z-value = −9.66, p = < 0.05) and LysoPC a C14:0 (z-value = −8.91, p = < 0.05) were downregulated implying that the membrane lipid metabolic process and phospholipid synthesis ability are inhibited during infection stress. These changes demonstrate the metabolic shift that occurs during infection.
The Venn diagram shows 212 metabolites that were commonly identified by univariate analyses, such as t-test and chemometric analyses such as PLS-DA and OPLS-DA, and advanced significances using SAM and EBAM, thereby giving strength to these results (Fig. 5e) (Table S2G). This overlap indicates a high degree of similarities in metabolite analysis via different methods. This further consolidates the reliability of those key metabolic alterations in response to E. coli infection.
Comprehensive chemometric and statistical profiling of metabolomic alterations post- E. coli challenge (24 h) in broiler chickens. (a) PLS-DA VIP scores for key discriminant metabolites in distinguishing between the E. coli-infected vs. control groups. (b) The OPLS-DA VIP scores further confirmed the findings of the PLS-DA, with adequate class discrimination. (c) SAM plot showing metabolites rated as significant by their differential abundance. (d) EBAM plot showing the significant metabolites and their posterior probabilities under E. coli infection. (e) Venn diagram: overlaps from metabolites identified by univariate, chemometric, and advanced significance analysis showing the consistency of findings.
8 h vs. 24 h: metabolite and pathway differences
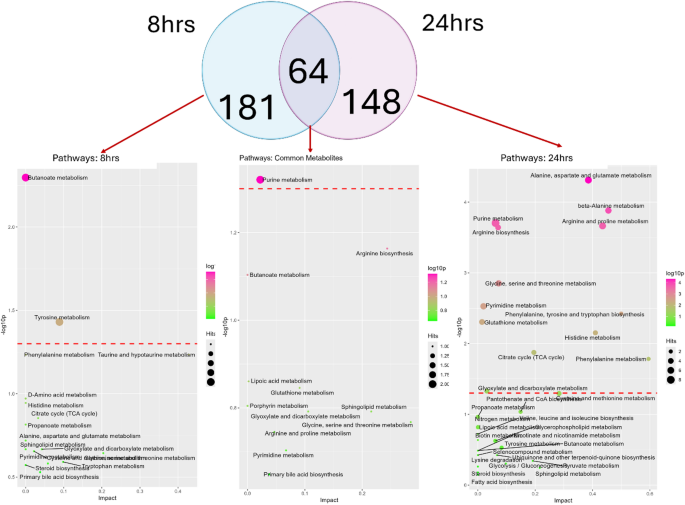
The pathway analysis of 245 metabolites at 8 h post-E.coli infection and 212 metabolites from 24 h post-infection data reveals distinct and overlapping metabolic changes (Fig. 6). In 8 h post-E. coli infection showed that butanoic acid and succinate metabolites were enriched in butanoate metabolism, which is associated with short-chain fatty acid metabolism. Furthermore, tyrosine metabolism was also enriched, involving L-noradrenaline and 4-hydroxyphenylacetate metabolites (p = < 0.03) (Table S3A). The list of unique metabolites specific to 24 h infection data consisted of 148 metabolites and showed several significantly enriched pathways. Alanine, aspartate, and glutamate metabolism had the highest significance: p = < 4.89E-05, impact = 0.38; metabolites include L-aspartate, L-alanine, L-glutamine, citrate, pyruvate, and 2-oxoglutarate. The following was beta-alanine metabolism: p = < 0.0001; key metabolites take part in beta-alanine, L-aspartate, L-histidine, spermidine, and spermine. Other significantly enriched pathways were those of purine metabolism, with p = 0.0001, involving metabolites like L-glutamine, adenosine, deoxyadenosine, deoxyinosine, hypoxanthine, guanine, urate, and guanosine; this represents large changes in nucleotide metabolism. Arginine biosynthesis (p = < 0.0002) was enriched with metabolites such as L-aspartate, L-ornithine, ammonia, L-glutamine, and 2-oxoglutarate, suggesting involvement in amino acid synthesis. Additional pathways, such as glutathione metabolism (p = < 0.004) with L-ornithine, putrescine, spermidine, and spermine, and pyrimidine metabolism (p = < 0.002) featuring L-glutamine, cytidine, deoxycytidine, thymidine, and beta-alanine, were also enriched, pointing toward changes in antioxidant defense and nucleotide metabolism. Histidine, phenylalanine, TCA cycle, glyoxylate and dicarboxylate metabolisms were also detected, highlighting shifts in energy metabolism and amino acid biosynthesis specific to batch 2 (p = < 0.05) (Table S3B). The enrichment analysis of the 64 common metabolites between 8 h and 24 h, identified purine metabolism as a significantly enriched pathway (p = 0.04), involving metabolites like deoxyguanosine and adenine (Table S3C).
Identification of common and unique metabolites and enriched pathways across 8 h and 24 h post-E. coli challenge: The Venn diagram shows 181 unique metabolites at 8 h, 148 at 24 h, and their overlap (64 common metabolites). Pathway enrichment analysis is shown for the unique as well as common metabolites of 8 and 24 h datasets.
Logistic regression (LR) highlights early biomarkers of E. coli challenge
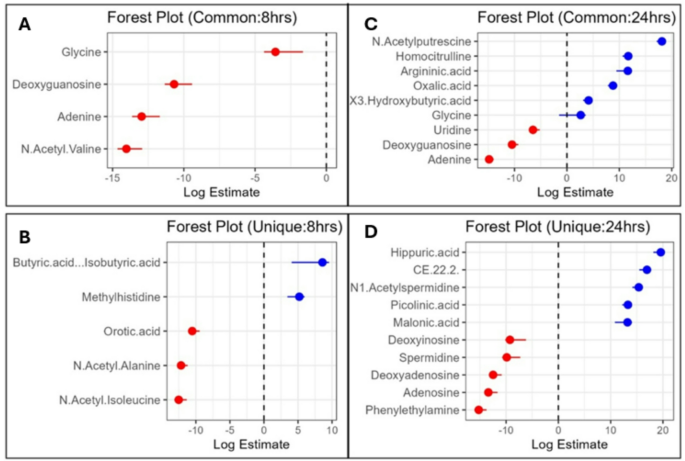
LR analysis can predict the infection status by weighing metabolite effect sizes, where odds ratios > 1 imply increased disease risk and < 1 indicate the protective effects, thereby facilitating in disease biomarker identification. The filtering of 393 metabolites between the two batches (8 h and 24 h post-E. coli infection) based on the pathway enrichment status identified a total of 91 metabolites, including 24 metabolites from 8 h, 51 metabolites from 24 h to 16 common metabolites (both batches). The LR analysis of these 91 metabolites identified 48 significantly enriched metabolites (p = < 0.05). Of these, batch 1 (8 h) had 9 metabolites (5 unique, 4 common), and batch 2 (24 h) had 39 metabolites (30 unique, 9 common) showing the different temporal metabolic signatures of infection progression (Table S3D and S3E) (Fig. 7a-d).
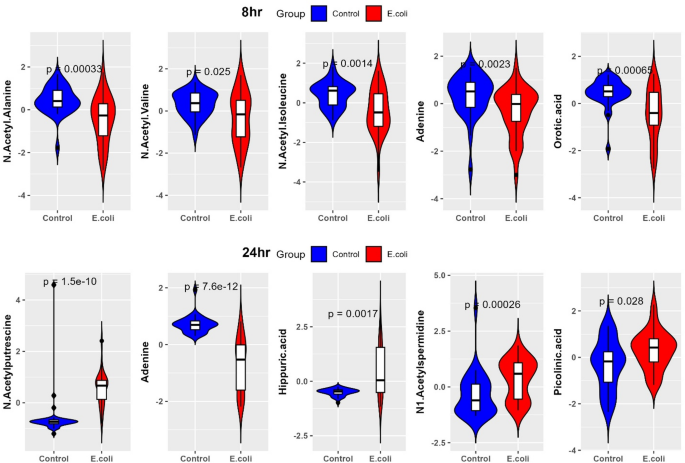
At 8 h post-E. coli challenge, LR results showed significant alterations in metabolites involving amino acid, fatty acid, and nucleotide metabolisms. Butyric acid/isobutyric acid (log estimate: 8.59, p = < 0.04) was significantly upregulated, suggesting early infection stage leads to the changes in short chain fatty acid metabolism and gut microbiome composition. Log estimate means for each 1 µM increase in butyric acid/isobutyric acid, the log odds of infection increase by 8.59 units. Methylhistidine (log estimate: 5.20, p = < 0.005) also showed positive association and may represent muscle protein catabolism as a response to infection. In contrast, N-acetyl-isoleucine (log estimate: −12.52, p = < 0.0004) and N-acetyl-alanine (log estimate: −12.18, p = < 6.65E-05) had strong negative associations indicating early dysregulation of amino acid metabolism (Table S3D). The marked depletion of orotic acid (log estimate: −10.53, p = < 0.0002) implies the altered nucleotide biosynthesis. However, based on the consensus scores (CS) calculated by combining effect size and statistical significance, the top metabolites were N-acetyl-alanine (CS = 50.86), N-acetyl-valine (CS = 50.34), N-acetyl-isoleucine (CS = 41.86), and orotic acid (CS = 38.66). Additionally, we identified the depletion of deoxyguanosine (CS = 33.25) and glycine (CS = 7.51). Their expression values are presented in the form of violin plots in Fig. 8. These findings suggest early dysregulation of nucleotide biosynthesis and amino acid metabolism as part of the host’s strategy to limit bacterial growth and manage oxidative stress during early E. coli infection.
At 24 h post E. coli challenge, the metabolites involving amino acid and energy metabolism were significantly altered. Hippuric acid (p = < 0.001, log estimate: 19.59, CS = 53.78) was the top-ranked metabolite, indicating increased metabolism of aromatic amino acids and potential alterations in gut microbiota. N1-acetylspermidine (p = < 0.0005, log estimate: 15.38, CS = 49.82) and picolinic acid (p = < 0.0002, log estimate: 13.31, CS = 48.38) were also ranked high, reflecting disruptions in polyamine metabolism and nicotinamide pathway activity, respectively. Furthermore, CE (22:2) (p = < 0.002, log estimate: 16.95, CS = 45.42) revealed profound changes in lipid metabolism, attesting to the metabolic adjustment of the host to infection. Additionally, downregulated metabolites like phenylethylamine, adenosine, deoxyadenosine, spermidine, and spermine indicated major changes in nucleotide, amino acid and stress response pathways (p = < 0.05). These findings indicate coordinated metabolic reprogramming during late stages of E. coli infection reflecting the host’s adaptation to counteract bacterial growth and manage inflammation.
A comparison of the common metabolites at 8 and 24 h post-infection revealed sustained dysregulation in nucleotide metabolism, with a significant downregulation of deoxyguanosine (8 h: log estimate: −10.68, p = < 0.0007; 24 h: log estimate: −10.47, p = < 0.0005) and adenine (8 h: log estimate: −12.96, p = < 0.0008; 24 h: log estimate: −14.84, p = < 1.40E-05). Glycine showed a significant shift from a negative association at 8 h (log estimate: −3.57, p = < 0.007) to a positive association at 24 h (2.63, p = < 0.03), suggesting temporal shifts in amino acid metabolism. At 24 h, some common metabolites such as N-acetylputrescine (log estimate: 18.12, p = < 9.49E-05), homocitrulline (log estimate: 11.70, p = < 0.0001), and argininic acid (log estimate: 11.61, p = < 0.01) were altered. Their expression values are presented in the form of violin plots in Fig. 8. These changes indicated progressive changes in the urea cycle and polyamine metabolism with the infection time course. While prominent at 24 h, these metabolites were not significantly altered at 8 h as per LR findings.
Forest plots of serum metabolite changes in broilers post-E. coli infection at 8 and 24 h. Panel A and B shows unique and common metabolites in 8-h data, whereas panels C and D show unique and common metabolites in 24 h data. Red and blue dots are for negative and positive log odds estimates, respectively, with error bars showing confidence intervals.
Violin plots showing expression levels of the metabolic biomarker panel in broiler chickens at 8 and 24 h post-E. coli infection. N-acetyl-alanine, N-acetyl-valine, N-acetyl-isoleucine, adenine, and orotic acid were downregulated at 8-h post-infection. N-acetylputrescine, hippuric acid, N1-acetylspermidine, and picolinic acid were upregulated, while adenine remained downregulated at 24 h post-E. coli infection.