Selection process and characteristics of included studies
Among 12 624 references identified, we assessed 578 studies on full-text to finally include 69 studies (Fig. 1; Table 1), reporting 2 917 IFI in 35 781 episodes. Reasons for exclusion based on full-text assessment are listed in additional file 6. The studies included were mainly single-center (n = 56, 81%) retrospective (n = 57, 90%) cohorts (n = 63, 91%) describing IFI based on 2008 EORTC/MSG criteria (n = 49, 71%) (Table 1). Publications mostly included patients from North America, Asia and Europe (n = 24, 35%, n = 23, 33%, and n = 18, 26%, respectively). The studies concerned mainly patients with allo-HSCT (n = 20, 29%), followed by myeloid malignancies (n = 18, 26%) (Table 1). Three studies with adjusted measure effects were performed on large databases (with more than 3 000 patients included each), but used other definitions than EORTC/MSG and, as a consequence, were not included in the meta-analysis (additional file 7). Eight studies reported results on episodes rather than patients. These episodes were based on chemotherapy courses for seven and on respiratory viral infections for one.
Methodological quality assessment for cohort and case control studies is described in additional files 8 and 9, respectively. Of the 69 included studies, 31 (45%) were considered at low risk of bias, 30 (43%) at unclear risk of bias and 8 (12%) at high risk of bias.
For the meta-analysis on PP-IFI, among 26 studies fulfilling criteria for potential inclusion, 20 were finally included (additional file 7 and Table 1), including 1 014 IFI in 9 270 episodes. For the PPP-IFI meta-analysis, 36 of the 41 considered studies were included, corresponding to 1 348 IFI in 11 247 episodes (Table 1). In additional file 10, we reported methods and variables on which the analyses were adjusted. In the PP-IFI meta-analysis, 16 (80%) studies were considered at low risk of bias, 2 (10%) at intermediate risk of bias and 2 (10%) at high risk of bias. In the PPP-IFI meta-analysis, 26 (72%) studies were considered at low risk of bias, 3 (8%) at unclear risk of bias and 7 (20%) at high risk of bias (additional files 8 and 9).
Qualitative synthesis
Overall, a total of 62 variables were assessed in at least one study. Age, neutropenia, corticosteroids and antifungal prophylaxis were the most frequently studied parameters (adjusted measure effects for 14, 17, 13 and 12 studies, respectively).
Having a pulmonary comorbidity was associated with IFI in the 3 studies assessing this variable in multivariate analysis. Namely, having an underlying pulmonary disease was associated with an increased risk of PP-IFI occurring in the first 40 days after HSCT (HR 7.57 [95%CI 2.05–27.77]) and with IFI regardless of a time period (HR 1.66 [95%CI 1.03–2.67]) and chronic obstructive pulmonary disease (COPD) was associated with PPP-IFI (OR 3.28 [95%CI 1.48–7.28]) (additional file 11). Interestingly, in five studies reporting diabetes (two in univariate analysis and three in multivariate analysis), none showed a statistically significant association with IFI (additional files 11 and 12). Regarding treatment, an intensified GvHD therapy was statistically associated with PP-IFI in multivariate analysis (OR 3.60 [95%CI 1.30–9.90]), both in the late period and very late period after HSCT (41–100 days after HSCT HR 3.45 [95%CI 1.15–10.31] and 101–365 days after HSCT HR 37.93 [95%CI 3.76-379.45], respectively) (additional file 11).
A synthesis of factors associated with IFI assessed in the studies, along with their definitions as reported by the original study authors, are displayed in additional files 11 and 12, based on univariate and multivariate analyses, respectively.
Quantitative synthesis
Patients’ characteristics
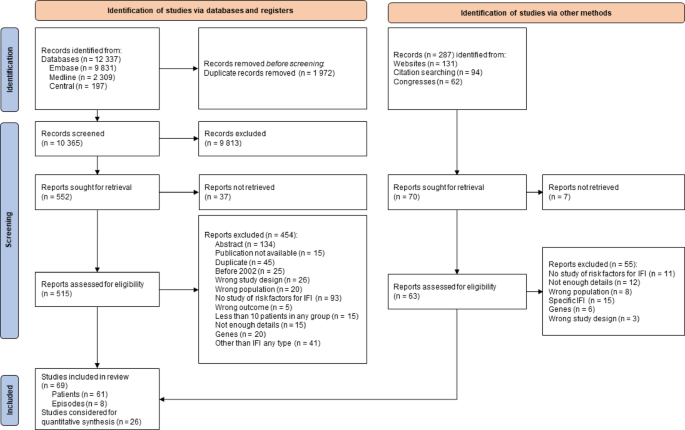
In the meta-analysis including PP-IFI, no patients’ characteristics were significantly associated with IFI (Fig. 2).
When considering PPP-IFI, age more than 60 years old was significantly associated with IFI (pooled OR with 3 studies 3.46 [95% CI 1.48–8.08]) as well as history of IFI (in the allo-HSCT population, pooled HR with 2 studies 5.49 [95% CI 2.52–11.97])21,22 (additional file 13). Nonetheless, with OR as the measure effect, with one study reporting multiple myeloma patients23 and the other reporting allo-HSCT patients24, history of IFI was not significantly associated with IFI (pooled OR with 2 studies 5.39 [95% CI 0.52–55.80]) (additional file 13).
Hematological malignancy or HSCT characteristics
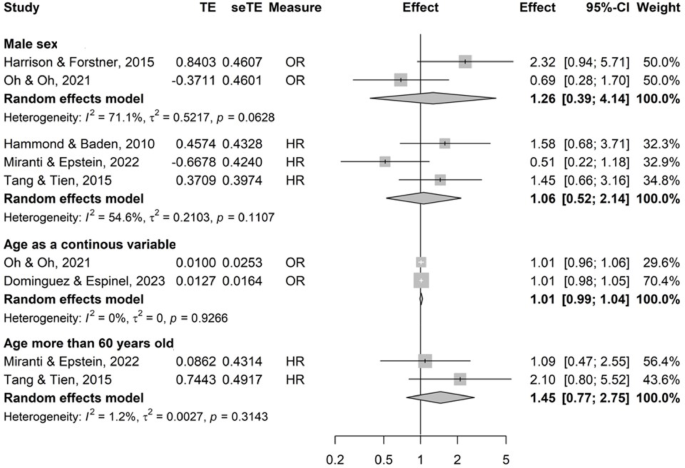
AML was not significantly associated with PP-IFI or PPP-IFI based on 3 and 5 studies, respectively (Fig. 3 and additional file 14) but was compared to other high-risk populations, namely transfusion-dependent MDS or ALL patients25,26. Moreover, this variable displayed moderate heterogeneity (I2 = 60%, phet = 0.08).
Forest plots of hemopathy or hematogenous stem cell transplantation status’ characteristics evaluated as risk factors for proven and/or probable invasive fungal infections. Abbreviations: AML acute myeloid leukemia; CI confidence interval; GvHD graft vs. host disease; HR hazard ratio; HSCT hematogenous stem cell transplant; OR odds-ratio; SE standard error, TE treatment effect. *High risk for mortality was defined in the studies according to the European Group for Blood and Marrow Transplantation or the HSCT-comorbidity index risk scores.
In hypomethylating agents-treated AML patients, the relapsed and/or refractory status of the hemopathy was significantly associated with PP-IFI (pooled OR 6.64 [95% CI 3.72–11.84], based on 2 studies) (Fig. 3).
Previous allo-HSCT was significantly associated with IFI (pooled HR with 2 studies 3.21 [95% CI 1.54–6.71]).
Allo-HSCT at higher risk for mortality according to consensus risk scores27,28 showed discrepant results based on association measure (pooled HR with 2 studies 2.52 [95% CI 1.36–4.67], and pooled OR with 2 studies 0.52 [95% CI 0.19–1.44]). Haploidentical donor was significantly associated with PP-IFI (pooled HR with 2 studies 2.41 [95% CI 1.27–4.57]).
Higher grades of acute GvHD (≥ 2) was significantly associated with PP-IFI whereas severe chronic GvHD was not (pooled HR 2.59 [95% CI 1.36–4.90] and pooled HR 1.49 [95% CI 0.55–4.07], with substantial heterogeneity, I2 = 66%, phet = 0.08, respectively, with 2 studies each, Fig. 3). When including PPP-IFI, GvHD regardless of its acute or chronic status, was significantly associated with IFI (pooled OR with 3 studies 4.84 [95% CI 2.34–10.05]) (additional file 14).
Biological parameters
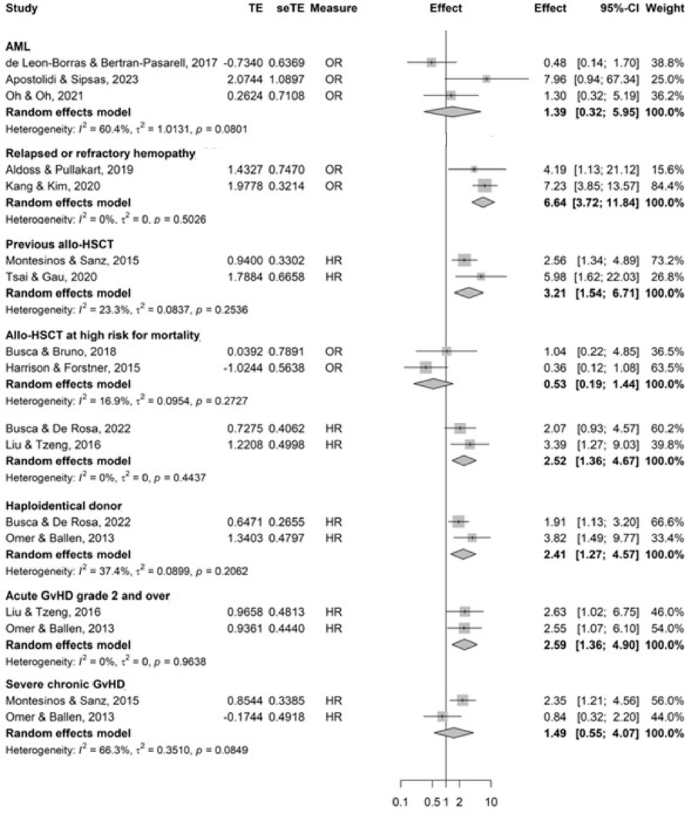
Prolonged neutropenia (> 10 days) was significantly associated with PP-IFI (pooled HR with 2 studies 2.71 [95% CI 1.35–5.44]), although when OR was the association measure the results were not significant but with a high heterogeneity (pooled OR with 2 studies 6.72 [95%CI 0.71–63.55], with substantial heterogeneity, I2 = 79%, phet = 0.03) (Fig. 4).
In addition, neutropenia, regardless of duration, was significantly associated with PPP-IFI (pooled OR with 3 studies 2.79 [95% CI 1.53–5.09]) (additional file 15).
Liver dysfunction was significantly associated with PPP-IFI (OR 3.16 [95%CI 1.14–8.74] with 2 studies) (additional file 15).
Treatment-related risk factors
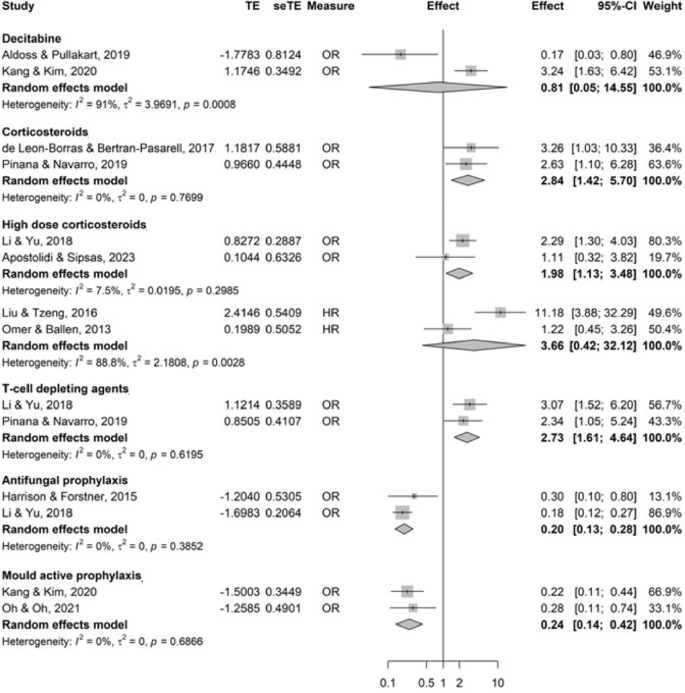
Decitabine was not significantly associated with PP-IFI, although it should be noted that there was considerable heterogeneity (OR 0.81 [95% CI 0.05–14.55] with 2 studies, I2 = 91%, phet < 0.01). Corticosteroids were significantly associated with PP-IFI (OR 2.84 [95% CI 1.42–5.70] with 2 studies) (Fig. 5). High dose corticosteroids were significantly associated with PP-IFI and PPP-IFI when considering OR as the association measure (pooled OR with 2 studies 1.98 [95% CI 1.13–3.48]; pooled OR with 5 studies 2.25 [95% CI 1.59–3.19]) but not with PP-IFI when considering HR (pooled HR with 2 studies 3.66 [95% CI 0.42–32.12], I2 = 89%, phet < 0.01). T-cell depleting agents were significantly associated with PP and PPP-IFI (pooled OR 2.73 [1.61–4.64], and pooled HR 2.00 [95% CI 1.07–3.76], with 2 studies respectively) (Fig. 5 and additional file 16).
Antifungal prophylaxis was a protective factor for PP-IFI (pooled OR with 2 studies 0.20 [95% CI 0.13–0.28]), including mould-active prophylaxis (pooled OR with 2 studies 0.24 [95% CI 0.14–0.42]) (Fig. 5).
Factors related to the hospital stay
Deep vein catheterization was statistically associated with PPP-IFI (pooled OR with 2 studies 3.11 [95%CI 1.27–7.63]) (additional file 17).
Sensitivity analyses and quality of evidence
Sensitivity analyses after exclusion of studies at high risk of bias did not significantly modify the results (additional file 18–25). Other sensitivity analyses performed, excluding studies with children and studies using episodes, gave similar results when data could be pooled (additional files 26–33).
Quality of evidence is summarized in additional file 34. There was a moderate quality of evidence for the association between IFI and corticosteroids and antifungal prophylaxis while the quality of evidence was low for haploidentical donor, acute GvHD, neutropenia, and T-cell depleting agents and very low for age > 60 years old, history of IFI, relapsed or refractory hemopathy, previous HSCT, a HSCT at high risk for mortality, and liver dysfunction.