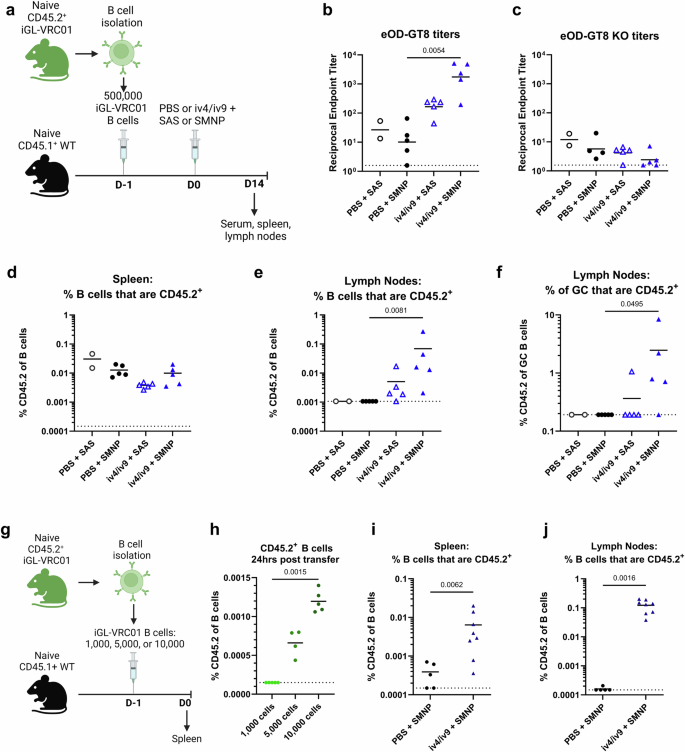
Optimizing an adoptive transfer model to test bispecific iv4/iv9
Using an adoptive transfer model where 500,000 iGL-VRC01 cells expressing the CD45.2 allele were engrafted into Wild-type (WT) mice expressing the CD45.1+ allele, we previously demonstrated that immunization with a bispecific germline targeting ai-mAb, iv4/iv9, formulated with sigma adjuvant system (SAS), successfully activated target B cells, initiated antigen-specific GC responses, and generated low serum binding titers to the VRC01 class germline-targeting immunogen eOD-GT834. However, under these conditions, we did not observe an increase in the overall frequency of transferred CD45.2+ cells after iv4/iv9 immunization34.
Therefore, we investigated whether formulation of iv4/iv9 with another adjuvant could more efficiently expand target B cells in the same adoptive transfer model. We evaluated SMNP, a nanoparticle adjuvant derived from saponin and monophosphoryl Lipid A, known to elicit larger GC and serum antibody responses than SAS60. Wild-type (WT) mice received 500,000 CD45.2+ iGL-VRC01 B cells34,40 on day -1 (D-1) (Fig. 1a). The next day (D0), they were immunized via intra-muscular (I.M.) injection in the quadriceps with PBS (negative control) or iv4/iv9, formulated with either SAS or SMNP. 14 days later, VRC01-class serum titers were assessed by measuring binding to eOD-GT8 and eOD-GT8 with mutations that disrupt VRC01 class binding (eOD-GT8 KO) by ELISA30. Additionally, CD45.2+ B cell populations were monitored in the spleen and lymph nodes using flow cytometry. Immunization with iv4/iv9 formulated with SMNP elicited eOD-GT8 serum antibody binding titers that were ~90-fold higher than those elicited with SMNP alone, while there was no statistical difference between iv4/iv9 formulated with SAS and SAS alone (Fig. 1b). All mice had low to absent titers to eOD-GT8 KO, indicating that the eOD-GT8-binding serum antibodies were against the CD4-BS (Fig.1c). Immunization of WT CD45.1 mice, lacking iGL-VRC01 B cells, with iv4/iv9 did not elicit eOD-GT8-binding serum antibodies (Supplementary Fig. 1), confirming that the measured eOD-GT8 titers arose from the transferred iGL-VRC01 cells (Fig. 1c). Neither regimen expanded the frequency CD45.2+ B cells in the spleen (Fig. 1d), while formulation with SMNP led to a significant expansion in the lymph nodes (LN, Fig. 1e). CD45.2+ B cells were observed among germinal center (GC) B cells in the LN of 4/5 mice immunized with iv4/iv9 + SMNP, while this population was only present in 1/5 mice immunized with iv4/iv9 formulated with SAS (Fig. 1f). Collectively, these data show that formulation of iv4/iv9 with SMNP elicits superior CD45.2+ iGL-VRC01-origin B cell and serum antibody responses as compared to SAS.
a–f CD45.1+ wild-type mice (WT) received 500,000 CD45.2+ iGL-VRC01 cells, followed by immunization with iv4/iv9 or PBS formulated with either sigma adjuvant system (SAS) or Saponin/Monophosphoryl Lipid A nanoparticle (SMNP) the next day as indicated. Blood, spleens, and lymph nodes were collected 14 days later (a). ELISA was used to measure total serum IgG titers against eOD-GT8 (b) and eOD-GT8 KO (c). Frequency of CD45.2+ cells as proportion of total B cells in the spleen (d) or lymph nodes (e). Frequency of CD45.2+ cells as a proportion of germinal center (GC) cells in the lymph nodes (f). g CD45.1+ mice received indicated number of CD45.2+ iGL-VRC01 cells. h 24 h later, the frequency of CD45.2+ cells present in the spleens was analyzed by flow cytometry. P values are reported as determined by the Kruskal–Wallis test with Dunn’s multiple comparisons, and the dashed line indicates half the lowest dilution tested. i and j CD45.1+ WT mice received 5000 CD45.2+ iGL-VRC01 cells and were immunized 24 h later with iv4/iv9 + SMNP or PBS + SMNP. The frequency of CD45.2+ cells was measured in the spleen (i) or lymph nodes (j) 14 days later. Each data point represents one mouse. P values determined by Mann-Whitney tests. a and g were created using BioRender.
Transfer of 500,000 CD45.2+ iGL-VRC01 B cells results in a frequency of ~1 in 20,000 among B-splenocytes at the time of immunization 24 h later34. This is likely supraphysiological since estimates of the frequency of VRC01 precursor B cells in humans range from ~1 in 10,000 to ~1 in 500,00030,61,62,63. We therefore sought to establish transfer conditions under which CD45.2+ iGL-VRC01 B cells are present at a more physiological frequency. We transferred 1000, 5000, or 10,000 CD45.2+ iGL-VRC01 B cells into CD45.1+ WT mice and measured their frequency in the spleen one day later (Fig. 1g). Although we were unable to reliably detect CD45.2+ cells when 1000 were transferred, delivery of 5000 and 10,000 cells resulted in measurable frequencies of 0.006%, or ~1 in 200,000 and 0.001% or ~1 in 100,000 B cells, respectively (Fig. 1h).
We next assessed whether iv4/iv9 + SMNP could effectively stimulate CD45.2+ iGL-VRC01 B cells when they are present at a more physiological frequency. CD45.1+ WT mice received 5000 CD45.2+ iGL-VRC01 cells and were immunized with iv4/iv9 + SMNP or adjuvant alone 24 h later. 14 days post immunization, we observed a 16-fold expansion of CD45.2+ B cells in the spleen that was statistically significant (Fig. 1i), and a significant expansion of CD45.2+ B cells in the LN (Fig. 1j) relative to the adjuvant control, confirming successful targeting of CD45.2+ iGL-VRC01 B cells. Given these results, all subsequent adoptive experiments were carried out in mice that received 5000 CD45.2+ iGL-VRC01 cells and immunizations were formulated with SMNP as adjuvant.
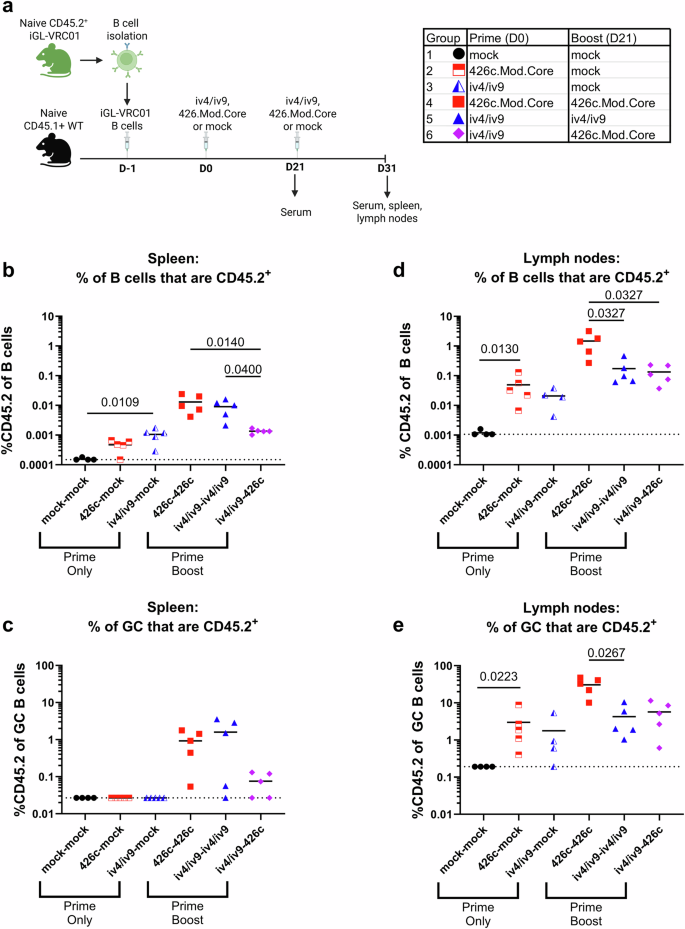
Comparing B cell responses to homologous and heterologous prime boost regimens
Next, we sought to test whether priming with a germline targeting ai-mAb would expand an initial pool of VRC01 class B cells and avoid off-target B cell responses directed at non-VRC01 epitopes on an Env-derived immunogen. We further sought to test whether this strategy would confer a competitive advantage to VRC01-class B cells over the more abundant off-target B cells during subsequent immunizations with a conventional germline targeting Env-derived immunogen. CD45.1+ WT mice harboring CD45.2+ iGL-VRC01 cells were immunized with either iv4/iv9, 426c.Mod.Core (herein called 426c), or PBS + SMNP (mock). Three weeks later, they received a second immunization with the same immunogen used in the prime (homologous boost), while one group of animals primed with iv4/iv9 were boosted with 426c (heterologous boost, Fig. 2a). Both immunogens were C-terminally tagged with a linear peptide, 2W1S, which is a known CD4+ T cell epitope in the C57BL/6 background64. We previously used this peptide fusion strategy to improve B cell responses elicited by an ai-mAb32 and reasoned that conjugating this peptide to both iv4/iv9 and 426c would ensure that there was at least one shared helper T cell epitope between the prime and boost immunogens to facilitate the acquisition of help from memory CD4+ T cells. Additional groups of animals were primed with iv4/iv9 or 426c at D0 but mock boosted with PBS + SMNP and were analyzed at day 31 as controls.
a Schematic of the experiment. CD45.1+ mice received 5000 CD45.2+ iGL-VRC01 cells. The next day (D0), they were immunized with either iv4/iv9-2W1S or 426c.Mod.Core-2W1S formulated with SMNP (n = 5 per group) or mock immunized (PBS + SMNP, n = 4 per group). 21 days later, the mice were bled and immunized a second time with the indicated immunogen. The mice were then euthanized 10 days later (D31). Created with BioRender. b Frequency of CD45.2+ cells as a proportion of total B cells in the spleen. c Frequency of CD45.2+ cells as a proportion of germinal center (GC) B cells in the spleen. d Frequency of CD45.2+ cells as a proportion of total B cells in the lymph nodes. e Frequency of CD45.2+ cells as a proportion of germinal center (GC) B cells in the lymph nodes. Each data point represents one mouse, bars represent the mean, and dashed lines indicate the limit of detection (LoD) in (b–e). P values are reported as determined by the Kruskal–Wallis test with Dunn’s multiple comparisons within each group. Statistical tests were performed separately on the Prime Only and Prime Boost groups, indicated by brackets in (b–e).
A single immunization with 426c or iv4/iv9 followed by mock boost led to a slight expansion of CD45.2+ B cells in the spleen measured at the time of euthanasia (D31, Fig. 2b). A second immunization with the same antigen used in the prime (homologous boost) further increased the frequency of CD45.2+ B cells to comparable levels in the spleen (Fig. 2b). The 426c homologous boost regimen induced splenic CD45.2+ GC responses (Fig. 2c), while boosting with 426c did not increase the frequency of CD45.2+ B cells that were primed by iv4/iv9 in the spleen (Fig. 2b), however a small population of splenic CD45.2+ GC B cells was present in some animals following the iv4/iv9 prime- 426c boost (Fig. 2c).
In the lymph nodes, a single immunization with either iv4/iv9 or 426c followed by a mock boost, led to a slight expansion of CD45.2+ B cells (Fig. 2d) and induction of CD45.2+ GC responses (Fig. 2e). For both antigens, a homologous boost further expanded CD45.2+ cells (Fig. 2d). This expansion was significant and ~10-fold higher in the homologous 426c prime-boost regimen as compared to the homologous iv4/iv9 prime-boost regimen (Fig. 2d). The homologous 426c regimen also elicited the highest frequency of CD45.2+GC B cells in the lymph nodes compared to the iv4/iv9 homologous regimen (Fig. 2e). The frequency of CD45.2+ B cells among all (Fig. 2d) and GC (Fig. 2e) lymph node B cells in the heterologous iv4/iv9–426c regimen were comparable to the homologous iv4/iv9 regimen.
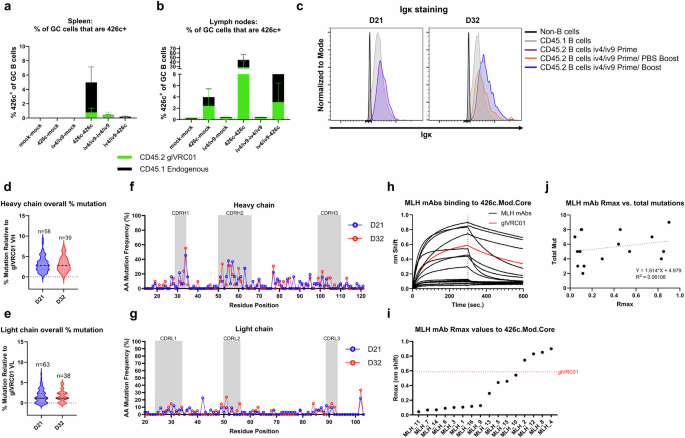
ai-mAb immunization induces somatic mutations that negatively impact 426c recognition by iGL-VRC01 B cells
To quantify on-target CD45.2+ B cells of iGL-VRC01 origin and off-target CD45.1+ 426c-specific GC B cell responses of WT origin, we stained GC B cells collected from the spleen and lymph nodes from the mice in Fig. 2 at D31 with 426c-tetramers, using a dual-labeling strategy. In the spleen, we observed the highest frequency, ~6%, of 426c + GC B cells in the group that received two immunizations with 426c (Fig. 3a). A minority of these, ~0.8%, were CD45.2+ (Fig. 3a). In mice immunized with a homologous iv4/iv9 prime-boost regimen, ~0.3% of the splenic GC B cells were 426c+ and nearly all were CD45.2+. The heterologous iv4/iv9-426c regimen elicited a very low frequency of 426c + B cells, most of which were off-target CD45.1 cells (Fig. 3a). The low frequency of CD45.2+ cells among 426c+ B cells in the iv4/iv9-426c regimen is consistent with smaller expansion of total CD45.2+ GC B cells in this group as compared to the homologous prime-boost groups (Fig. 2c).
a and b Proportion of GC B cells from mice in Fig. 2 that stain 426c.Mod.Core+ in spleen (a) and lymph nodes (b); the height of the bars indicates the mean total frequency of 426c.Mod.Core+ GC cells, and the colors indicate whether they are of CD45.1+ (black) or CD45.2+ (green) origin. Bars represent the mean of n = 5 mice per group, and the error bars show the standard deviation. c Overlaid histograms showing IgK staining by flow cytometry of indicated cell populations at the indicated timepoints. Data normalized to mode. d–g Sequence and mutation analysis of single-cell sorted CD45.2+ B cells from the lymph nodes of iv4/iv9 prime, iv4/iv9 boost animals at D21 and D32. d and e Violin plots of overall percent mutation in the heavy chain (IgH, d) or light chains (IgK, e) compared to iGL-VRC01 with the number of chains analyzed above each plot. h–j Binding of 16 recombinant mAbs corresponding to BCRs from sorted cells was assessed for binding to a 500 nM solution of 426c.Mod.Core by BLI. h Representative BLI traces of sorted mAbs (black) compared to iGL-VRC01 IgG (red). i Rmax values of sorted mAbs to 426 c.Mod.Core from (h). Rmax of iGL-VRC01 to 426c.Mod.Core shown by the red dotted line. j Linear regression analysis of Rmax values from (i) vs. number of mutations relative to iGL-VRC01.
In the lymph nodes, the 426c-426c regimen elicited the highest frequency of total antigen-specific GC B cells, ~45%, most of which (~30%) were CD45.2+ (Fig. 3b). In comparison, the heterologous iv4/iv9 prime-426c boost yielded a lower frequency of 426c + GC B cells in the lymph nodes (~7%), with the majority being off-target (Fig. 3b). Although the iv4/iv9-iv4/iv9 and iv4/iv9-426c regimens led to a comparable expansion of CD45.2+ B cells in the LN GCs at D31 (Fig. 2e), 426c+/CD45.2+ LN GC B cells were not detected in mice immunized twice with iv4/iv9 (Fig. 3b) suggesting that immunization with iv4/iv9 negatively affects subsequent CD45.2+ B cell binding to 426c. In support of this notion, we observed that only ~30% of splenic CD45.2+ B cells in mice immunized twice with iv4/iv9, bound 426c, while ~70–80% bound 426c in all other groups (Supplementary Fig. 2a). Similarly, ~70–80% of lymph node CD45.2+ B cells bound 426c in animals immunized once or twice with 426c while only ~20% bound when the animals were immunized once or twice with iv4/iv9 (Supplementary Fig. 2b).
To better understand the mechanism underlying the reduced binding of CD45.2+ GC B cells to 426c following iv4/iv9 immunization, we repeated the iv4/iv9 prime only, and iv4/iv9 prime-boost immunizations (i.e., Fig. 2a, Groups 3 and 5) and focused our analyses on LN B cells. Igκ staining was observed on non-iGL VRC01 (CD45.1+) and CD45.2+ iGL-VRC01 origin cells at the time of the second immunization (D21) or 11 days after a second immunization (D32), indicating that the reduced 426c binding observed in GC LNs is not attributed to loss of BCR expression on the cell surface (Fig. 3c).
Lymph node cells from iv4/iv9 immunized mice were pooled, and CD45.2+ GC B cells were subjected to antigen-unbiased single-cell sorting (Supplementary Fig. 3), and their variable heavy (VH) and variable light (VL) BCR transcripts were recovered by RT-PCR and sequenced. Among all recovered transcripts, we observed higher frequencies of heavy chain mutations as compared to light chain mutations with no statistical differences in these frequencies between the time of the boost (D21) and 11 days later (D32, Fig. 3d and e). Most of the mutations were found in the CDR1 and CDR2 regions of the heavy and light chains (Fig. 3f and g, Supplementary Fig. 4, and Supplementary Tables 2–4). 22/62 or 35% of paired sequences had at least one mutation found in VRC01 class antibodies. We randomly selected 16 representative heavy and light chain pairs (roughly 25% of the total pairs recovered), four from each quartile based on the HC mutation rate (Fig. 3d and e), isolated at either D21 or D32 (see Supplementary Fig. 4 for more details). This included some pairs that had mutations found in mature VRC01 (Supplementary Figs. 4 and 5, and Supplementary Tables 2–4). These pairs were produced as recombinant mAbs and evaluated for binding to 500 nM of 426c by biolayer interferometry (BLI). This analysis revealed that some mAbs clearly had reduced, or absent binding to 426c as compared to iGL-VRC01 with discernible differences in the association and dissociation rates (Fig. 3h). Using the maximum response (Rmax) measured at the end of the dissociation phase as a proxy for binding affinity we found that only 4/16 (25%) had stronger binding than iGL-VRC01 (Fig. 3i). 8 (50%) had negligible binding to 426c, while 4 (25%) retained some binding Fig. 3i). There was no obvious correlation between Rmax and the frequency of total (Fig. 3j), heavy (Supplementary Fig. 6a), or light (Supplementary Fig. 6b) chain mutations. 4 of the mAbs had mutations in VH1-2*02 encoded contact residues, W50, N58, R71, and W100B found in potent VRC01 like antibodies as defined in ref. 65 (Supplementary Fig. 4a), all of which showed near-complete abrogation of 426c binding (Supplementary Fig. 6c). Among the recombinant mAbs we produced, 4 acquired heavy chain mutations found in mutated VRC01: MLH_2 G30D, MLH_4 Y91F, MLH_6 Q105R and MLH_11 I51L (Supplementary Fig. 4a). Among these, only MLH_2 and MLH_4 showed enhanced binding to 426c.Mod.Core, while the binding of the other two was reduced (Fig.3i). Among the mAbs that bound 426c.Mod.Core only one, MLH_2 acquired a key VRC01 class mutation (G31D), but it lost a germline-encoded germline contact residue (R66W), Supplementary Fig. 5 and Supplementary Tables 2 and 3). None of the mAbs that lost affinity acquired key VRC01-class mutations (Supplementary Figs. 4 and 5, and Supplementary Tables 2 and 3). Interestingly, mAbs that lost affinity to 426c do not appear to gain affinity towards iv4 or iv9, but most maintain binding as determined by BLI (Supplementary Fig. 6d–h).
Collectively, these analyses reveal quantitative and qualitative differences in the frequencies of CD45.1 and CD45.2 B cell responses measured in the spleen and lymph nodes. They also indicate that in this model, the homologous 426c prime-boost regimen, despite eliciting off-target B cell responses, elicited a higher frequency of on-target B cell responses (total and GC) as compared to a homologous iv4/iv9, or heterologous iv4/iv9-426c boost. These data also indicate that immunization with the ai-mAb induces BCR mutations that negatively impact binding to 426c.
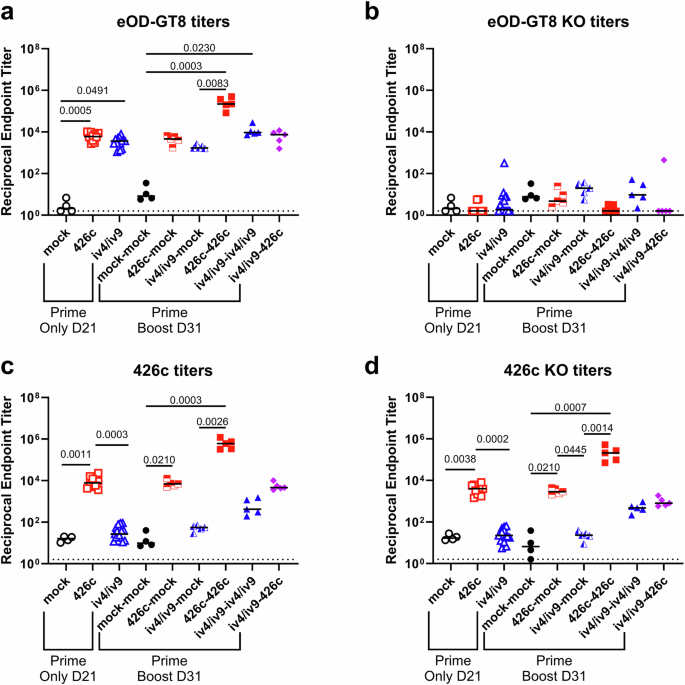
Comparing plasma responses to homologous and heterologous prime-boost regimens
To assess both circulating VRC01-epitope-specific (on-target) and non-epitope-specific (off-target) antibody responses from immunized animals in Fig. 2, serum binding titers to eOD-GT8 and eOD-GT8-KO, as well as 426c and 426c-KO were measured by ELISA (Fig. 4). A single immunization with either iv4/iv9 or 426c elicited epitope-specific antibodies to eOD-GT8 (D21, Fig. 4a). In contrast, only mice immunized with 426c showed strong binding titers to 426c (Fig. 4c) after a single immunization. We note that the affinity of iGL-VRC01 for eOD-GT8 is in the pM range30 while 426c is 0.3µM28, making eOD-GT8 a more sensitive reagent for detecting serum responses in this assay. The same mice had slightly lower (~2-fold) titers to 426c-KO (Fig. 4d and Supplementary Fig. 7). Control (CD45.1) mice that were immunized with 426c without receiving CD45.2+ iGL-VRC01 B cells had lower plasma antibody binding titers to 426c (~2.6-fold) and 426c-KO (~2.2-fold) relative to those harboring iGL-VRC01 (Supplementary Fig. 7). The titers were ~1.8-fold lower to 426c-KO relative to 426c in the no transfer control group (Supplementary Fig. 7). Collectively these data indicate that a substantial fraction of the circulating antibodies in 426c immunized mice are directed at off-target non-CD4-BS epitopes irrespective of the presence of CD45.2+ iGL-VRC01 B cells.
a–d Serum was collected at D21 and D31 from the experiment shown in Fig. 2 and analyzed by ELISA. Endpoint binding titers were measured to eOD-GT8 (a), eOD-GT8-KO (b), 426c.Mod.Core (c), and 426c.Mod.Core-KO (d). Dashed line indicates half of the lowest dilution tested. Each data point represents one mouse. n = 5 mice per group, except the PBS only control group, where n = 4 mice. Mean is plotted. P values are reported as determined by the Kruskal–Wallis test with Dunn’s multiple comparisons. Statistical tests were performed separately on the Prime Only and Prime Boost groups, indicated by brackets.
Titers to eOD-GT8 significantly increased in groups primed with either iv4/iv9 or 426c followed by a homologous boost (Fig. 4a). However, titers to 426c only significantly increased in animals that received homologous 426c prime-boost, while those that received iv4/iv9 homologous prime-boost increased but did not reach statistical significance (Fig. 4c). In contrast, mice which received an immunogen at D0 but were mock-boosted with PBS + SMNP at D21 had unchanged titers to eOD-GT8 and 426c at D31 confirming that the measured changes in serum titers at D31 were due to the second immunization with either iv4/iv9 or 426c (Fig. 4a and c). The homologous 426c prime-boost regimen elicited the highest binding titers to eOD-GT8, and to both 426c and 426c-KO (Fig. 4a, c, and d), consistent with the expansion of both on-target and off-target B cell populations among GC B cells in the lymph nodes (Figs. 2e and 3b).
Animals that received a heterologous iv4/iv9 prime-426c boost regimen had considerably higher 426c-specific titers compared to animals that were only primed with iv4/iv9 (Fig. 4c). However, the titers to 426c-KO also increased after the boost in the iv4/iv9prime-426c-boost group (Fig. 4d), while the eOD-GT8 titers remained unchanged (Fig. 4a), suggesting that most of the serum responses elicited by the second 426c immunization were de novo responses directed at off-target epitopes.
Circulating on-target antibodies modestly affect the priming of iGL-VRC01 B cells
Similar adoptive transfer systems have demonstrated that circulating antibodies can inhibit or enhance GC populations of naive B cells depending on BCR affinity, epitope specificity and antibody titer48,56. We observed that priming with iv4/iv9 and 426c both elicited high titers of on-target iGL-VRC01-origin antibodies (Fig. 4a), while the latter also elicited high titers of off-target non-VRC01 antibodies, which includes those that bind outside and within the CD4-BS (Fig. 4d and Supplementary Fig. 7). We therefore sought to investigate the influence of these circulating on- and off-target antibodies on the B cell recall responses elicited upon boosting in this model.
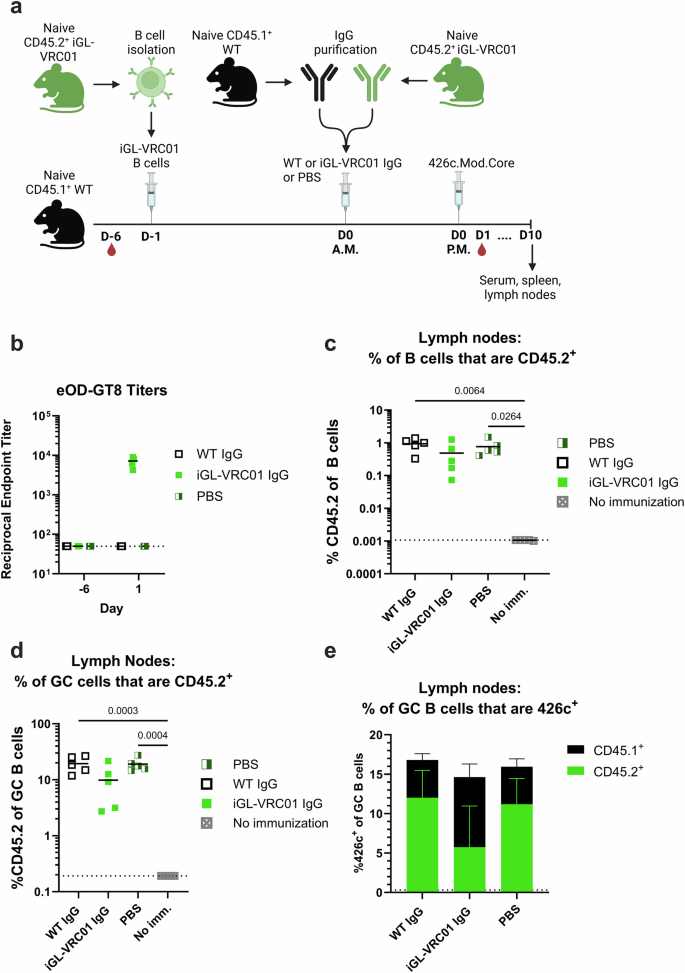
We first investigated whether elevated titers of circulating iGL-VRC01 antibodies impacted the ability of the CD45.2+ iGL-VRC01 B cells to respond to immunization with 426c. A pre-bleed was collected on recipient WT CD45.1+ mice; and then 6 days later, they received CD45.2+ iGL-VRC01 B cells on day -1 (D-1). The next morning (D0, A.M.), they received PBS, or 500 µg of IgG purified from naive iGL-VRC01 mice or naïve wildtype mice. ~8 hours later (D0, P.M.), they were immunized with 426c. The timing of the IgG immunization and transfer was done to minimize the effects of antibody decay from serum and is based on a similar adoptive transfer model48. Serum was then collected on D1, and the mice were euthanized nine days later (Fig. 5a).
a Schematic of the experiment. CD45.1+ WT mice (n = 5 per group) received 5000 CD45.2+ iGL-VRC01 cells (D-1). The next morning (D0 A.M.), they received IgG purified from naïve CD45.1+ WT mice, iGL-VRC01 mice or PBS, followed by an immunization with 426c.Mod.Core in the afternoon (D0 P.M.). Created with BioRender. b Endpoint binding titers to eOD-GT8 were measured by ELISA in serum collected from the indicated groups on D-6 and D1. Dashed line indicates half the lowest dilution tested. c Frequency of total B cells that are CD45.2+ in the lymph nodes at day 10. d Frequency of germinal center (GC) B cells that are CD45.2+ in the lymph nodes at day 10. Each data point represents one mouse (n = 5), and the mean is indicated by a bar. P values are reported as determined by the Kruskal–Wallis test with Dunn’s multiple comparisons. Dashed lines indicate the limit of detection in (b–d). e Mean percentage of 426c.Mod.Core+ GC B cells in the lymph node at day 10. The height of the bar indicates the total frequency of 426c.Mod.Core+ cells, and the colors indicate whether they are CD45.1+ (black) or CD45.2+ (green). Bars represent the mean of n = 5 mice per group, and the error bars represent the standard deviation in (e).
Sera from mice that received iGL-VRC01 IgG had no eOD-GT8 binding titers at baseline (D-6); while at one day post transfer, they were comparable to those observed at day 21 in mice primed with either iv4/iv9 or 426c alone (compare Figs. 5b and 4a), indicating that the IgG transfer was successful. 10 days after 426c-immunization, we observed a comparable frequency of CD45.2+ B cells in the lymph nodes of animals that received either PBS, or control WT IgG (Fig. 5c). Mice which received iGL-VRC01 IgG had marginally lower frequencies of CD45.2+ cells in the lymph nodes; both as a proportion of total B cells and of GCs as compared to the control groups, however the differences were not statistically significant (Fig. 5c and d).
The frequency of 426c-specific GC B cells was comparable in all 3 groups at D10 (Fig. 5e). ~66% of these were CD45.2+ B cells in the PBS and control IgG transfer groups, while only ~33% were CD45.2+ B cells in mice that received iGL- VRC01 IgG prior to immunization (Fig. 5e). We did not observe any difference in the frequency of iGL-VRC01 B cells in the spleens of immunized mice relative to unimmunized mice (Supplementary Fig. S8c–e). Collectively, these results indicate that under these experimental conditions, pre-existing iGL-VRC01 serum antibodies had a modest inhibitory effect on CD45.2+ iGL-VRC01 responses in the lymph nodes of immunized animals.
Circulating on-target antibodies do not affect the ability to boost iGL-VRC01 B cells
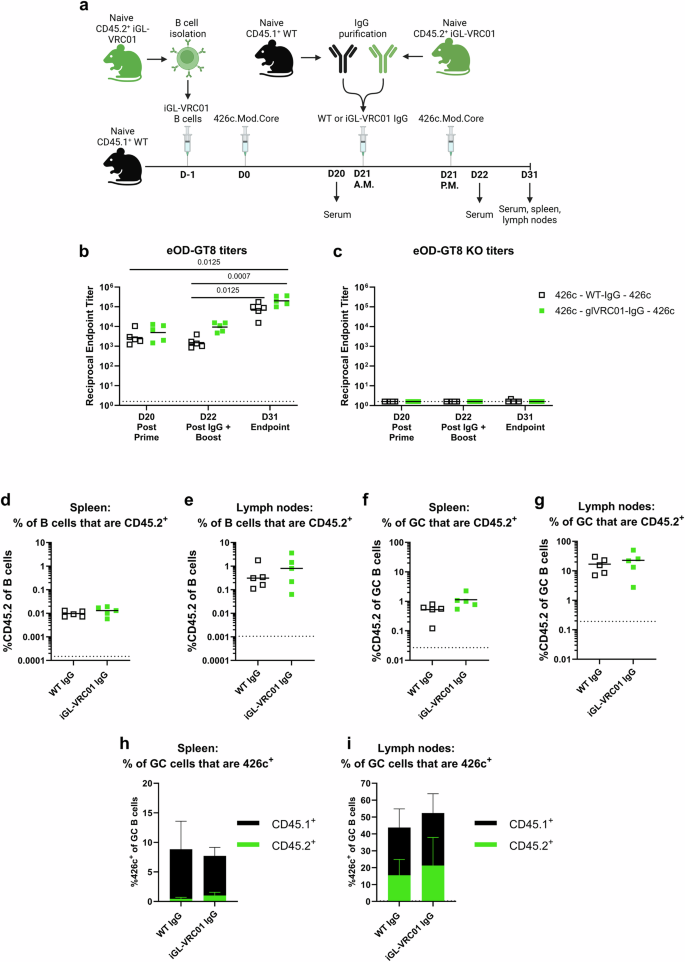
We next asked whether supraphysiological titers of on-target antibodies affect iGL-VRC01 B cell responses to a homologous boost (Fig. 6). Here, CD45.2+ iGL-VRC01 cells were transferred into CD45.1+ WT mice on D-1. The next day (D0), they were immunized with 426c. Three weeks later (D21), the mice received purified IgG from either naïve WT CD45.1+ or CD45.2+ iGL-VRC01 mice and were then immunized with 426c a second time (Fig. 6a). Mice which received IgG from iGL-VRC01 donors had higher titers to eOD-GT8 at D22 than those which received WT-IgG (~6 fold higher), while none of the plasma bound to eOD-GT8 KO consistent with successful antibody transfer (Fig. 6b and c).
a Schematic of the experiment. CD45.1+ WT mice received 5,000 iGL-VRC01 CD45.2+ cells and were immunized the next day (D0) with 426c.Mod.Core. Twenty-one days later (D21), they received IgG from naïve CD45.1+ WT or from iGL-VRC01 mice in the morning (A.M.), followed by an immunization with 426c.Mod.Core in the afternoon (P.M.). Created with BioRender. b and c Endpoint binding titers to eOD-GT8 (b) or eOD-GT8 KO (c) were measured by ELISA in serum collected on D20, D22, and D31. Dashed line represents half of the lowest dilution tested. d and e Frequency of total CD45.2+ B cells in the spleen (d) and lymph nodes (e) at D31. f and g Frequency of CD45.2+ B cells among GC B cells in the spleen (f) and lymph nodes (g) at D31. Each data point represents one mouse (n = 5). P values are reported as determined by the Kruskal–Wallis test with Dunn’s multiple comparisons, and dashed lines indicate limit of detection (d–g). h and i Percentage of 426c.Mod.Core+ B cells among GC B cells in the spleen (h) and lymph nodes (i) at day 31. Bars represent the mean of n = 5 mice per group, and the error bars represent the standard deviation. The height of the bar indicates the total frequency of 426c.Mod.Core+ cells, and the colors indicate whether they are CD45.1+ (black) or CD45.2+ (green).
Following the second immunization with 426c, we observed an increase in eOD-GT8 titers from D22 to D31, in both the control WT IgG and iGL-VRC01 IgG transfer groups (Fig. 6b), indicating that transfer of on-target IgG did not inhibit the serum antibody response to a homologous boost. When we assessed the prevalence of CD45.2+ B cells, no differences were observed in the frequencies of total or GC CD45.2+ B cells in the spleen or lymph nodes (Fig. 6d–g and Supplementary Fig. S9). Addition of either control WT or iGL-VRC01 IgG resulted in comparable frequencies of total 426c-specific GC B cells in both the spleen and lymph nodes (Fig. 6h and i). Transfer of iGL-VRC01 IgG resulted in a slightly higher proportion of CD45.2+ 426c-specific B cells in the spleen and lymph nodes (Fig. 6h and i). Collectively, these data demonstrate that in this experimental setting, administration of supraphysiological levels of iGL-VRC01 IgG following a 426c prime does not negatively affect CD45.2+ iGL-VRC01 B cell responses to a homologous boost.
Circulating off-target antibodies promote on-target iGL-VRC01 B cell responses
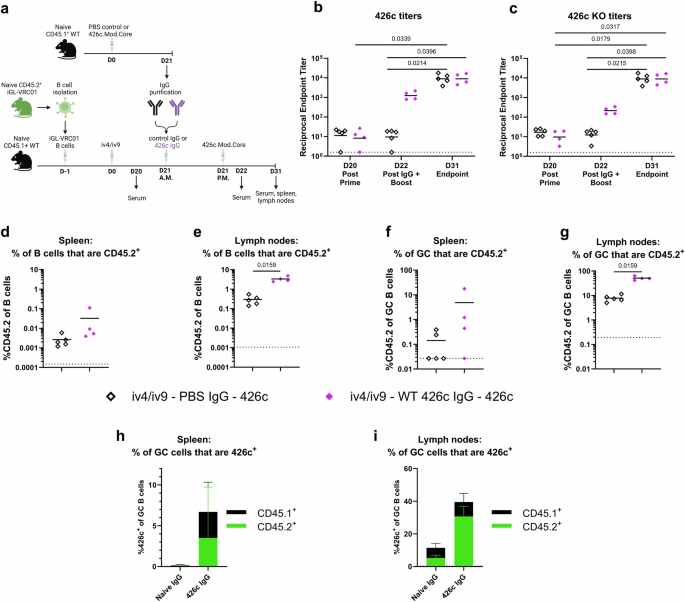
To further evaluate the effect of pre-existing antibodies on the iGL-VRC01 B cell response to immunization, we investigated the influence of off-target serum antibodies to 426c on iGL-VRC01 B cell recall responses following an iv4/iv9 prime. To generate off-target antibodies, CD45.1+ WT mice lacking CD45.2+ iGL-VRC01 B cells were immunized with 426c. Three weeks later, the serum was collected. We verified that both CD4-BS and non-CD4-BS antibodies were present in the serum (Supplementary Fig. 7), and the IgG was subsequently purified. IgG purified from CD45.1+ WT mice mock immunized with PBS + SMNP was collected as a control. Experimental groups of CD45.1+ WT mice harboring CD45.2+ iGL-VRC01 B cells were immunized with iv4/iv9. Three weeks later (D21) in the morning, they received either control IgG from naïve WT mice or off-target 426c-elicited, followed by an immunization with 426c in the afternoon. Serum was collected from the recipients the day before (D20) and the day after (D22) the second immunization (Fig. 7a). Prior to IgG transfer, the 426c and 426c-KO titers were equivalent (Fig. 7b and c). At D22, mice that received 426c -elicited IgG (solid purple diamonds) had higher titers to both 426c and 426c-KO than those that received control IgG (open black diamonds), consistent with a successful transfer of antibody (Fig. 7b and c). At this timepoint, the titers were approximately 10-fold higher to 426c than they were to 426c-KO, indicating that both CD4-BS and non-CD4-BS antibodies were transferred. At the endpoint (D31), the serum titers to 426c and 426c -KO were boosted to equivalent titers in both groups, indicating that the second 426c immunization elicited 426c-specific serum responses that augmented those from the prime and the IgG transfer. The addition of off-target IgG, between the prime and the boost, resulted in a slight increase in the frequency of total and GC CD45.2+ B cells in the spleen that was not significant (Fig. 7d and f, and Supplementary Fig. 10). Meanwhile, the addition of off-target IgG resulted in a significantly higher frequency of total and GC CD45.2+ B cells in the lymph nodes (Fig. 7e and g and Supplementary Fig. 10). There, almost half of the GC B cells were CD45.2+, while only ~5% were CD45.2+ in the mice that received control IgG (Fig. 7g).
a Schematic of the experiment. CD45.1+ wild-type mice received 5000 CD45.2+ iGL-VRC01 B cells and were immunized the next day (D0) with iv4/iv9. Twenty-one days later (D21), they received IgG from CD45.1+ wild-type mice immunized with PBS + SMNP (control, black n = 5) or with 426c.Mod.Core (purple n = 4) in the morning (A.M.), followed by an immunization with 426c.Mod.Core in the afternoon (P.M.). Created with BioRender. b and c Endpoint binding titers to 426c.Mod.Core (b) and 426c.Mod.Core KO (c) was measured by ELISA in serum collected on D20, D22, and D31. P values are reported as determined by the Kruskal–Wallis test with Dunn’s multiple comparisons, and the dashed line indicates half the lowest dilution tested. d and e Frequency of total CD45.2+ B cells in the spleen (d) and lymph nodes (e) at D31. f and g Frequency of CD45.2+ B cells among germinal center B cells in the spleen (f) and lymph nodes (g) at D31. Each data point represents one mouse, and bars represent the means. P values are reported as determined by the Mann–Whitney test, and the dashed line indicates the limit of detection (d–g). h and i Percentage of 426c.Mod.Core+ B cells among GC B cells in the spleen (h) and lymph nodes (i) at day 31. Bars represent the mean of n = 5 mice per group in the naïve IgG group and n = 4 in the 426c IgG group, and the error bars represent the standard deviation. The height of the bar indicates the total frequency of 426c.Mod.Core+ cells, and the colors indicate whether they are CD45.1+ (black) or CD45.2+ (green).
The addition of off-target IgG, as compared to control IgG also led to a higher total frequency of 426c+ GC B cells in the spleen and lymph nodes (Fig. 7h, i). These were dominated by CD45.1+ WT B cells in the spleen (Fig. 7h), and by CD45.2+ B cells in the lymph nodes (Fig. 7i). The relative proportion of off-target and of CD45.2+ cells elicited by the heterologous iv4/iv9-426c regimen in the presence of transferred exogenous off-target IgG mirrored those of the homologous 426c prime-boost regimen (Compare Figs. 3a, b, 7h, i and Supplementary Fig. 11). Collectively, these data indicate that circulating polyclonal off-target antibodies to 426c, including those that bind to the CD4-BS, promote the expansion of on-target iGL-VRC01 class B cells and their recruitment to GCs, particularly in the lymph nodes.