Design
To compare the risks of death and incident MACE, such as myocardial infarction or stroke, within 365 days following COVID-19 between individuals fully vaccinated against SARS-CoV-2 and unvaccinated individuals, we specified and emulated a target trial (Supplementary, Table 1)36.
Setting and data sources
We utilized observational data from Estonia’s nationwide electronic health databases. The study period spanned from April 2021 to March 2023. In Estonia, COVID-19 vaccination began in January 2021, with a cumulative vaccination uptake of the primary vaccination series of approximately 70% among the adult population by June 2022. The accrual of COVID-19 cases for the study period coincides with the epidemics of COVID-19 variants Alpha, Delta, and Omicron37.
The Estonian health insurance fund (EHIF)
At the end of 2021, universal public health insurance covered 95.2% of Estonia’s population of 1.3 million38. The EHIF maintains a complete record of the healthcare services provided. Diagnoses are defined according to the International Classification of Diseases, tenth revision (ICD-10). The EHIF database records sex, date of birth, and healthcare utilization information (incl. dates of service, diagnoses, treatment type: in- or outpatient).
Estonian national health information system (ENHIS)
Data on COVID-19 vaccination (dates, the type of vaccine), SARS-CoV-2 testing (dates) and laboratory confirmed (real-time polymerase chain reaction (PCR) or antigen testing) cases of SARS-CoV-2 infections (dates) were retrieved from ENHIS39. According to law, all health-care providers and laboratories in Estonia are obligated to report health claims data to ENHIS. Thus, the expected coverage is 100%.
The population register
Estonian citizens and foreign nationals residing in Estonia are listed in the Population Register maintained by the Ministry of the Interior, based on their residence permit40. This register provided data on the study subjects’ education, native language, and ethnicity.
Estonian causes of death registry
We used data obtained from the Estonian Causes of Death Registry to identify the dates of deaths. The Estonian Causes of Death Registry collects information on all deaths that occur on Estonian territory and in foreign missions41.
The unique personal identification code assigned to all Estonian residents (at birth or immigration) was used to link data between health databases.
Study population
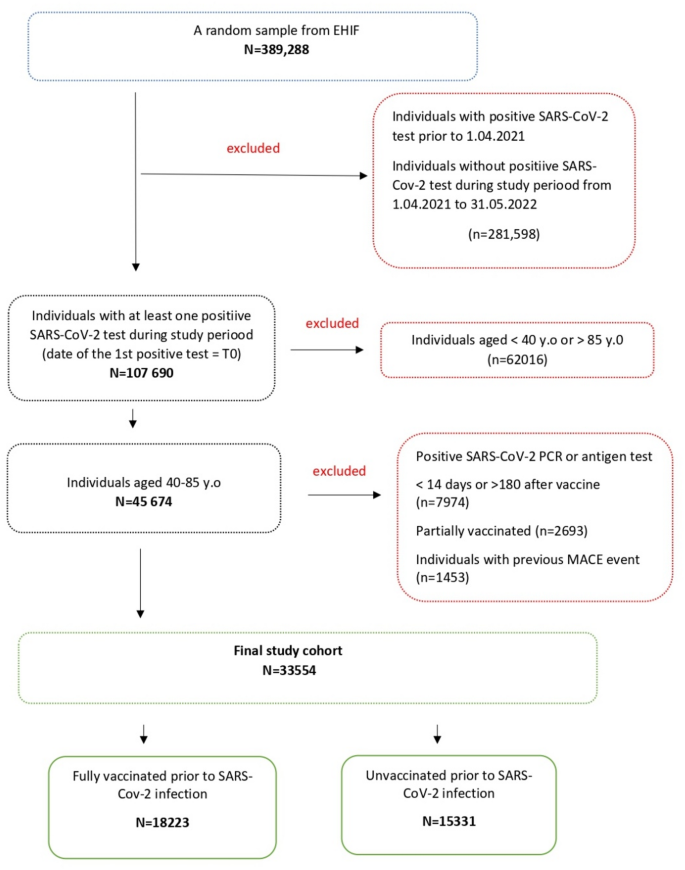
A study cohort was drawn from a random sample of 389,288 individuals covered by the EHIF. The study’s eligibility criteria included individuals aged 40 to 85 years who had a first positive SARS-CoV-2 test between April 1, 2021, and May 31, 2022 (baseline, T0), and who had not received any COVID-19 vaccination before the index date. Individuals with diagnoses of acute cardiovascular events (see Supplementary, Table 2 for clinical diagnoses included in MACE codes, as classified by International Classification of Disease, 10th Revision (ICD-10)) during the five years preceding the index date, or a positive SARS-CoV-2 test < 14 days or > 180 days after COVID-19 vaccination, were also excluded. Only the first positive SARS-CoV-2 test was considered, regardless of possible reinfections (Fig. 3).
Exposure
We considered COVID-19 vaccination status as a treatment (or exposure) in our study. The vaccinated cohort consisted of individuals who completed the primary COVID-19 vaccine series (two doses of BNT162b2 (Pfizer/BioNTech), mRNA-1273 (Moderna), AZD1222 (Oxford/AstraZeneca), or one dose of Ad26.COV2 (Janssen/Johnson & Johnson) 14–180 days before SARS-CoV-2 infection. Supplementary vaccine doses administered after the primary series were not considered in this analysis.
The unvaccinated cohort consisted of individuals who had not received a vaccination before their first SARS-CoV-2 infection.
Outcomes
Primary outcome: incident MACE
MACE (major acute cardiovascular events) is a composite of clinical events previously described in various clinical trials aimed at evaluating the effectiveness and safety of cardiovascular interventions42,43.
We used a two-point MACE outcome, defined as a composite of acute myocardial infarction (AMI) and stroke occurring 0-365 days after the 1 st positive SARS-CoV-2 test, as the primary outcome. Our focus on two-point MACE in COVID-19 sequelae research is driven by its significant impact on individuals and health systems, potentially causing long-term disability, reduced quality of life, and premature death. We defined MACE components based on the ICD-10 codes in EHIF data (MACE diagnosis codes are presented in Supplementary Table 2).
Secondary outcome: All-cause mortality
We defined all-cause mortality as any death occurring 0-365 days after the 1 st positive SARS-CoV-2 test.
Follow-up period
Follow-up began at baseline (T0 or index date; the date of the 1 st positive SARS-CoV-2 test) and continued until the occurrence of outcome (MACE, all-cause death), a competing event (all-cause death for analysis of MACE only), or 365 days post-baseline, whichever occurred first. Thus, we incorporate all symptomatic and asymptomatic COVID-19 cases, as well as outcomes resulting in the acute and post-acute phases of COVID-19.
Covariates
Sociodemographic characteristics
The analysis included the age (in years), sex (male, female), and education level of the study participants. Study participants’ education levels were categorized into three groups: primary (basic education or below), secondary (general secondary or vocational education), and higher education (higher or tertiary education)44.
Pre-COVID-19 comorbidities
We obtained data about diabetes (type 1 and 2), chronic pulmonary diseases, and cardiovascular diseases, recognizing their potential to increase the risk of contracting COVID-19 and experiencing severe disease course, as well as the potential of these diseases to increase the risk of MACE and mortality both during and after the acute phase of infection.
Comorbidities were identified using ICD-10 codes (see detailed description in Supplementary, Table 3), as any primary or secondary diagnosis code in the claim or diagnosis of any type (hospital or outpatient) health care claims during the five years preceding the index date.
Severity of COVID-19
Severe COVID-19 was defined as hospitalization occurring within 3 days before to 14 days after a positive SARS-CoV-2 test. We set additional criteria for COVID-19-related hospitalization to avoid misclassification of COVID-19 cases identified through routine screening during hospitalization for other reasons (e.g., childbirth). The diagnoses associated with hospitalization had to include an ICD-10-based COVID-19 diagnosis (U07.1, U07.2) and at least one additional diagnosis indicating a COVID-19-related condition, such as acute upper or lower respiratory tract infection or acute respiratory failure (Supplementary, Table 4)45.
Causal inference strategies
This study was designed in the target trial emulation framework, as described in the supplementary material (Supplementary Tables 1 and Supplementary discussion)36,46. Briefly, we developed a protocol for a hypothetical target trial in which the treatment is vaccination against COVID-19 or no vaccination; the outcomes are MACE and all-cause death. Our target trial investigates the average causal effect of COVID-19 vaccination on the one-year risk of MACE and all-cause mortality in individuals who have had a SARS-CoV-2 infection. To obtain causal effect estimates, we employed a directed acyclic graph (DAG) – based inverse probability of treatment weighted marginal structural Poisson survival model with flexible time functions to adjust for confounding and capture time-varying effects on outcomes.
This method enabled us to systematically map the assumed causal relationships between exposure (vaccination) and outcomes (MACE or death). In the final DAG (see Supplementary Fig. 2), we identified and incorporated variables that could plausibly confound the association between vaccination and the outcomes.
Selected known comorbidities, language, education level, age, and sex were incorporated into the DAG based on their known associations with both exposure and outcomes47,48,49,50,51,52,53. These variables formed the basis of our adjustment set to minimize confounding bias in estimating the effect of vaccination on MACE and mortality.
Statistical analysis
Participant characteristics are presented as means with standard deviation (SD) for continuous variables and frequencies and percentages for categorical variables. We estimated the crude cumulative incidence (cIR) of death to assess the influence of age group and sex on outcomes following COVID-19 diagnosis, stratified by vaccination status. A competing risks model was employed to evaluate the cumulative incidence of MACE over 365 days54,55 (Supplementary Fig. 1).
As individuals were not randomly allocated to the different exposure groups, causal inferences from exposure to outcomes can be biased. Inverse probability of treatment weighting (IPTW) was used to adjust for confounding56. IPTW is a propensity score-based method, with the propensity scores reflecting their probability of belonging to the fully vaccinated group, which were calculated from logistic regression models that included the minimally sufficient set of covariates: calendar time, sex, age, education (primary, secondary, higher), native language (Estonian, Russian/Ukrainian, other), previous diabetes, cardiovascular disease and pulmonary disease (Supplementary, Fig. 2 for the DAG and Table 3 for the definitions of comorbidities). These covariates were derived from a directed acyclic graph (DAG) constructed in daggity.net57.
IPTW creates a pseudo-population in which the exposure is independent of measured confounders58. We used the IP weights in a marginal structural Poisson survival model, where the time from T0 and the vaccination status were the sole predictors, with an interaction term included in the spline model (see code in Supplement 2 for details).
Weighted standardized mean differences (SMDs) were calculated using the tidysmd package v. 0.2.0 and were used to assess the balance of covariates between groups59. SMDs ≤ 0.1 were regarded as sufficient balance60,61.
The occurrence of primary outcomes was assessed using incidence rates for 100 person-years (IR), and the treatment effects were evaluated using incidence rate ratios averaged over the entire one-year follow-up period (IRR). We employed a marginal structural model with IPT weights to estimate adjusted incidence rates (wIRs) per 100 person-years and incidence rate ratios (wIRRs, averaged over the one-year follow-up period) (Table S5). The model utilized Poisson regression with an unequally split timescale (0, 10, 30, 50, 75, 100, 150, 200, 300, 365 days) and modelled the follow-up time with penalized splines from the R::mgcv package, incorporating an interaction with vaccination status. The adjusted models were used to estimate IRs and IRRs, along with their corresponding 95% confidence intervals, which were calculated using the delta method from the marginal effects package62. The results were stratified by sex and age group (< 70 and ≥ 70 years). We then analyzed the results for individuals with severe and non-severe COVID-19 separately to control for the effect of disease severity on the primary outcomes. Missing data (education for 1003 individuals) were addressed through single imputation using the R::mice package63.
All statistical analyses were done in R v.4.3.264. The R code for reproducing the figures, including the modelling code, as well as datasets (slightly modified for privacy reasons) that can be used to run the code, can be found at https://datadoi.ee/handle/33/667 (DOI: https://doi.org/10.23673/re-505).
All research was conducted in accordance with relevant guidelines and regulations. The Research Ethics Committee of the University of Tartu approved the study on the 15th of March 2021 (No. 337/M-27). The need for informed consent was waived by the Ethics Committee of the University of Tartu due to the retrospective nature of the study.
Data sharing
According to Estonian legislative regulations and data protection law, the authors are prohibited from publicly releasing the raw data received from the health data registers in Estonia. However, data are available from the corresponding author ([email protected]) upon reasonable request and with permission of EHIF, ENHIS, The Causes of Death Registry, and Population Registry.