Antibody response analysis
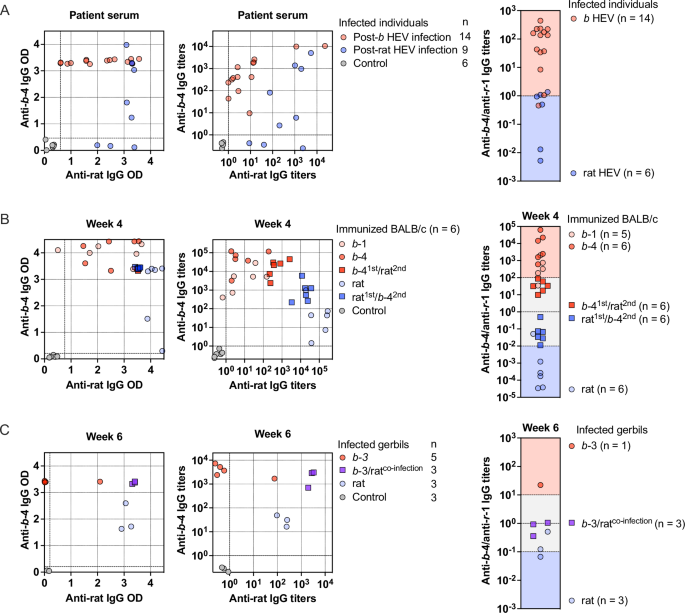
HEV 239 protein (p239) is a truncated form of the capsid protein that self-assembles into virus-like particles (VLPs) and mimics the conformation of natural HEV particles52. We first detected HEV IgG levels in rat and b HEV RNA-positive serum samples using recombinantly expressed ratti HEV genotype 1 (r-1; rat) p239 and b HEV genotype 4 (b-4) p239 coated in microplates. The clinical properties of rat and b HEV-infected patients are listed in Table S1. Based on the OD values of anti-HEV IgG for rat p239 and b-4 p239, we created a two-dimensional (2D) scatter plot. We observed that the OD values of most samples reached the upper plateau range, making it difficult to distinguish the true HEV IgG levels (Fig. 1A). We next used serially diluted samples to detect. Among rat HEV RNA-positive samples, 22.22% (2 of 9) of the samples showed significantly higher anti-rat HEV IgG levels than anti-b-4 HEV IgG, while 44.44% (four of nine) of the samples showed comparable anti-HEV IgG titer levels between rat p239 and b-4 p239. Anti-b-4 HEV IgG was undetectable in 33.33% (three of nine) of the samples (Fig. 1A). In b HEV RNA-positive samples, 78.6% (11 of 14) showed significantly higher anti-b-4 HEV IgG levels than anti-rat HEV IgG, and 21.4% (3 of 14) of the samples showed comparable anti-HEV IgG titer levels between rat p239 and b-4 p239 (Fig. 1A). A previous study reported an immunoassay system to differentiate rat HEV- and b HEV-infected patient samples based on the anti-HEV IgG ratio of 2.947 (ODb-4/ODrat)53. The immunoassay system was developed using individuals with active rat HEV infection and b HEV infection, which limits the utility of the anti-HEV IgG ratio of ODb-4/ODrat when applied to convalescent sera. Another limitation of this system is that it can be confusing when serum samples show comparable anti-HEV IgG levels between rat p239 and b-4 p239. Due to the high prevalence of b HEV in humans54, individuals are likely to have been exposed to both b HEV and rat HEV (dual exposure), exhibiting comparable anti-HEV IgG titer levels in serum samples. An overlapping range of b-4/rat values of 100 was found in samples from b and rat HEV RNA-positive patients (Fig. 1A), suggesting possible dual immunogen exposure. These results highlight that it is crucial to distinguish the antibody response elicited by either rat HEV, b HEV, or both HEV.
Two-dimensional scatter plot of HEV IgG levels in HEV RNA-positive samples (A), immunized BALB/c samples at week 4 (B), and infected gerbil samples at week 6 (C). HEV IgG levels were evaluated using serial dilutions of serum samples. Samples were simultaneously tested with rat p239 (blue dots) and b−4 p239 (red dots). The X-axis represents anti-rat HEV IgG titer levels; the Y-axis represents anti-b−4 HEV IgG titer levels. Healthy controls were individuals unexposed to either HEV. Each serum sample was tested in duplicate. The dashed line indicates the HEV IgG assay cutoff, defined as 2.5 times the mean reading of healthy control samples. Binding trends for anti-rat and anti-b−4 HEV IgG were evaluated in HEV RNA-positive samples (A), immunized BALB/c samples at week 4 (B), and infected gerbil samples at week 6 (C). The ratio values of anti-rat HEV IgG titer levels divided by anti-b−4 HEV IgG titer levels were calculated to assess HEV exposure characteristics in various situations. Initial exposure to b HEV (red) or rat HEV (blue) immunogens for BALB/c samples and gerbil samples are shown, with a single and dual exposures designated as circles and squares, respectively. Source data are provided as a Source Data file. b HEV species HEV balayani, b-1 species HEV balayani-1, b-3 species HEV balayani-3, b−4 species HEV balayani−4, HEV hepatitis E virus, rat HEV species HEV ratti-1.
To exclude unknown background interference, BALB/c mice were vaccinated with HEV p239 immunogen using multiple schemes (Fig. S1A). Vaccination of mice with either a single (b-1, b-4, rat) or dual (b-41st/rat2nd, rat1st/b-42nd) immunogen was designed to simulate naturally occurring HEV exposure. Anti-rat and anti-b-4 IgG titer levels in serum samples were detected weekly, with increased IgG titer levels (Fig. S1B). Overlapping clusters were observed in samples vaccinated with single and dual immunogens based on OD values (Fig. 1B). We next used serially diluted samples to detect. Samples formed separated clusters on the 2D scatter plot when classified according to the immunogen to which they were first exposed, with clear demarcation. However, samples from single and dual immunogen groups with the same initial exposure showed overlapping clusters, with no demarcation (Fig. 1B). We assessed the ratio (b-4/rat value) of anti-HEV IgG titer levels to further quantitatively visualize the characteristics of HEV exposure. A b-4/rat value of 100 was able to distinguish samples with different initial exposures (Fig. 1B). In the case of the same initial immunogen exposure, samples from a secondary heterologous immunogen exposure did not significantly increase in IgG titers to heterologous immunogens due to immune imprint. This resulted in overlapping clusters on the 2D scatter plot with similar ratio values (Fig. 1B). A b-4/rat value of higher than 102 or lower than 10−2 was able to distinguish samples from groups with a single (b-1, b-4) and dual (b-41st/rat2nd) immunogen exposure, as well as samples from groups with a single (rat) and dual (rat1st/b-42nd) immunogen exposure. However, samples with ratio values between 102 and 10−2 could not be distinguished (Fig. 1B).
Mongolian gerbils are highly susceptible to both b HEV and rat HEV infections55,56,57. The gerbil model can be used to mimic typical antibody response features in humans and exclude potential background interference in patients. Gerbils were challenged with HEV using multiple schemes (Fig. S2A). Infection of gerbils with either a single (b-3, rat) or dual (b-3/ratco-infection) HEV was designed to simulate naturally occurring HEV exposure. Anti-rat and anti-b-4 IgG titer levels in serum samples were detected weekly, with increased IgG titer levels (Fig. S2B). Separated clusters on the 2D scatter plot were observed in gerbil samples infected with single HEV and co-infected with dual HEV based on OD values and titer levels (Fig. 1C). We assessed the ratio (b-4/rat value) of anti-HEV IgG titer levels to further quantitatively visualize the characteristics of HEV exposure. A b-4/rat value of higher than 101 or lower than 10−1 was able to distinguish gerbil samples from groups with a single (b-3, rat) and dual (b-3/ratco-infection) HEV exposure (Fig. 1C). Considering the b-4/rat values generated in vaccinated BALB/c and infected gerbil samples, a robust b-4/rat value of higher than 102 or lower than 10−2 potentially identify single HEV exposure (b HEV or rat HEV). However, we sought to address this issue by developing a reliable and effective method to distinguish the antibody response elicited by rat HEV exposure.
DARE method establishment
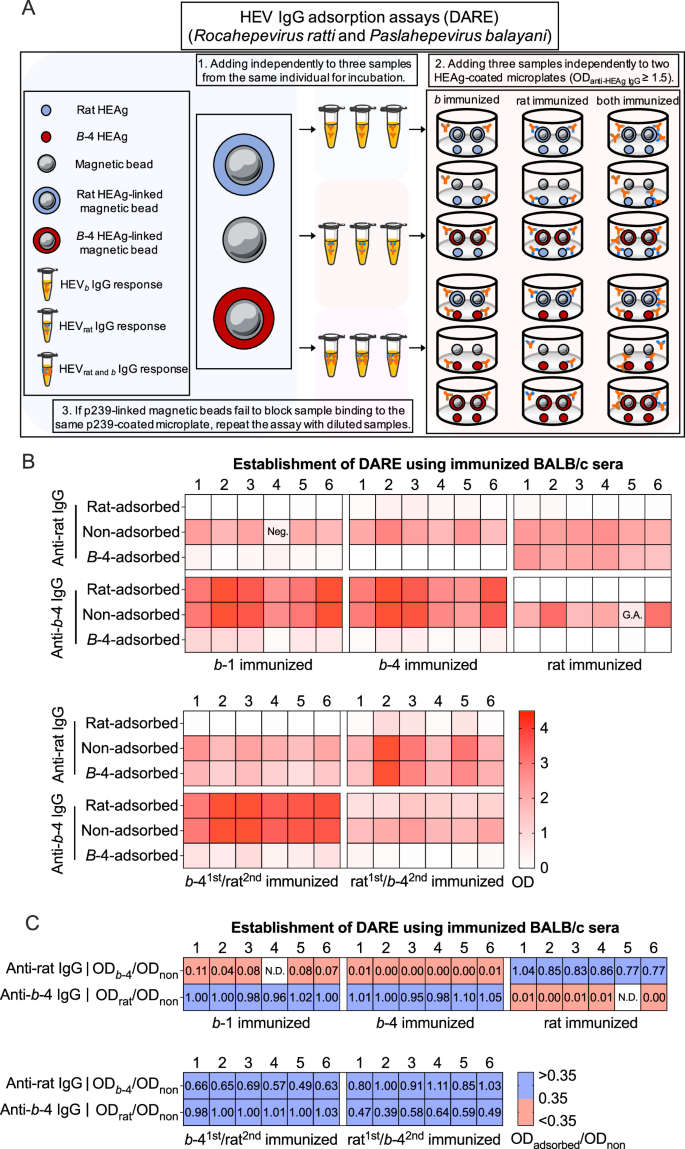
We developed a method called DARE (Fig. 2A). Samples from a single immunogen exposure were categorized as either b HEV-elicited IgG response (HEVb IgG response) or rat HEV-elicited IgG response (HEVrat IgG response). Samples from dual immunogen exposure were classified as both rat and b HEV-elicited IgG response (HEVrat and b IgG response). Three samples from the same individual were separately incubated with two p239-linked magnetic beads and unlinked magnetic beads. After the adsorption process was completed, the three samples from the same individual were independently added to two p239-coated microplates. If the individual had an HEVrat IgG response, their sera might contain IgG that specifically recognized rat p239 while showing undetectable or cross-reactive IgG against b-4 p239. It was expected that rat p239-linked magnetic beads would completely absorb the IgG from the HEVrat response, resulting in almost complete blocking of binding to the two p239-coated microplates. On the other hand, b-4 p239-linked magnetic beads would only adsorb a small portion of cross-reactive IgG from the HEVrat response, leading to partial blocking of binding to rat p239-coated microplates and almost complete blocking of binding to b-4 p239-coated microplates. If the individual had a HEVb IgG response, the situation would be reversed. If the individual had a HEVrat and b IgG response, their sera might contain IgG that specifically recognized rat p239 and b-4 p239, while also showing undetectable or cross-reactive IgG. In this case, rat p239-linked magnetic beads would adsorb cross-reactive IgG and IgG that specifically recognized rat p239, resulting in almost complete blocking of binding to rat p239-coated microplates and partial blocking of binding to b-4 p239-coated microplates. On the other hand, b-4 p239-linked magnetic beads would adsorb cross-reactive IgG and IgG that specifically recognizes b-4 p239, leading to partial blocking of binding to rat p239-coated microplates and almost complete blocking of binding to b-4 p239-coated microplates.
A Establishment of the DARE method. The overall scheme is shown. Before detecting anti-rat and anti-b−4 HEV IgG simultaneously, three samples from the same individual were separately incubated with two p239-linked magnetic beads and unlinked magnetic beads. After the adsorption process, the three samples from the same individual were independently added to two p239-coated microplates. B Adsorption of serum samples from immunized BALB/c at week 4. Samples were separately incubated with rat p239- and b−4 p239-linked magnetic beads, then simultaneously tested in rat p239 and b-4 p239. Samples adsorbed to unlinked magnetic beads served as controls. C Algorithm setup. The ODadsorbed/ODnon ratio values, representing the OD values after adsorption with two p239-linked magnetic beads (ODadsorbed) divided by the OD values after adsorption with one unlinked magnetic beads (ODnon), were calculated to analyze the exposure situation. An ODadsorbed/ODnon ratio value of 0.35 was used to identify the IgG response, effectively distinguishing between single and dual immunogen/HEV recent exposures. Blue boxes represent samples incubated with p239-linked magnetic beads, where an ODadsorbed/ODnon ratio value is greater than 0.35 (insufficient adsorption). Red boxes represent samples incubated with p239-linked magnetic beads, where an ODadsorbed/ODnon ratio value is less than or equal to 0.35 (sufficient adsorption). Source data are provided as a Source Data file. b HEV species HEV balayani, b−1 species HEV balayani-1, b−4 species HEV balayani−4, DARE distinguishing antibody response elicitation, GA gray area, HEAg hepatitis E virus antigen, HEV hepatitis E virus, Neg. negative, rat HEV species HEV ratti-1.
Based on the above principles, we used mouse samples for identifying IgG response using DARE. As expected, our DARE method preliminarily distinguished mouse samples with a single or dual immunogen exposure based on the changes in IgG (Fig. 2B). Mouse samples with a single and dual immunogen exposure, were further analyzed to assess the characteristics of HEV exposure by evaluating the b-4/rat ratio values. The ODadsorbed/ODnon ratio values, representing the OD values after adsorption with two p239-linked magnetic beads (ODadsorbed) divided by the OD values after adsorption with one unlinked magnetic beads (ODnon), were calculated to analyze the exposure situation. An ODadsorbed/ODnon ratio value of 0.35 was used to identify the IgG response, effectively distinguishing between a single and dual immunogen/HEV recent exposures in samples (Fig. 2C). These results demonstrated that our DARE method can effectively distinguish individuals with a single or dual HEV exposure.
DARE method validation
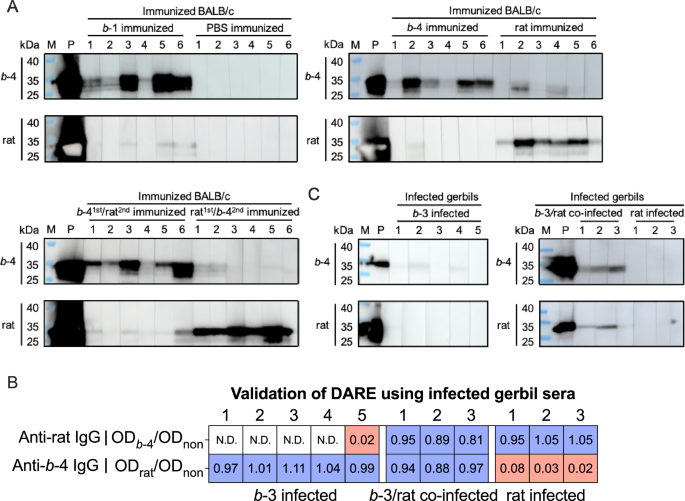
Given the unknown accuracy of the established DARE method, the previously reported immunoblot method was applied to validate in immunized BALB/c samples44,53. None (0 of 6) of the samples from PBS immunized BALB/c exhibited a visible band at parallel b-4 p239 and rat p239 immunoblots (Fig. 3A), demonstrating the specificity of the DARE method. Among samples from b-1 and b-4 immunized BALB/c, 50% (three of six) and 83.33% (five of six), respectively, reacted exclusively with b-4 p239 immunoblot and did not cross-react with rat p239 immunoblot (Fig. 3A). Additionally, 66.67% (four of six) of samples from rat immunized BALB/c reacted exclusively with rat p239 immunoblot and did not cross-react with b-4 p239 immunoblot (Fig. 3A). Since the immunoblot method was developed without considering the possibility of dual exposure, we hypothesize that positive visible bands on both the parallel b-4 p239 and rat p239 immunoblots indicate HEVrat and b IgG response. It is important to note that this hypothesis may incorrectly classify BALB/c samples immunized with a single antigen as dual immunized. Among samples from b-41st/rat2nd and rat1st/b-42nd immunized BALB/c, 66.67% (four of six) and 50% (three of six) of sera cross-reacted with b-4 p239 and rat p239 immunoblots (Fig. 3A).
A Immunoblot using immunized BALB/c samples at week 4. Immunoblot was used to validate the accuracy of the DARE method established with immunized BALB/c samlpes at week 4. Parallel b−4 p239 and rat p239 immunoblots were conducted using immunized BALB/c samples at week 4 to identify HEV antibody response. M represents the lane labeled with the marker. P indicates the positive control, representing the lane labeled with an antibody that cross-reacts with both b HEV and rat HEV. Two times each experiment was repeated independently with similar results. B Algorithm validation using infected gerbil samples at week 6. An ODadsorbed/ODnon ratio value of 0.35 was used in the algorithm setup to identify the IgG response, effectively distinguishing immunized BALB/c samples at week 4 between single and dual immunogen/HEV recent exposures. Infected gerbil samples at week 6 were used to validate the algorithm setup with an ODadsorbed/ODnon ratio value of 0.35. Blue boxes represent samples incubated with p239-linked magnetic beads, where an ODadsorbed/ODnon ratio value is greater than 0.35 (insufficient adsorption). Red boxes represent samples incubated with p239-linked magnetic beads, where an ODadsorbed/ODnon ratio value is less than or equal to 0.35 (sufficient adsorption). C Immunoblot using infected gerbil samples at week 6. An immunoblot was used to validate the accuracy of the DARE method established with immunized BALB/c at week 4. Parallel b−4 p239 and rat p239 immunoblots were conducted using infected gerbil samples at week 6 to identify HEV antibody response. M represents the lane labeled with the marker. P indicates the positive control, representing the lane labeled with an antibody that cross-reacts with both b HEV and rat HEV. Two times each experiment was repeated independently with similar results. Source data are provided as a Source Data file. b HEV species HEV balayani, b-1 species HEV balayani-1; b-3 species HEV balayani-3, b−4 species HEV balayani−4, DARE distinguishing antibody response elicitation, HEV hepatitis E virus, rat HEV species HEV ratti-1.
Since BALB/c samples from week 4 post-immunization were still in the phase of a steady increase in antibody response, which could limit the credibility of the DARE method established by the immunization model. Serum samples from immunized BALB/c at week 6, with steady antibody response levels, were validated in parallel using the DARE method and the immunoblot method. An ODadsorbed/ODnon ratio value of 0.35 established using immunized BALB/c samples at week 4 was used in the algorithm to identify the IgG response, effectively distinguishing between single and dual immunogen/HEV recent exposures in immunized BALB/c samples at week 6 (Fig. S3A). None (0 of 6) of the BALB/c samples at week 6 exhibited a visible band at parallel b-4 p239 and rat p239 immunoblots (Fig. S3B). Among samples from week 6 of b-1 and b-4 immunized BALB/c, 16.67% (1 of 6) and 100% (6 of 6), respectively, reacted exclusively with b-4 p239 immunoblot and did not cross-react with rat p239 immunoblot (Fig. S3B). Additionally, 33.33% (2 of 6) of samples from week 6 of rat immunized BALB/c reacted exclusively with rat p239 immunoblot and did not cross-react with b-4 p239 immunoblot (Fig. S3B). Among samples from week 6 of b-41st/rat2nd and rat1st/b-42nd immunized BALB/c, 100% (6 of 6) and 100% (6 of 6) of sera cross-reacted with b-4 p239 and rat p239 immunoblots (Fig. S3B).
The gerbil infection model can be used to mimic typical antibody response features in humans and was also validated in parallel using the DARE method and the immunoblot method. An ODadsorbed/ODnon ratio value of 0.35 established using immunized BALB/c samples at week 4 was used in the algorithm to identify the IgG response, effectively distinguishing between single and dual immunogen/HEV recent exposures in infected gerbil samples (Fig. 3B). Among samples from b-3 infected gerbil, 40% (2 of 5) of sera reacted exclusively with b-4 p239 immunoblot and did not cross-react with rat p239 immunoblot (Fig. 3C). Additionally, 33.33% (1 of 3) of samples from rat infected gerbil reacted exclusively with rat p239 immunoblot and did not cross-react with b-4 p239 immunoblot (Fig. 3C). Among samples from b-3/ratco-infection infected gerbil, 100% (3 of 3) of sera cross-reacted with b-4 p239 and rat p239 immunoblots (Fig. 3C). These results demonstrate that the DARE method has higher sensitivity and comparable specificity to the immunoblot method.
The algorithm of the DARE method
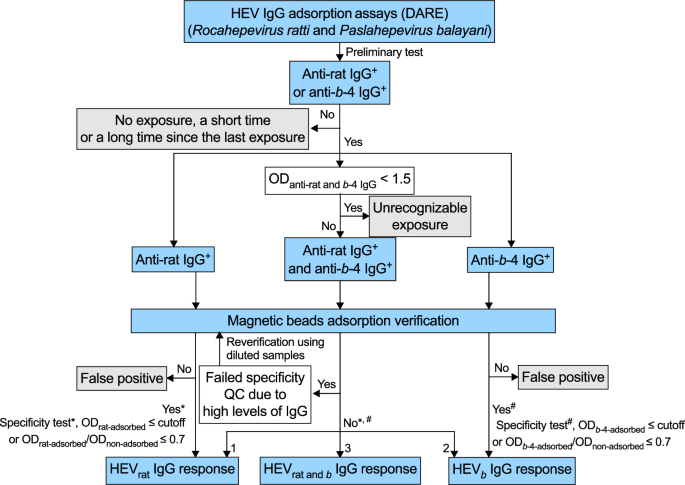
To further facilitate the reliability and convenience of the DARE method for large populations, a set of optimized workflows and algorithms is proposed based on rigorous experimental data (Fig. 4). Samples from large populations were preliminarily tested for anti-rat and anti-b-4 HEV IgG. Samples negative for both anti-rat and anti-b-4 HEV IgG were considered as having either no exposure or having had a short time or a long time since the last exposure. If a sample is only anti-rat IgG positive or only anti-b-4 IgG positive, it is designated as HEVrat IgG response (indicating rat HEV exposure within the recent 5 years) or HEVb IgG response (indicating b HEV exposure within the recent 5 years), respectively. Further specificity test (*, #) is required to determine the specificity of binding. An ODadsorbed value (using the same p239-linked magnetic beads as the p239-coated microplate) less than or equal to the cutoff, or an ODadsorbed/ODnon ratio value less than or equal to 0.7 (twice the value of distinguishing algorithm) is used for identification. For samples positive for both anti-rat and anti-b-4 HEV IgG, weakly positive samples can interfere with the identification of the IgG response. This is a limitation of our DARE method, so we defined samples with both anti-rat and anti-b-4 HEV IgG OD values less than 1.5 (cutoff/0.35) as unrecognizable exposure. Samples with both anti-rat and anti-b-4 HEV IgG OD values higher than 1.5 could be identified as HEVrat IgG response, HEVrat and b IgG response (indicating both rat and b HEV exposure within the recent 5 year) or HEVb IgG response based on the specificity test and the algorithm (label 1, 2, and 3). The remaining samples (ODanti-rat IgG ≥ 1.5 and ODanti-b-4 IgG < 1.5, ODanti-rat IgG < 1.5 and ODanti-b-4 IgG ≥ 1.5) that were positive for both anti-rat and anti-b-4 HEV IgG could be identified as HEVrat IgG response or HEVb IgG response based on the specificity test and the algorithm for IgG OD values higher than 1.5. In addition, for these remaining samples, a b-4/rat value of anti-HEV IgG titer levels higher than 102 or less than 10−2 can be directly designated as HEVb IgG response or HEVrat IgG response. For samples with high HEV IgG levels, the ODadsorbed value (using the same p239-linked magnetic beads as the p239-coated microplate) must meet the specificity test. If it does not, the DARE method needs to be repeated with diluted serum samples for accurate HEV IgG response identification.
The algorithm was based on rigorous experimental data. To further facilitate the reliability and the convenience of the DARE method in large populations, an optimized workflow and algorithm are proposed. In the figure, the labels 1, 2, and 3 indicate the algorithms applied when ODanti-rat and b−4 IgG ≥ 1.5. Label 1 corresponds to the condition where ODb-4-adsrobed/ODnon-adsorbed > 0.35 (anti-rat IgG) and ODrat-adsrobed/ODnon-adsorbed ≤ 0.35 (anti-b-4 IgG), which is designated as HEVrat IgG response. Label 2 corresponds to the condition where ODb-4-adsrobed/ODnon-adsorbed ≤ 0.35 (anti-rat IgG) and ODrat-adsrobed/ODnon-adsorbed > 0.35 (anti-b-4 IgG), which is designated as HEVb IgG response. Label 3 represents all cases except those of labels 1 and 2, which is designated as HEVrat and b IgG response. For the cases where ODanti-rat IgG ≥ 1.5 and ODanti-b-4 IgG < 1.5, different algorithms are applied. Label 1 corresponds to the condition where ODb-4-adsrobed/ODnon-adsorbed > 0.35 (anti-rat IgG) or anti-rat/anti-b-4 IgG titers > 102, which is designated as HEVrat IgG response. All other conditions are designated as unrecognizable exposure. For the cases where ODanti-rat IgG < 1.5 and ODanti-b-4 IgG ≥ 1.5, different algorithms are applied. Label 1 corresponds to the condition where ODrat-adsrobed/ODnon-adsorbed > 0.35 (anti-b-4 IgG) or anti-b-4/anti-rat IgG titers >102, which is designated as HEVb IgG response. All other conditions are designated as unrecognizable exposure. b HEV species HEV balayani, b-4 species HEV balayani-4, DARE distinguishing antibody response elicitation, HEV hepatitis E virus, OD optical density, QC quality control, rat HEV species HEV ratti−1.
Burden of rat HEV spillover
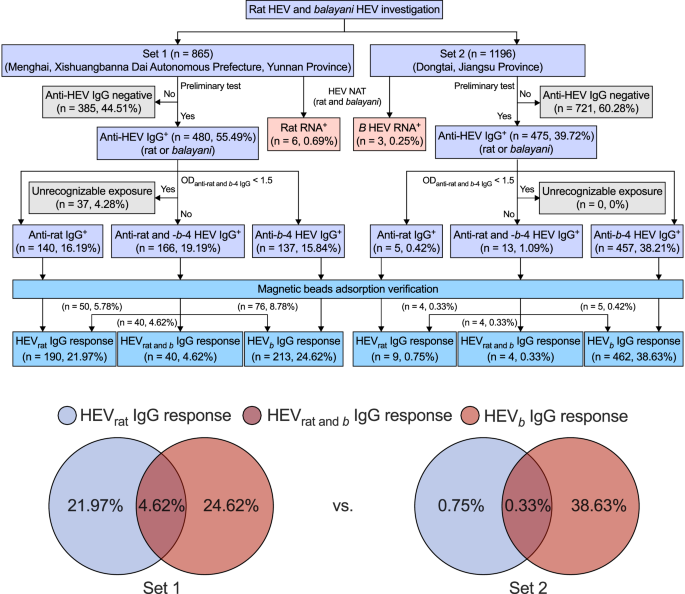
To gauge the exposure risk of rat HEV, population samples were collected from two sets: set 1 from Menghai, a city on China’s southwestern border in Yunnan province, and set 2 from Dongtai, a city on China’s eastern coast in Jiangsu province.
In set 1, preliminary anti-HEV IgG testing showed that 55.49% (480 of 865) of individuals were positive for anti-rat or anti-b-4 HEV IgG (Fig. 5). We identified samples with both anti-rat and anti-b-4 HEV IgG OD values less than 1.5 as unrecognizable exposure, accounting for 4.28% (37 of 865) of set 1. Using the DARE method, we analyzed individuals who were positive for only anti-rat IgG, only anti-b-4 IgG, and those who were positive for both anti-rat and anti-b-4 IgG based on the specificity test and the algorithm (Fig. S4A–J). In set 1, HEV exposures were comprehensively documented, with 21.97% (190 of 865) showing rat HEV exposure, 5.43% (47 of 865) showing both rat and b HEV exposure and 23.82% (206 of 865) showing b HEV exposure (Fig. 5). To confirm the accuracy of the DARE method in human population samples, we applied the previously developed immunoblot method to five types of samples distinguished by the DARE method44,53. These serum samples were randomly selected. None (0 of 41) of sera from individuals with anti-HEV IgG negative exhibited visible band at parallel b-4 p239 and rat p239 immunoblots (Fig. S5), demonstrating specificity of the DARE method. Additionally, none (0 of 36) of the sera from individuals with unrecognizable exposure showed a visible band (Fig. S6), further highlighting the higher sensitivity of the DARE method. Among sera from individuals with HEVrat IgG response, 24.44% (11 of 45) of sera only reacted with rat p239 immunoblot and did not cross-react with b-4 p239 immunoblot (Fig. S7). Among sera from individuals with HEVb IgG response, 4.44% (2 of 45) of sera only reacted with b-4 p239 immunoblot and did not cross-react with rat p239 immunoblot (Fig. S8). Among sera from individuals with HEVrat and b IgG response, 5.56% (2 of 36) of sera cross-reacted with b-4 p239 and rat p239 immunoblots (Fig. S9). These results support that the DARE method has higher sensitivity and comparable specificity to the immunoblot method.
The summary of HEV spillover risk and infection in the two sets is displayed on the left and right, respectively. Purple boxes represent the results of a preliminary test using anti-HEV IgG assays. Gray boxes represent identification of anti-HEV IgG negative individuals and unrecognizable exposure. Blue boxes represent identification of HEVrat, HEVrat and b, and HEVb IgG responses using the DARE method. Additionally, red boxes represent results of HEV nucleic acid testing. Source data are provided as a Source Data file. b HEV species HEV balayani, b−4 species HEV balayani−4, HEV hepatitis E virus, NAT nucleic acid testing, rat HEV species HEV ratti−1.
In set 2, preliminary anti-HEV IgG testing showed that 39.72% (475 of 1196) of individuals were positive for anti-rat or anti-b-4 HEV IgG (Fig. 5). No unrecognizable exposure was identified. The specificity test and algorithm revealed that 0.75% (9 of 1196) had rat HEV exposure, 0.33% (4 of 1196) had both rat and b HEV exposure, and 38.63% (462 of 1196) had b HEV exposure (Fig. 5 and Fig. S10A–G). The profiles of HEV exposure differed significantly between set 1 and set 2, indicating that rat HEV is endemic in humans and spreads regionally. The spillover burden posed by rat HEV is notably higher in the hotspot.
We further analyzed the demographic characteristics of the HEV IgG response in the two sets. Compared to anti-HEV IgG negative individuals, we found that individuals with HEVrat IgG response, HEVb IgG response, or both HEVrat and b IgG response generally had an older median age, except for those with HEVrat IgG response in set 2 (Table 1). Cochran-Armitage trend test revealed significant differences in age distribution. In the two sets, the proportion of individuals with HEVrat and b IgG response and HEVb IgG response increased with age, demonstrating higher HEV and b HEV exposure with age. In set 1, the subgroup aged 20–29 with HEVrat IgG response had a higher composition ratio compared to those with HEVb IgG response or HEVrat and b IgG response. Conversely, the subgroup aged 60 and above showed a lower composition ratio than the other two subgroups. This suggests that, unlike b HEV whose prevalence increases with age, the prevalence of rat HEV may be due to specific exposure patterns among younger individuals. We also conducted a retrospective analysis of laboratory test indicators over the past five years for set 1 (Table 2). Individuals with indicators at two or more time points (more than two months apart) were included, accounting for 13.3% (115 of 865) of set 1. The proportion of individuals with an IgG response or anti-HEV IgG negative roughly matches that of set 1. During the retrospective period, abnormal ALT levels were observed in 39.5% (15 of 38) of individuals with rat HEV exposure, 60.0% (6 of 10) of those with both rat and b HEV exposure, 63.0% (17 of 27) of those with b HEV exposure and 22.5% (9/40) of those with anti-HEV IgG negative. The relative risks (RRs) were 1.76, 2.67, and 2.80, respectively. This indicates that rat HEV infection in humans increases the hepatitis burden.
Spillover infection of rat HEV to humans
In set 1, 0.69% (6 of 865) of individuals had detectable rat HEV RNA (Fig. 5). Among these six individuals, 33.33% (2 of 6) had HEVrat IgG response, 50% (2 of 6) had HEVrat and b IgG response, 16.67% (1 of 6) had unrecognizable exposure, and 16.67% (1 of 6) were anti-HEV IgG negative. In set 2, 0.25% (3 of 1196) of individuals had detectable b HEV RNA. Among these three individuals, 33.33% (1 of 3) had an HEVb IgG response, and 66.67% (2 of 3) were anti-HEV IgG negative. These findings indicate the infection risk posed by rat HEV is significantly higher in the hotspot.
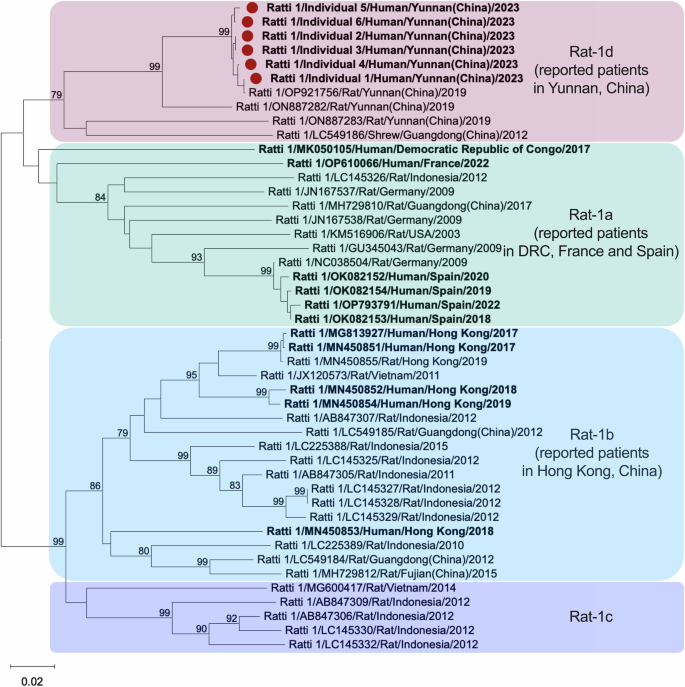
We amplified a portion of the ORF1 genome using broad-spectrum nested RT-PCR assays targeting the rat HEV RdRp gene. Phylogenetic analysis showed that the rat HEV strains formed four distinct subclades, labeled rat-1a to rat-1d. Human-derived rat HEV strains are isolated into subclades rat-1a (the Democratic Republic of Congo, France, and Spain) and rat-1b (Hong Kong, China). In our study, the human-derived strains were classified into subclade rat-1d (GenBank nos. PQ001569-PQ001574) (Fig. 6). By BLAST analysis, these strains showed 99.35–100% nucleotide identity with rat HEV strains (GenBank nos. OP921755 and OP921756) isolated from rats in Yunnan province collected in 201958. These results suggest zoonotic potential in subclade rat-1d, with rat HEV infections closely related to local rats. Three rat HEV subclades with zoonotic crossover to humans have been discovered worldwide.
Phylogenetic tree was reconstructed by the neighbor-joining method based on a partial RdRp gene of the rat HEV ORF1. Viral species and genotype, GenBank accession number, virus host, country (province or state) of origin, and year of collection are indicated. Sequences obtained in rat HEV-infected individuals from set 1 are highlighted with a red circle (GenBank nos. PQ001569-PQ001574). HEV strains derived from human-infected are marked in bold. Bootstrap values >70 are shown. Source data are provided as a Source Data file. HEV hepatitis E virus, ORF1 the open reading frame 1, rat HEV species HEV ratti−1, RdRp RNA-dependent RNA polymerase.
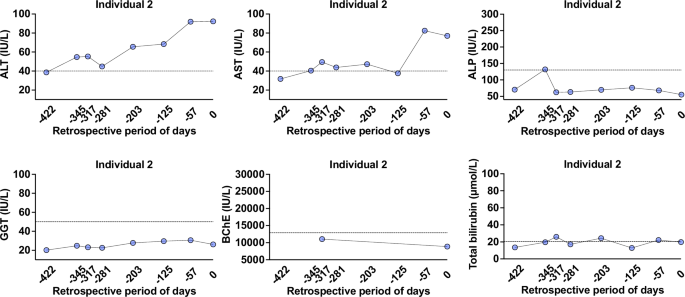
The median age of six rat HEV RNA-positive individuals was 47.0 years (IQR 27.0–59.0) and 66.67% (four of six) were female (Table 3). None had HBV and immunosuppression, and none presented with hepatitis. Three out of four women were pregnant, and two men were admitted for pulmonary infection and acute tonsillitis. Of the six individuals, one (individual 2) had a chronic medical condition (type 2 diabetes mellitus) and abnormal ALT levels (Table 3). According to retrospective analysis of laboratory test indicators, individual 2 showed abnormal ALT levels in the past 203 days (day 0, rat HEV RNA positive) (Fig. 7). This suggests that potential liver damage appears not to be caused by rat HEV infection. Individual 2 cleared rat HEV RNA and did not progress to chronic infection. None of the individuals had abnormal renal markers (Table 3 and Fig. S11). These results indicate that these rat HEV RNA-positive individuals are considered asymptomatic carriers with concomitant disease.
Liver markers in individual 2 with positive rat HEV RNA were traced. Liver markers include ALT, AST, ALP, GGT, BChE, and total bilirubin. Data points are marked as blue circles. Source data are provided as a Source Data file. ALP alkaline phosphatase, ALT alanine aminotransferase, AST aspartate aminotransferase, BChE butyrylcholinesterase, GGT gamma-glutamyl transferase, rat HEV species HEV ratti−1.
Public health burden of rat HEV
Population samples with ALT abnormality were collected from two sets: set 3 from Dali, a city on China’s southwestern border in Yunnan province, and set 4 from Dongtai, a city on China’s eastern coast in Jiangsu province. Demographic characteristics for the populations in these two sets are summarized in Table S2.
In set 3, 13.97% (70 of 501) showed rat HEV exposure, 4.99% (25 of 501) showed both rat and b HEV exposure, 41.92% (210 of 501) showed b HEV exposure and 1.60 (8 of 501) showed unrecognizable exposure (Table S3 and Fig. S12A–J). This exposure spectrum was similar to that of set 1. Testing for rat and b HEV RNA revealed that 0.4% (2 of 501) of patients had detectable b HEV RNA. In set 4, 1.91% (15 of 786) of individuals had detectable b HEV RNA, demonstrating the high prevalence of b HEV in this region (Table S4). We amplified a portion of the ORF1 genome from b HEV RNA-positive patients in set 4 and phylogenetic analysis showed that these b HEV strains belonged to subtype 4d (Fig. S13).