Patients
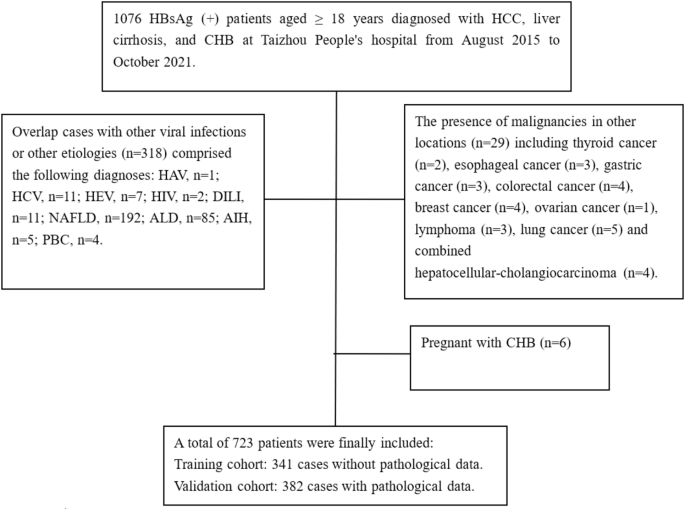
A total of 358 patients with HBV-HCC, 124 patients with chronic hepatitis B (CHB), and 241 patients with hepatitis B cirrhosis were recruited from August 2015 to October 2021 at Taizhou People’s Hospital. Among them, 382 patients had liver pathological data. All patients signed the informed consent for liver puncture. The inclusion criteria were as follows: (1) Older than 18 years old; (2) Complete clinical data; (3) The diagnosis of CHB and hepatitis B cirrhosis adhered strictly to the Chinese “Guidelines for the Prevention and Treatment of Chronic Hepatitis B (version 2022)”17, and the diagnosis of HCC was in accordance with the “Guidelines for the Diagnosis and Treatment of Primary Liver Cancer (2022 Edition)”18. The exclusion criteria were as follows: (1) Overlapping with other viral infections or liver diseases caused by other factors, such as HCV infection, human immunodeficiency virus infection, drug-induced liver injury, nonalcoholic fatty liver disease, alcoholic fatty liver disease, and autoimmune hepatitis; (2) The presence of malignancies in other primary locations, including secondary liver cancer; (3) Pregnant and lactating women. None of the patients consumed vitamin K or vitamin K antagonists. The patient screening process is shown in Fig 1.
At baseline, 403 out of 723 enrolled patients received antiviral treatment. Among those who received antiviral treatment, 48 received interferon treatment, while the remaining patients were treated with nucleoside analogs or a combination of antiviral therapies. Patients who had not received antiviral treatment at baseline were given antiviral treatment after completing relevant examinations and meeting the criteria for antiviral therapy.
This study was approved by the ethics committee of Taizhou People’s Hospital (ky2021-081-01). This study was a retrospective study, the data was the routine examinations of patients, and the patient’s privacy was not involved, so informed consent was exempted.
Data collection
Clinical data included sex, age, laboratory tests (routine blood tests, biochemical detection, tumor marker detection, coagulation function, pathogen detection, HBV marker detection, and HBV DNA quantification), imaging examinations (dynamic contrast-enhanced computed tomography (CT) or magnetic resonance imaging (MRI)), and liver histopathology.
Examination of the specimen: A blood cell analyzer was used for complete blood count, and an Abbott i2000SR automated chemiluminescent immunoassay analyzer was used to detect AFP and PIVKA-II, and GP73 was measured using the up-converting phosphor technology immunoassay analyzer UPT-3 A produced by Beijing Hotgen Biotech Co., Ltd. along with the corresponding quantitative detection kit. The reference concentrations of AFP, PIVKA-II, and GP73 were 0-8.78 ng/ml, 0–32 mAU/ml, and 0-150 ng/ml, respectively. Liver function and kidney function were tested by an Olympus 5421 automatic biochemical analyzer. HBV markers were detected by a Roche Elecsys electrochemiluminescence automatic immune analyzer. HBV DNA was quantified by a Roche Cobas TaqMan 48 virus quantification system, and the lower limit of quantification was 1.3 lg IU/ml (20 IU/ml).
Pathological detection
The pathological data were obtained from multiple sources. First, they involve puncture biopsies of liver nodules which cannot rule out the presence of HCC and do not exhibit the characteristic imaging features of HCC, with the aim of obtaining a definitive pathological diagnosis. Second, pathological data of resected liver cancer specimens were collected. Third, liver biopsy evaluations were conducted to assess inflammation and fibrosis in HBV-infected patients, as well as in those with liver disease who were serum HBsAg-negative and HBcAb-positive. Liver biopsy was performed under the guidance of ultrasound, utilizing a TSK 16G liver biopsy needle to procure liver nodule(s). Informed consent and signatures were obtained from the patients before the procedure. The seven-point baseline sampling method was used for the resected specimens. The pathological specimens were evaluated by an intermediate and a senior pathologist, initially examined by the intermediate pathologist and subsequently reviewed by the senior pathologist. In the event of any queries, the researcher and pathologist jointly deliberated and reached a consensus based on the criteria.
Grouping
The training cohort for screening of indicators
A total of 341 patients with clinical diagnoses and lacking pathological data were selected as the training cohort. These patients were divided into the CHB group (56 patients with HCC and 74 without), the Child‒Pugh A hepatitis B cirrhosis group (43 patients with HCC and 39 without), and the Child‒Pugh B&C hepatitis B cirrhosis group (88 patients with HCC and 41 without). The baseline data was shown in Table 1.
The validation cohort for validation of the screened indicators
The validation cohort consisted of 382 patients with liver pathology data, including 171 HCC patients. The median age was 56 (48, 64). The number of males and female was 307 and 75, respectively. Among the 171 HCC patients, 124 underwent surgical resection. These patients were categorized into the CHB group (55 patients with HCC and 50 without), the Child‒Pugh A hepatitis B cirrhosis group (56 patients with HCC and 91 without), and the Child‒Pugh B&C hepatitis B cirrhosis group (60 patients with HCC and 70 without).
The staging of HCC
This study adopted the China liver cancer (CNLC) staging system. The CNLC staging system is divided into stages Ia, Ib, IIa, IIb, IIIa, IIIb, and IV. The recommended treatment for patients with CNLC stages Ia, Ib, and IIa HCC and enough liver function reserve is surgical resection18. Among the 358 HCC patients in the study, there were 191 patients with CNLC stages Ia or Ib, 51 patients with CNLC stage IIa, 38 with CNLC stage IIb, 41 with CNLC stage IIIa, 18 with CNLC stage IIIb, and 19 with CNLC stage IV. Approximately 70% of patients belong to the early to middle stage of HCC.
Statistical analysis
SPSS 26.0 statistical software was used for data analysis and processing, and P < 0.05 indicated that the results were significantly different. The chi-square test was used to analyze the level of concordance between patients with and without HCC in each group, and the Kolmogorov‒Smirnov test was used to assess the normality of the measurement data. The measurement data in this study exhibited a non‒normal distribution; thus, the median and quartiles were used to describe the non‒normally distributed data. The constituent ratio was used to describe the count variable, and statistical charts were drawn. The Mann‒Whitney U test was used to compare the measurement data of patients with and without HCC within each group. The Pearson chi-square test was used to analyze the differences in classification data across groups. After identifying indicators with statistically significant results from the univariate analysis, a collinearity analysis was carried out and combined with references to exclude the mutual influence of individual variables. These refined variables were subsequently included in a multivariate analysis. Binary logistic regression was used to analyze the relationships between the levels of pertinent indicators and clinical variables in patients with HCC. The receiver operating characteristic (ROC) curve was generated for diagnostic combinations of multiple indicators, and the area under the curve (AUC) was calculated. Using liver pathology as the gold standard, the consistency between the screened indicators and liver pathology for HCC diagnosis was analyzed with Cohen’s kappa.