γδ T cells affect early expansion of CD8+ and CD4+ T cells
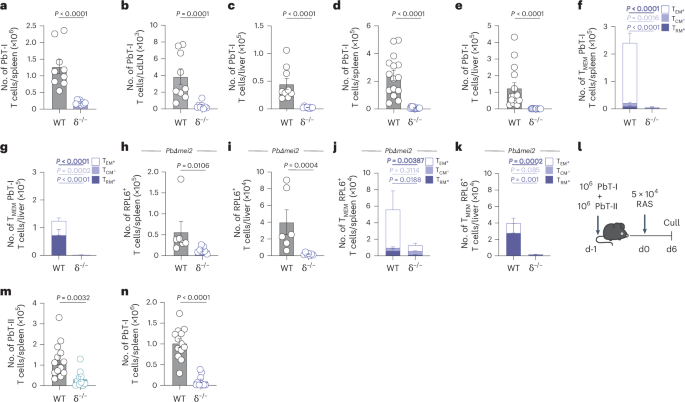
Intravenous RAS injection initiates priming and expansion of T cells in the spleen, followed by recirculation and resultant accumulation of activated T cells in the liver14, a proportion of which will then differentiate into memory subsets, including protective liver TRM cells15. To understand if the previously described role for γδ T cells in memory T cell responses in RAS-vaccinated mice16 was due to a role for γδ T cells in T cell priming or the generation of memory T cells, we used PbT-I TCR transgenic T cells to study the response at an antigen-specific level. These T cells recognize an H-2Kb-restricted epitope (PbRPL6120-127) from the P. berghei-derived RPL6 protein14,29. PbT-I cells were adoptively transferred into B6 (wild-type (WT)) or TCRδ−/− (δ−/−) mice and responses assessed 6 days after vaccination with RAS (Fig. 1a–c and Extended Data Fig. 1a). This revealed a defect in the accumulation of PbT-I cells in the spleen (Fig. 1a), the liver-draining lymph node (Fig. 1b) and, consequently, the liver (Fig. 1c) of δ−/− mice. As a result of this early failure to respond to RAS, fewer memory PbT-I cells were found in the spleen (Fig. 1d) and liver (Fig. 1e) after 3 weeks, with impaired formation of memory T cells subsets, including liver TRM cells (Fig. 1f,g).
A total of 106 RPL6-specific transgenic CD8+ T cells (PbT-I) were transferred into mice 1 day before vaccination with 5 × 104 RAS. PbT-I cells were analyzed either 6 or 23 days later. a–c, Enumeration of PbT-I cells in the spleen (a), liver-draining lymph nodes (LdLN) (b) or liver (c) of WT (B6) (n = 9), or Tcrd−/− (δ−/−) (n = 8) mice 6 days after immunization. d,e, Numbers of PbT-I cells in the spleen (d) or liver (e) of WT (n = 15), or δ−/− (n = 14) mice 23 days after vaccination. f,g, Quantified memory T cell (TMEM) subsets; central memory (TCM; CD62L+, CD69−), effector memory (TEM; CD62L−, CD69−) and resident memory (TRM; CD62L−, CD69+) within the CD44+ PbT-I cell compartment of WT (n = 15) or δ−/− (n = 14) spleens (f) or livers (g) at day 23. Mice were vaccinated with 2 × 104 PbΔmei2 sporozoites; 35 days later, mice were culled. h,i, Numbers of RPL6 tetramer+ cells in the spleen (h) and liver (i) of WT (n = 6) or δ−/− (n = 7) mice 35 days after vaccination. j,k, Quantified TMEM subsets; TCM (CD62L+, CD69−), TEM (CD62L−, CD69−) and TRM (CD62L−, CD69+) within the CD44+ RPL6+ T cell compartment of WT (n = 6) or δ−/− (n = 7) spleens (j) or livers (k) at day 35. l, Experimental design. m,n, Transgenic CD8+ (PbT-I) and CD4+ (PbT-II) T cells were transferred into mice 1 day before vaccination with RAS. PbT-II (m) or PbT-I (n) cell counts in the spleen at day 6 (WT, n = 13; δ−/−, n = 10). Data show two (a,c,h–k) or three (b,d–g,m,n) independent experiments where points represent individual mice and bars represent mean. Error bars indicate mean + s.e.m. Data were log-transformed and compared using an unpaired two-tailed Welch’s t-test.
CD8+ T cells within the endogenous repertoire specific for PbRPL6120-12729 also showed impaired accumulation in the spleen and liver of δ−/− mice at 6 days (Extended Data Fig. 1b–d) and did not form memory in the spleen or liver (Extended Data Fig. 1e–h). γδ T cells were thereby required for the initiation phase of the CD8+ T cell response to RAS.
To assess whether genetically attenuated parasite vaccination was also γδ T cell dependent, B6 or δ−/− mice were vaccinated with P. berghei gene deletion mutants lacking the mei2 gene (PbΔmei2), which do not differentiate into blood-stage parasites but persist late into the liver stage, like the Δmei2 P. falciparum parasites30. A single dose of PbΔmei2 generated impaired memory CD8+ T cells against RPL6 in the spleen (Fig. 1h,j) and the liver (Fig. 1i,k) of δ−/− mice. Thus, vaccination with genetically attenuated parasites also required γδ T cells. Of note, the poor initial response in δ−/− mice did not extended to vaccination with blood-stage parasites (irradiated infected red blood cells) (Extended Data Fig. 1i), despite this response being categorized as relatively weak based on its CD4+ T cell help dependence31.
We next assessed whether the initiation of the CD4+ T cell response to RAS vaccination was also γδ T cell dependent. Splenic accumulation of MHC-II restricted P. berghei-specific PbT-II cells was dependent upon γδ T cell-mediated help in response to RAS vaccination (Fig. 1l,m), also resulting in fewer PbT-II cells in the livers of δ−/− mice (Extended Data Fig. 1j). PbT-I cell accumulation in the spleen (Fig. 1n) and liver (Extended Data Fig. 1k) of the δ−/− mice was also lower than the controls, demonstrating that the addition of large numbers of naive antigen-specific CD4+ T cells could not rescue the response of CD8+ PbT-I cells when γδ T cells were absent.
Vγ1+ γδ T cells initiate immunity to RAS
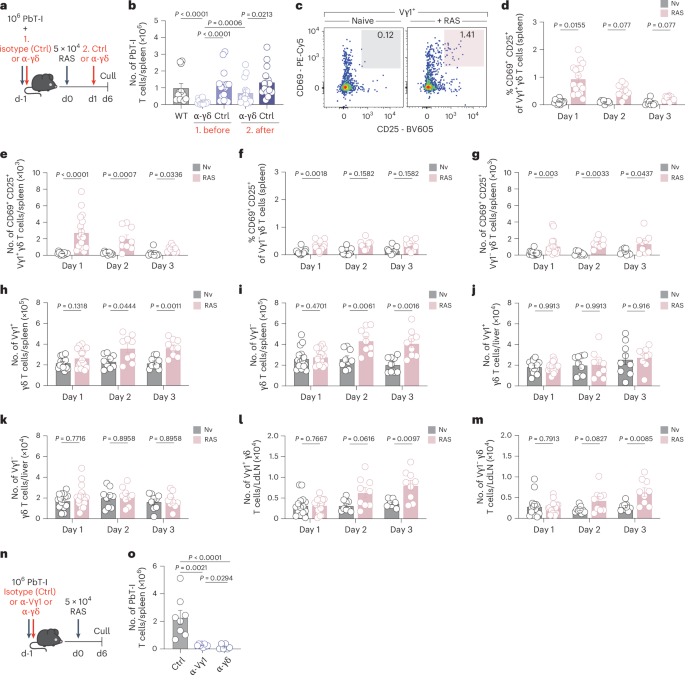
Blocking γδ T cell function before RAS vaccination with the pan-TCRδ blocking antibody (α-γδ, clone GL3) (Fig. 2a) impaired the PbT-I response comparably to that previously observed in δ−/− mice (Fig. 2b). In contrast, blocking γδ T cell function from 24 h after RAS injection had a milder effect (Fig. 2b), suggesting that the first 24 h are the crucial window of γδ T cell activation. We thereby examined early γδ T cell activation by splitting them into the two major populations found in the periphery: Vγ1+ or Vγ1− populations (Extended Data Fig. 2a). In the spleen, Vγ1+ γδ T cells upregulated the canonical T cell activation markers CD69 and CD25 1 day after RAS vaccination (Fig. 2c) with a transient increase in the proportion (Fig. 2d) and number (Fig. 2e) of activated cells. A small increase in the number and proportion of activated Vγ1− γδ T cells was also observed in the spleen (Fig. 2f,g and Extended Data Fig. 2a). γδ T cell expansion within both Vγ1+ and Vγ1− populations was observed from day 2 (Fig. 2h,i). In the liver, a small increase in the activation of Vγ1+, but not Vγ1−, γδ T cells, was detected at day 1 (Extended Data Fig. 2b–f), and an equivalent small increase in activated Vγ1+ γδ T cells seen in the liver dLN at day 2 (Extended Data Fig. 2g–k), indicating a slight delay in the initiation of the liver dLN response. Expansion of both Vγ1+ and Vγ1− γδ T cell populations was seen in the liver dLN but not in the liver (Fig. 2j–m). To confirm that downregulation of the TCR due to γδ T cell activation did not result in an inability to detect activated cells, we also examined γδ T cell activation in TCRδ-GDL mice, in which green fluorescent protein (GFP) can be used to detect γδ T cells. These analyses showed a similar level of activation of Vγ1+ γδ T cells within the spleen (Extended Data Fig. 2l,m). As specific activation of Vγ1+ γδ T cells was suggested, the functional consequence of impairing Vγ1+ γδ T cell activity was tested (Fig. 2n). Blockade of Vγ1+ γδ T cells had a similar effect on PbT-I accumulation as blockade of the entire γδ T cell population (α-γδ) (Fig. 2o). Vγ1+ γδ T cells therefore initiate the CD8+ T cell response to RAS in mice.
a, Experimental design. PbT-I cells were transferred into recipient mice 1 day before RAS vaccination (WT, n = 12). Mice were treated with an α-pan-γδ-TCR (α-γδ, clone GL3) (i.v.) or an isotype control mAb (Ctrl) (i.v.) either (1) before RAS (α-γδ, n = 16; Ctrl, n = 15) or (2) 24 h after RAS (α-γδ, n = 15; Ctrl, n = 15). b, Numbers of PbT-I cells in the spleen were assessed at day 6 post-vaccination. c, Vγ1+ γδ T cells in naive (Nv) (left) or RAS-vaccinated (right) WT (B6) mice 1 day after 5 × 104 RAS vaccination. d,f, Frequency of activated (CD69+ CD25+) Vγ1+ (d) or Vγ1− (f) γδ T cells in naive or RAS-vaccinated WT spleens at days 1 (Nv, n = 15; RAS, n = 17), 2 (Nv, n = 9; RAS, n = 9) or 3 (Nv, n = 9; RAS, n = 9) post-RAS. e,g, Number of activated (CD69+ CD25+) Vγ1+ (e) or Vγ1− (g) γδ T cells in naive or RAS-vaccinated WT spleens at days 1 to 3 post-injection. h,i, Number of splenic Vγ1+ (h) or Vγ1− (i) γδ T cells. j,k, Number of liver Vγ1+ (j) or Vγ1− (k) γδ T cells. l,m, Number of LdLN Vγ1+ (l) or Vγ1− (m) γδ T cells. n, Experimental design. o, Numbers of PbT-I cells at day 6 post-vaccination in isotype control mAb-treated (Ctrl) (n = 8), α-Vγ1-treated (n = 8) (i.v.) or α-pan-γδ-TCR-treated (n = 6) (i.v.) mice. Data show three (a,b) or four (c–o) independent experiments where points represent individual mice and bars represent mean. Error bars indicate mean + s.e.m. Data were log-transformed and compared using an ordinary one-way ANOVA or multiple unpaired two-tailed Welch’s t-tests (b,o) and corrected with Holm–Sidák multiple comparisons test (d–m).
Antigen presentation is intact in the absence of γδ T cells
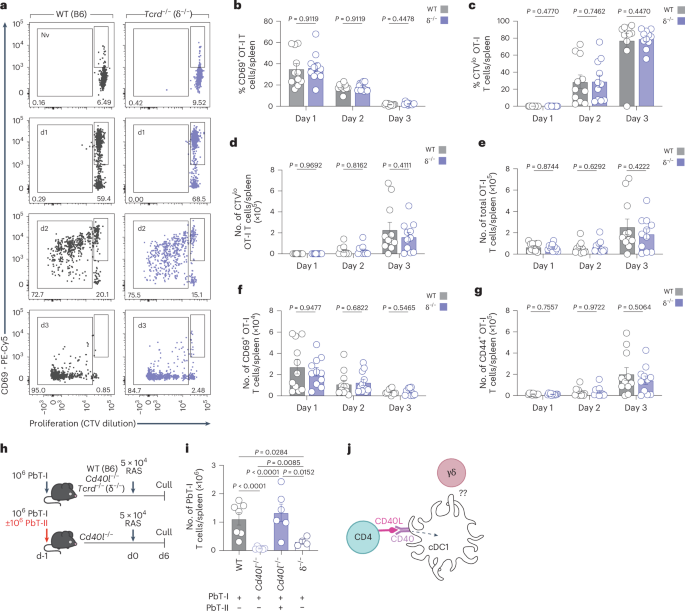
Due to the very early timing of γδ T cell activation, we hypothesized that the Vγ1+ γδ T cells may impact the ability of cDC1 to present antigen to CD8+ T cells in response to RAS vaccination. We therefore examined the initial upregulation of CD69 within OT-I cells following injection of SIINFEKL expressing CS5M sporozoites (CS5M-RAS). OT-I cells were used because adoptive transfer leads to spontaneous upregulation of CD69 on PbT-I cells for reasons yet to be determined, but OT-I cells allow for sensitive detection of CD69 upregulation in this system. OT-I T cells recapitulated the phenotype of PbT-I cells in δ−/− mice, with impaired responses in the spleen, liver dLN and liver 6 days after vaccination with CS5M-RAS (Extended Data Fig. 3a–c). At early time points post-CS5M-RAS vaccination, equal proportions and numbers of cell trace violet (CTV) labeled OT-I cells were present, upregulated cell surface expression of CD69 and divided in the spleens of B6 and δ−/− mice (Fig. 3a–g). We observed little evidence of OT-I cell activation in the liver (Extended Data Fig. 3d–j) and a small level of activation in the liver dLN, but this was equivalent between B6 and δ−/− mice (Extended Data Fig. 3k–q). These data indicated that responding T cells had access to antigenic signals capable of upregulating CD69 and initiating some proliferation even when γδ T cells were absent. Failure to accumulate at day 6 (Extended Data Fig. 3a–c), however, suggested a lack of signals for extended T cell expansion, differentiation and/or survival. This was suggestive of a lack of co-stimulation or cytokines in the T cell priming process, raising the intriguing possibility that γδ T cells were crucial for triggering upregulation of co-stimulation-like signals on cDC1.
A total of 106 CTV-labeled OVA-specific CD8+ (OT-I) T cells were transferred into WT or δ−/− recipient mice, which were vaccinated 1 day later with 5 × 104 CS5M-OVA RAS. OT-I cell numbers were assessed at days 1 to 3 (WT n = 11; Tcrd−/− n = 11, per day) after transfer. a, Flow cytometry plots of total OT-I cells in the spleen. b, Frequency of CD69+ OT-I cells in the spleen of either WT or δ−/− mice. c,d, Frequency (c) and number (d) of CTVlo OT-I cells in the spleen of either WT or δ−/− mice. e–g, Number of total (e), CD69+ (f) or CD44+ (g) splenic OT-I cells. h, Experimental design. i, WT (n = 7), δ−/− (n = 4) and Cd40l(Cd154)−/− mice received PbT-I cells with (n = 6) or without co-transferred PbT-II cells (n = 6) 1 day before vaccination with RAS. i, PbT-I cell counts in the spleen at day 6 post-vaccination. j, CD40 signaling to DCs is intact when CD40L-sufficient T cells are provided, showing that CD4+ T cells can provide CD40L in this system. Data show two (i) or three (a–g) independent experiments where points represent individual mice and bars represent mean. Error bars indicate mean + s.e.m. Data were log-transformed and compared using multiple unpaired two-tailed Welch’s t-tests and corrected using Holm–Sidák multiple comparisons test (b–g)or an ordinary one-way ANOVA (i).
γδ T cells are not required to supply the CD40L signal to cDC1s
We next hypothesized that γδ T cells may provide CD40L for signaling CD40 on cDC1s (ref. 32). To investigate this possibility, PbT-I cells were transferred into WT, δ−/− or Cd40lg−/− (Cd40l−/−) mice. CD8+ T cell expansion is dependent on help from CD4+ T cells in the RAS model9,10, and these CD4+ T cells were previously presumed to provide the CD40L signal. To test this assumption, one group of Cd40l−/− mice was also given CD40L-sufficient PbT-II cells (Fig. 3h). PbT-I cell accumulation in the spleen was severely diminished in Cd40l−/− mice (Fig. 3i), confirming that CD8+ T cell accumulation in this model is dependent on CD40L signaling. Addition of CD40L-sufficient PbT-II cells, however, rescued the PbT-I cell response, even though γδ T cells lacked expression of CD40L in these mice. Therefore, CD4+ T cells can provide the CD40L signal required, suggesting that γδ T cell-derived CD40L is not essential to this response. Furthermore, rescue by CD40L-sufficient PbT-II cells showed that this signal is normally provided by CD4+ T cells but is insufficient to ensure an appropriate CD8+ T cell response if γδ T cells are absent (Fig. 3i,j).
IL-4 is required for the CD8+ T cell response to RAS vaccination
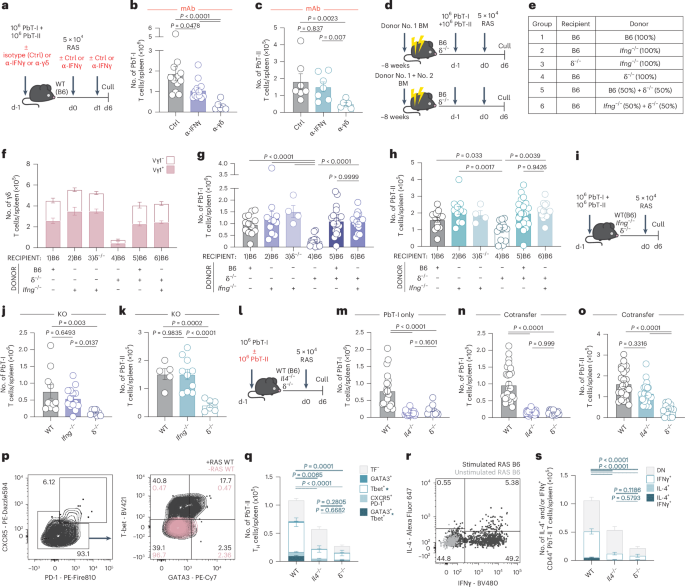
As CD40L was not the crucial signal provided by γδ T cells, we rationalized that they may provide ‘signal 3’ that is a cytokine signal, for differentiation and expansion of responding T cells. As IL-4 was identified as a crucial factor in the generation of memory CD8+ T cells in the liver following RAS vaccination12,13 and splenic γδ T cells can secrete IL-4 and IFNγ24,33, we next investigated the role of these cytokines. Six days after RAS vaccination, there was an increase in the number and proportion of splenic Vγ1+ γδ T cells that produced both IFNγ and IL-4 (Extended Data Fig. 4a–c). Vγ1− γδ T cells made IFNγ in response to RAS but no detectable IL-4 (Extended Data Fig. 4d–f). IFNγ blockade (Fig. 4a) impaired the accumulation of PbT-I but did not completely recapitulate the effect of γδ T cell blockade (α-γδ) (Fig. 4b) and had no effect on the accumulation of PbT-II T cells (Fig. 4c). To investigate if γδ T cells were a crucial source of IFNγ for PbT-I cell accumulation, we generated several groups of mixed bone marrow (BM) chimeras (Fig. 4d,e). Eight weeks after reconstitution, the γδ T cell compartment was reconstituted as expected (Fig. 4f). Assessment of the PbT-I and PbT-II cell responses 6 days after RAS vaccination demonstrated equal PbT-I (Fig. 4g) and PbT-II (Fig. 4h) cell accumulation in the spleens of Ifng−/−+ δ−/−→B6 chimeras (Group 6) when compared to B6 + δ−/−→B6 chimeras (Group 5) (Fig. 4g,h), suggesting that γδ T cells were not an essential source of IFNγ for PbT-I T cell accumulation. Accumulation of both PbT-I and PbT-II T cells was also similar between B6 → B6 (Group 1) and Ifng−/−→B6 (Group 2) chimeras (Fig. 4g,h), suggesting either that the source of IFNγ was not BM derived, or that the transferred PbT-I and/or PbT-II T cells were providing IFNγ. Co-transfer of PbT-I and PbT-II T cells into IFNγ−/− hosts (Fig. 4i) further demonstrated no impairment in PbT-I or PbT-II T cell accumulation in the spleen at day 6 (Fig. 4j,k). When combined, these data suggest that IFNγ contributes to PbT-I T cell accumulation in response to RAS vaccination, but γδ T cells are not the essential source of this cytokine, which is likely derived from CD4+ and/or CD8+ T cells.
a, Experimental design. b,c, PbT-I and PbT-II cells were transferred into WT (B6) mice treated with an α-IFNγ blocking antibody (i.p.) or an isotype control (Ctrl) (i.p) or α-pan-γδTCR (i.v.) 1 day before immunization with RAS. Shown is the number of PbT-I cells (Ctrl, n = 12; α-IFNγ, n = 12; α-γδ, n = 6) (b) and PbT-II cells (Ctrl, n = 7; α-IFNγ, n = 7; α-γδ, n = 6) (c) in the spleen on day 6 post-vaccination. d, Experimental design. e–h, Single and mixed BM chimeras were prepared using BM from WT, δ−/−, and Ifng−/− donors as indicated. Eight weeks later, chimeras were given PbT-I and PbT-II cells 1 day before RAS vaccination and then analyzed 6 days later. e, Chimera groups relating to f–h. f, Spleen γδ T cell numbers in chimeras separated by Vγ1 expression (Group 1, n = 13; Group 2, n = 11; Group 3, n = 4; Group 4, n = 14; Group 5, n = 20; Group 6, n = 15). g,h, Splenic PbT-I (g) and PbT-II (h) cell numbers. i, Experimental design. j,k, WT, Ifng−/− and δ−/− mice received PbT-I and PbT-II cells before immunization with RAS. j,k, Number of PbT-I cells (j) in the spleen of WT (n = 10), Ifng−/− (n = 14) and δ−/− (n = 9) mice or PbT-II cells (k) in WT (n = 5), Ifng−/− (n = 10) and δ−/− (n = 6) mice. l, Experimental design. m–o, PbT-I cells were transferred into WT, Il4−/− or δ−/− mice 1 day before immunization. An additional cohort of Il4−/− mice received both PbT-I and PbT-II cells. m, Number of PbT-I cells in the spleen of WT (n = 21), Il4−/− (n = 20) or δ−/− (n = 11) mice, 6 days post-vaccination. n,o, Numbers of PbT-I cells (n) and PbT-II cells (o) in the spleen of WT (n = 26), Il4−/− (n = 22) or δ−/− (n = 12) mice that received both PbT-I and PbT-II cells. p, Transcription factor gating in the spleen. q, Number of spleen CD44+ PbT-II cells expressing no transcription factors tested (TF-), GATA3 or Tbet alone, or co-expressing CXCR5 and PD-1, or GATA3 and Tbet from WT (n = 16), Il4−/− (n = 9) or δ−/− (n = 7) mice. Splenocytes at day 6 post-vaccination were restimulated ex vivo with PMA/ionomycin for 5 h to assess cytokine production. r, IL-4 and IFNγ co-expression in spleen CD44+ PbT-II cells from WT (n = 16), Il4−/− (n = 9) or δ−/− (n = 7). s, Number of IL-4- and/or IFNγ-expressing CD44+ PbT-II cells. Data were pooled from three independent experiments where points represent individual mice and bars represent mean. Flow cytometry plots are representative. Error bars indicate mean + s.e.m. Data were log-transformed and compared using an ordinary one-way ANOVA.
Strikingly, IL-4 had a profound effect on PbT-I accumulation either when α-IL-4 antibody was used to mediate blockade (Extended Data Fig. 4g,h) or responses were measured after transfer of PbT-I cells into Il4−/− hosts (Fig. 4l,m). IL-4 was therefore crucial for the response, and the absence of IL-4 mirrored the effect of γδ T cell deficiency.
To further investigate if, as previously reported, CD4+ T cells were the relevant source of IL-412, we transferred IL-4-sufficient PbT-I cells with or without IL-4-sufficient PbT-II cells into B6 or Il4−/− mice (Fig. 4l). Accumulation of PbT-I cells was impaired in the Il4−/− mice even in the presence of IL-4-sufficient antigen-specific CD4+ T cells (Fig. 4n). In contrast, antigen-specific CD4+ T cells expanded in the spleen in both B6 and Il4−/− mice (Fig. 4o), demonstrating two phenomena: 1) the CD4+ T cell response was not IL-4 dependent and 2) IL-4-sufficient CD4+ T cells could not rescue the antigen-specific CD8+ T cell response. CD4+ T cells, therefore, are not the crucial source of IL-4 for CD8+ T cell accumulation following RAS vaccination.
To assess the effect of IL-4 on the T helper phenotype, we examined IFNγ and IL-4 secretion and transcription factor expression in PbT-II cells. Very few PbT-II cells isolated from vaccinated B6 mice differentiated into Tfh phenotype cells, based on expression of CXCR5 and PD-1, with no difference in the number or proportion of Tfh cells detected in the absence of IL-4 (Fig. 4p,q and Extended Data Fig. 4i). In contrast, larger numbers and proportions of PbT-II cells expressed either Tbet or both Tbet and GATA3, suggesting a mixed Th1 and Th1/2 phenotype, with the latter dependent on both IL-4 and γδ T cells (Fig. 4p,q and Extended Data Fig. 4i). Cytokine expression analysis showed that PbT-II cells fell into three categories: IFNγ+ only, IFNγ+/IL-4+ or double negative (DN) (Fig. 4r,s). Strikingly, PbT-II cells isolated from δ−/− or Il4−/− mice did not produce IL-4 and showed a reduction in IFNγ (Fig. 4s and Extended Data Fig. 4j), suggesting that both IL-4 and γδ T cells were required for CD4+ T cells to produce IL-4. Collectively, these data show that CD4+ T cells proliferate in response to RAS vaccination in the absence of IL-4 but are impaired in their differentiation into IL-4 producing effectors.
γδ T cells produce IL-4 in experimental and clinical malaria
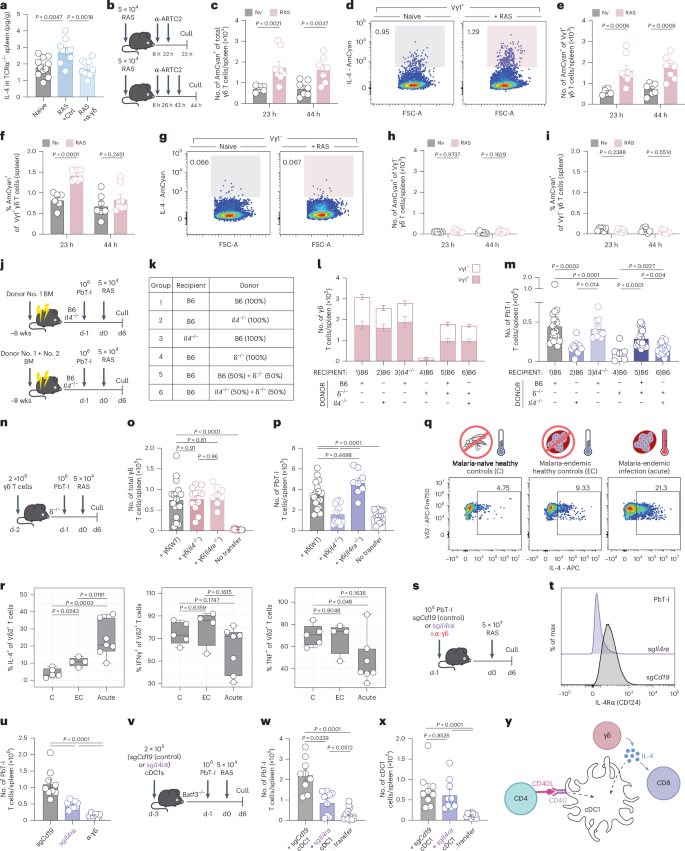
To determine if γδ T cells provide IL-4 early in the response to RAS vaccination in mice, we took two experimental approaches. First, IL-4 protein measured in the spleen of RAS-vaccinated TCRα−/− mice at 6 h was significantly increased only when γδ T cell function was intact (Fig. 5a). Secondly, 4C13R dual IL-4/IL-13 reporter mice that were RAS vaccinated and injected with α-ARTC2 antibody to protect the cytokine-secreting cells33 (Fig. 5b) showed an increase in the total number of γδ (Fig. 5c) and a specific increase in the number (Fig. 5d,e) and proportion (Fig. 5d,f) of Vγ1+ IL-4 producing γδ T cells at 23 h. The γδ T cell activation seen in the 4C13R mirrored that observed in B6 mice in previous experiments (Extended Data Fig. 5a–d and Fig. 2). We did not detect an increase in IL-4 production by Vγ1− γδ T cells (Fig. 5g–i) or an increase in IL-4 production by γδ T cells isolated from the liver at either timepoint (Extended Data Fig. 5e–h). Splenic Vγ1+ γδ T cells therefore produce IL-4 in response to RAS vaccination in vivo.
TCRα−/− mice were injected with α-pan-γδTCR (or isotype, Ctrl) (i.v.) 1 day before RAS vaccination. After 6 h, IL-4 concentration was assessed. a, Concentration per gram of spleen in naive (n = 11), vaccinated Ctrl (n = 10) or vaccinated α-pan-γδTCR (n = 11) TCRα−/− mice. b, Experimental design (c–i). B6.4C13R mice were vaccinated with RAS. Naive (23 h, n = 6; 44 h, n = 7) and vaccinated (23 h or 44 h, n = 8) mice were treated with α-ARTC2 (i.v.) and then culled at 23 or 44 h post-RAS. c, IL-4+ (AmCyan+) γδ T cell numbers in enriched spleen. d, IL-4 expression in splenic Vγ1+ γδ T cells. e, IL-4+ (AmCyan+) Vγ1+ γδ T cell numbers in enriched spleen. f, IL-4+ (AmCyan+) cell frequency in splenic Vγ1+ γδ T cells. g, IL-4 expression in splenic Vγ1− γδ T cells. h, IL-4+ (AmCyan+) Vγ1− γδ T cell numbers in enriched spleen. i, IL-4+ (AmCyan+) cell frequency in splenic Vγ1− γδ T cells. j, Experimental design (k–m). Chimeras were given PbT-I cells 1 day before RAS vaccination and then analyzed 6 days later. k, Chimeras were reconstituted with B6, δ−/−, Il4−/− donor cells (Group 1, n = 20; Group 2 or 3, n = 10; Group 4, n = 6; Group 5 or 6, n = 20). l, Spleen γδ T cell numbers at day 6 post-RAS. m, PbT-I cell numbers in the spleen. n, Experimental design. o,p, δ−/− mice received γδ T cells from WT, Il4−/− or Il4ra−/− donors 1 day before transfer of PbT-I cells. Recipient mice were vaccinated with RAS and analyzed 6 days later. o, Quantified splenic γδ T cells 6 days after RAS vaccination in δ−/− mice receiving γδ T cells from WT (+γδ WT) (n = 17), Il4−/− (+γδ Il4−/−) (n = 12), Il4ra−/− (+γδ Il4ra−/−) (n = 8), or no γδ T cells (no transfer) (n = 13). p, PbT-I cell counts in the spleen 6 days after RAS vaccination. q, PBMCs were collected from malaria-naive healthy controls (C, n = 5), malaria-endemic healthy controls (EC, n = 4) and malaria-endemic patients with acute malaria infection (acute, n = 7). r, Frequency of IL-4, IFNγ- and TNF-producing Vδ2+ γδ T cells after PMA/ionomycin stimulation. s, Experimental design. t,u, CRISPR-Cas9 ablated sgCd19 (control) or sgIl4ra PbT-I cells were transferred into recipients 1 day before RAS vaccination. t, Surface IL-4Rα expression on sgCd19 (control) or sgIl4ra PbT-I cells at day 6. u, Numbers of PbT-I cells in the spleen of mice receiving sgCd19 (n = 9) or sgIl4ra (n = 9) PbT-I cells or sgCd19 cells with α-pan-γδTCR (n = 6) at day 6. v, Experimental design. w,x, Batf3−/− recipients received sgCd19 (control) or sgIl4ra gene-edited CD24+ cDC1 2 days before T cell transfer. Recipient mice were vaccinated with RAS and analyzed 6 days later. w, Spleen PbT-I cell numbers in Batf3−/− mice that received sgCd19 (control) (n = 9) or sgIl4ra cDC1s (n = 9) or no cDC1s (- transfer) (n = 10). x, Splenic cDC1 counts six days after RAS vaccination. y, IL-4 acts directly on CD8+ T cells and cDC1s for CD8+ T cell accumulation. Data show two (a–i,s–x), four (l–p) or three (c,d,j,k) independent experiments where points represent individual mice and bars represent mean. Flow cytometry plots are representative. Box and whisker plot center line represents the median; box limits indicate the upper and lower quartiles and whiskers extend to 1.5x the interquartile range. Error bars indicate mean + s.e.m. Data were log-transformed and compared using an ordinary one-way ANOVA (a,l–p,u–x), multiple unpaired two-tailed Welch’s t-tests and Holm–Sidák multiple comparisons correction (c–i) or multiple unpaired two-tailed t-test or (TNF) Mann–Whitney U test after Shapiro–Wilk normality testing (r). h, hours.
To determine if γδ T cell-derived IL-4 had a functional impact on PbT-I T cell accumulation, we generated mixed BM chimeras (Fig. 5j,k). Eight weeks after reconstitution, the γδ T cell compartment in the spleen (Fig. 5l) and the liver (Extended Data Fig. 5i) reconstituted as expected. Six days after RAS vaccination, impaired PbT-I cell accumulation in the spleens of Il4−/−+δ−/−→B6 chimeras (group 6) was observed when compared to B6 + δ−/−→B6 chimeras (group 5) (Fig. 5m). These data demonstrated that γδ T cells were the important source of IL-4, as B6 + δ−/−→B6 chimeras contained γδ T cells that could produce IL-4, whereas the Il4−/−+δ−/−→B6 chimeras lacked these cells. There was a small but significant increase in PbT-I accumulation when δ−/−→B6 (Group 4) chimeras were compared with Il4−/−+δ−/−→B6 (Group 6) chimeras, suggesting an additional small contribution of γδ T cells that was not IL-4 dependent, but the nature of this contribution is yet to be determined. Nonetheless, these data strongly suggest that γδ T cells are the essential source of IL-4 for the initiation of an effective CD8+ T cell response to RAS vaccination.
To further confirm that γδ T cells are the crucial initial source of IL-4, we reconstituted δ−/− mice with splenic γδ T cells from either WT or Il4−/− or Il4ra−/− donors and tested whether these cells could rescue the PbT-I response. TCRδ-deficient mice received either splenic γδ T cells or no γδ T cell transfer (Fig. 5n). Six days after RAS vaccination (8 days after γδ T cell transfer), γδ T cells were recovered from the spleens demonstrating that the transfer was effective (Fig. 5o). As hypothesized, B6-derived but not Il4−/−-derived γδ T cells supported PbT-I cell accumulation in the spleen in response to RAS vaccination, confirming that these cells provided IL-4 in this vaccination setting (Fig. 5p). Support for PbT-I T cell accumulation by Il4ra−/− γδ T cells (Fig. 5p) implied that γδ T cells do not need to sense IL-4 to provide it, suggesting these cells are the crucial initiators of IL-4 production.
In humans, the population of γδ T cells that expand and correlate with protection following RAS vaccination are Vγ9+Vδ2+ γδ T cells, the most abundant γδ T cell population in the peripheral blood16,17,19,20 and also found in the spleen34. We therefore asked if Vγ9+Vδ2+ γδ T cells also produce IL-4, or other inflammatory cytokines, in the context of natural infection. Peripheral blood mononuclear cells (PBMCs) from individuals with an uncomplicated P. falciparum malaria infection were PMA-stimulated and cytokine production quantified (Fig. 5q). IL-4 production by Vδ2+ γδ T cells was higher in individuals with malaria compared to either healthy, currently uninfected individuals from the same area or healthy, malaria-naive Australians (Fig. 5r and Extended Data Fig. 6a–c). In contrast, IFNγ and TNF production was comparable or reduced between malaria-infected and healthy controls (Fig. 5r). CXCR5 and CCR7 are associated with lymphoid tissue homing in Vγ9+Vδ2+ γδ T cells34,35. Analysis of IL-4 expression showed that this cytokine was largely produced by cells expressing CXCR5 and CCR7, two chemokine receptors that assist with entry to lymphoid tissues including the spleen (Extended Data Fig. 6c). This was different from the pattern seen in IFNγ-producing Vδ2+ T cells with a higher proportion of IFNγ producers within the CXCR5− CCR7− subset (Extended Data Fig. 6c). These data suggest that during human malaria initiated by sporozoite infection, Vδ2+ T cells that may have spleen homing properties also have the capacity to produce IL-4.
IL-4 signals both CD8+ T cells and cDC1s to initiate the CD8+ response
IL-4 is a prototypical T helper 2 cytokine that has potent effects on CD4+ T cell differentiation and function36 but less well-studied impacts on CD8+ T cells. We therefore asked if the RAS-induced γδ T cell-derived IL-4 acted directly on the responding CD8+ T cells. CRISPR-Cas9 deletion of the IL-4Rα gene (Il4ra) (or Cd19 as a control) from naive PbT-I cells before transfer and immunization with RAS (Fig. 5s,t) showed that direct IL-4Rα signaling was important for PbT-I cell accumulation in the spleen (Fig. 5u).
PbT-I cells lacking the IL-4R still retained a modest capability to accumulate when compared to a complete failure in the absence of γδ T cells (Fig. 1a) or IL-4 itself (Fig. 4m), suggesting an additional activity that contributes to CD8+ T cell accumulation. To investigate whether this involved direct signaling of cDC1 by IL-4, we developed an in vivo model for cDC1 manipulation. Donor cDC1 were expanded in vivo in B6 mice37 before enrichment, CRISPR-Cas9-mediated deletion of either Cd19 or Il4ra and transfer into Batf3−/− mice for replenishment of the cDC1 pool (Fig. 5v). Splenic PbT-I cell accumulation was impaired when cDC1 did not express IL-4Rα (sgIl4ra) (Fig. 5w), suggesting direct IL-4 signaling in cDC1. Numbers of cDC1 recovered were equivalent between groups (Fig. 5x and Extended Data Fig. 6d). Collectively, these experiments demonstrate that IL-4 acts directly on DC and on CD8+ T cells for optimal CD8+ T cell expansion in the context of RAS vaccination (Fig. 5y).
IL-4 and IFNγ synergize with CD40 to drive IL-12 production by cDC1s
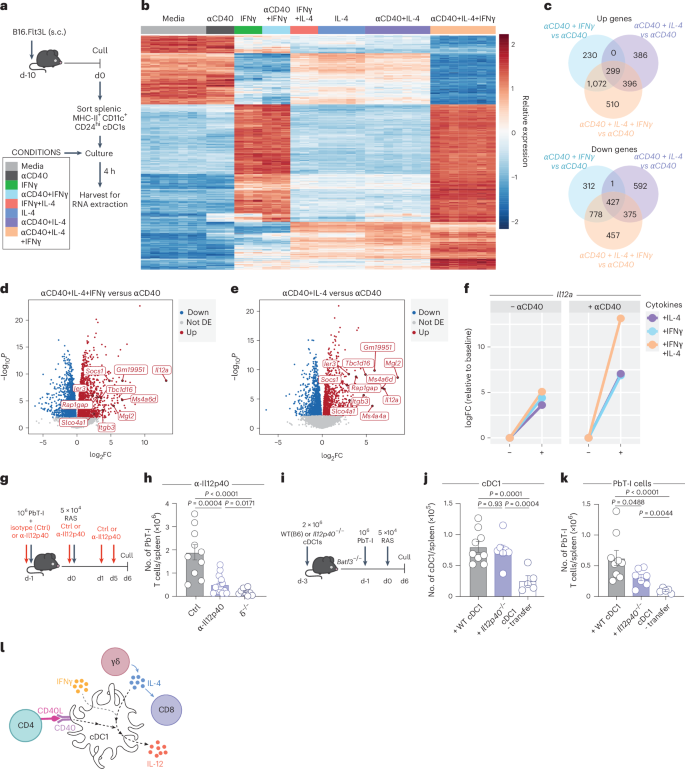
To identify the molecules induced by IL-4 in cDC1s, gene expression was assessed in vitro following stimulation with media alone, αCD40, IFNγ, IL-4, IFNγ+IL-4, αCD40 + IFNγ, αCD40 + IL-4 or αCD40 + IL-4 + IFNγ. Expanded cDC1s were sort purified and then cultured in each condition before isolation of RNA at 4 h (Fig. 6a).
a, Experimental design. b–f, Sorted cDC1s were cultured for 4 h in media alone (n = 7), αCD40 (n = 3), αCD40 + IL-4 (n = 7), αCD40 + IFNγ (n = 3), αCD40 + IL-4 + IFNγ (n = 7), IL-4 (n = 5), IFNγ (n = 3) or IL-4 + IFNγ (n = 3). s.c., subcutaneous. b, Heatmap of differentially expressed genes (DEGs) between media alone and αCD40 + IL-4 + IFNγ, across the different stimulation conditions. c, Upregulated (Up) and downregulated (Down) DEGs between cDC1s stimulated with αCD40 + IL-4, αCD40 + IFNγ or αCD40 + IL-4 + IFNγ relative to αCD40 only. d,e, Volcano plots of DEGs between αCD40 only and αCD40 + IL-4 + IFNγ (d) or αCD40 + IL-4 (e) stimuli. f, Log fold change (FC) in Il12a expression in cells subjected to IL-4 or IFNγ, or a combination of both relative to baseline (no cytokine) when in the absence (left) or presence (right) of αCD40. g, Experimental design. h, WT (B6) or δ−/− (n = 10) mice received PbT-I cells 1 day before RAS vaccination and WT mice were treated with either α-IL-12p40 (i.p.) (n = 14) or an isotype control mAb (Ctrl) (i.p.) (n = 10). h, Number of PbT-I cells in the spleen at day 6 post-RAS vaccination. i, Experimental design. j,k, CD24+ cDC1 from WT (B6) or Il12p40−/− mice were transferred into Batf3−/− recipients followed by PbT-I cells 2 days later. Mice were vaccinated with RAS and analyzed at day 6 post-RAS. j, Splenic cDC1 counts in Batf3−/− mice that received WT (n = 9), Il12p40−/− (n = 7) cDC1 or no cDC1s (- transfer) (n = 5). k, Number of PbT-I cells in spleens of Batf3−/− mice that received WT (n = 9), Il12p40−/− (n = 7) cDC1 or no cDC1s (- transfer) (n = 5). l, γδ T cell-derived IL-4 acts on cDC1s with IFNγ to promote IL-12 production that, along with IL-4, is required for CD8+ T cell accumulation. Data show seven (a–f) or two (h,j,k) independent experiments where points show pooled biological replicates (f) and individual mice (h,j,k). Error bars indicate mean + s.e.m. Data were log-transformed and compared by an ordinary one-way ANOVA.
Analysis of sequenced RNA revealed a marked transcriptomic shift in the presence of IL-4 that was augmented by the addition of αCD40 (Fig. 6b,c), a shift further augmented by the addition of IFNγ (Fig. 6b). Specifically, there were 1,081 upregulated and 1,395 downregulated genes in response to αCD40 + IL-4 when compared to αCD40 alone (Fig. 6c and Supplementary Table 1). This was substantially increased by the addition of IFNγ, with 2,277 genes upregulated and 2,037 genes downregulated in the presence of αCD40 + IL-4 + IFNγ (Fig. 6c and Supplementary Table 1). Il12a was the most upregulated gene when comparing αCD40 with αCD40 + IL-4 + IFNγ (LFC 13.14, FDR 9.03 × 10−8) and the third most upregulated gene when comparing αCD40 + IL-4 with αCD40 alone (LFC 7.055, FDR 8.97 × 10−6) (Fig. 6d,e). This gene encodes the p35 subunit of IL-12, which, when combined with the p40 subunit (which is constitutively expressed by cDC1), makes bioactive IL-12. Efficient upregulation of Il12a was dependent on the combination of CD40, IFNγ and IL-4 signals (Fig. 6f).
IL-12 blockade in vivo (Fig. 6g) showed significant impairment in PbT-I accumulation (Fig. 6h). To confirm that cDC1 were the crucial source of IL-12, Batf3−/− mice were reconstituted with cDC1 from Il12b−/− (Il12p40−/−) mice (Fig. 6i,j), showing that PbT-I cell accumulation was impaired when cDC1 were unable to produce bioactive IL-12 (Fig. 6k). This supports a model where CD40 signaling synergizes with IL-4 and IFNγ in cDC1 to induce IL-12 that is crucial for the accumulation of CD8+ T cells in response to RAS vaccination (Fig. 6l).
IL-4 promotes CD8+ T cell expansion by increasing IL-12R expression
IL-4 and IL-12 were both essential for enhanced accumulation of CD8+ T cells, suggesting synergistic actions in CD8+ T cell expansion. To isolate the effects of the cytokines, we first examined the impact of IL-4 and IL-12 on CD8+ T cell expansion in vitro.
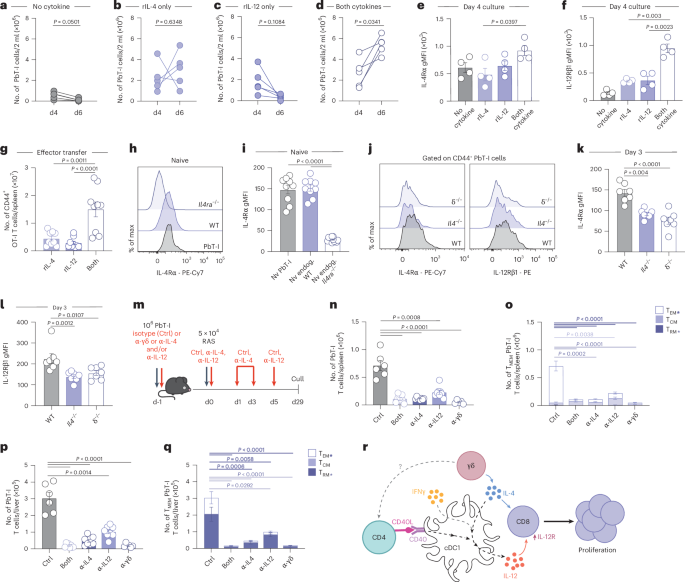
PbT-I cells activated by peptide-coated antigen presenting cells showed limited cell growth in the absence of exogenous cytokine (Fig. 7a). Addition of either IL-4 or IL-12 only modestly impacted the number of cells recovered (Fig. 7b,c), but addition of both IL-4 and IL-12 resulted in significantly higher cell recovery at day 6 (Fig. 7d), suggesting that IL-4 and IL-12 act synergistically to increase the T cell response in vitro.
a–d, PbT-I cells were peptide-activated in vitro in media alone (no cytokine) (n = 5) (a) or in the presence of 60 ng ml−1 rIL-4 (rIL-4 only) (n = 5) (b), 10 ng ml−1 rIL-12 (rIL-12 only) (n = 5) (c) or both rIL-4 and rIL-12 (both cytokines) (n = 5) (d). e,f, IL-4Rα (e) and IL-12Rβ1 (f) surface expression on activated PbT-I cells 4 days after culture in the presence of different cytokines (n = 4). OT-I cells were activated in vitro in the presence of 60 ng ml−1 rIL-4 (n = 9), 10 ng ml−1 rIL-12 (n = 9), or both rIL-4 and rIL-12 (both) (n = 9) and then transferred into recipient mice at day 4 of culture. g, Number of OT-I cells in the spleen 3 days after effector transfer. h, Surface IL-4Rα expression on endogenous naive CD8+ T cells in Il4ra−/− (top), WT (B6) mice (middle), or on naive PbT-I cells transferred into naive WT recipients (bottom). i, Geometric mean fluorescence intensity (gMFI) of surface IL-4Rα expression on endogenous naive CD8+ T cells in Il4ra−/− (n = 12), WT mice (n = 9) or on naive PbT-I cells transferred into naive WT recipients (n = 9). B6, Il4−/− and δ−/− mice received 106 PbT-I cells 1 day before RAS vaccination. PbT-I cells were then assessed 3 days later. j, Surface IL-4Rα (left) or IL-12Rβ1 (right) expression on PbT-I cells at day 3. k,l, gMFI of surface IL-4Rα (k) or IL-12Rβ1 (l) expression on PbT-I cells at day 3 post-vaccination (n = 7). m, Experimental design. n–q, Mice received PbT-I cells 1 day before RAS vaccination and were treated with an isotype control (Ctrl) (i.p.) (n = 6), α-pan-γδTCR (i.v.) (n = 5), α-IL-12p40 (i.p.) (n = 8), α-IL-4 (i.p.) (n = 8) or both α-IL-4 and α-IL-12p40 (n = 8). n,p, Number of PbT-I cells in the spleen (n) and liver (p), 29 days after vaccination. o,q, Frequency of TCM, TEM and TRM within the CD44+ PbT-I cell compartment in the spleen (o) and liver (q) 29 days after RAS vaccination. r, γδ T cell-derived IL-4 acts on cDC1s, along with CD40 signals and IFNγ, to promote IL-12 production, which, together with IL-4, signals directly on CD8+ T cells to drive proliferation and therefore enhance liver TRM formation. Data show five (a–d), four (e,f), three (g) or two (i–k,h–q) independent experiments where points show biological replicates (a–f) and individual mice (e–q). Histogram plots are representative. Error bars indicate mean + s.e.m. Data were log-transformed then compared using a paired two-tailed t-test (a–d) or an ordinary one-way ANOVA (e–q).
To explore whether the synergy between IL-4 and IL-12 was mediated by changes in cytokine sensitivity resulting from alterations in receptor expression, we assessed IL-4R and IL-12R expression on T cells at day 4 of culture. Compared to addition of IL-4 alone, a combination of IL-4 and IL-12 induced a small increase in IL-4R (Fig. 7e). More substantially, the addition of IL-4 + IL-12 increased expression of IL-12R over either cytokine alone (Fig. 7f), suggesting that the combination of IL-4 and IL-12 amplifies the sensitivity of CD8+ T cells to IL-12 signaling through increased expression of IL-12R. Further, in vitro activated OT-I T cells stimulated with IL-4 and IL-12 were maintained in greater numbers when transferred into mice (Fig. 7g).
To extend these findings in vivo, we first confirmed that naive PbT-I cells expressed detectable levels of IL-4Rα (Fig. 7h,i). PbT-I cells adoptively transferred into either B6, Il4−/− or δ−/− mice and exposed to RAS vaccination only showed full upregulation of IL-4Rα and IL-12R in the presence of IL-4 or γδ T cells (Fig. 7j–l), supporting our in vitro findings and implicating γδ T cells as the mediator.
γδ T cell-derived IL-4 and cDC1-derived IL-12 promote liver TRM cells
To confirm that removal of both IL-4 and IL-12 in vivo would recapitulate the lack of γδ T cells and therefore inhibit the formation of protective liver TRM cells (Fig. 1), we blocked IL-4 or IL-12, or both, in the context of RAS vaccination and then examined TRM numbers in the liver 29 days later (Fig. 7m). Enumeration of the number of liver TRM cells showed that IL-4 had a dominant effect on liver TRM formation, but blockade of both cytokines phenocopied blockade of γδ T cells (with α-γδ Ab) in the spleen (Fig. 7n,o) and liver (Fig. 7p,q). These data show that γδ T cells drive the response to RAS via delivery of IL-4 to DCs, which in turn acts with IFNγ to drive IL-12 production. The γδ T cell-derived IL-4 and DC-derived IL-12 then synergize to enhance the expansion of CD8+ T cells (Fig. 7r), a proportion of which will differentiate into protective liver TRM cells.