Experimental animals
A total of 81 five-week-old male BALB/c mice were purchased from the National Laboratory Animal Center, Taipei, Taiwan. All mice were used at 5 weeks of age when they weighed 20–22 g. Food and water were provided ad libitum. Euthanize experimental animals using carbon dioxide in accordance with approved protocol. The animal care and handling protocols were approved by the Animal Use Protocol Chung-Shan Medical University Animal Center Affidavit (IACUC Approval No: 2572). All animal experiments were carried out under institutional policies and guidelines for the care and use of laboratory animals, and all efforts were made to minimize animal suffering. The procedures fully complied with the approved guidelines and the ARRIVE guidelines (https://arriveguidelines.org).
Toxocara canis larval preparation
Toxocara canis was prepared via the Lu method with slight modifications22. The eggs of T. canis were obtained from the uterus of adult female parasites from infected dogs. The embryonated eggs were incubated in 2% formalin at room temperature with gentle weekly agitation for 3–5 weeks to obtain the infective eggs. The eggs were washed three times with deionized water to remove the formalin. The mixture was centrifuged for 10 min at 1400 × g. For eggshell lysis, the supernatant was discarded and the pellet was agitated in 10 ml of 3% sodium hypochlorite for 1 h. The mixture was centrifuged for 10 min at 1400 × g. The eggs were washed three times with deionized water and collected for experimental studies.
Animal infection
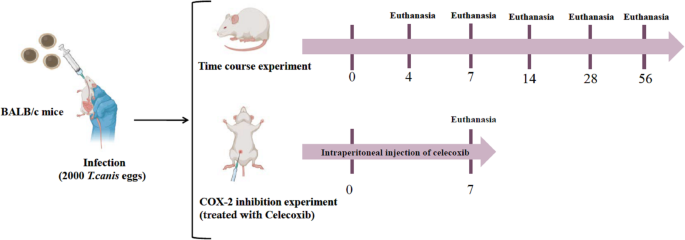
For the time course experiment, 54 BALB/c mice were randomly assigned to 6 groups, each consisting of 9 mice. The mice in the T. canis infection group were sacrificed on days 4, 7, 14, 28, and 56 after infection. In the infection groups, each mouse received 2000 embryonated T. canis eggs through a stomach tube. The noninfected control group received an equivalent volume of deionized water on day 0, as did the infected groups, and was sacrificed on day 56. For the COX-2 inhibition experiment, 27 BALB/c mice were randomly assigned to 3 groups, each consisting of 9 mice. These groups included the uninfected control group, the T. canis infection, and the T. canis infection group treated with pharmacological inhibition of COX-2 (Celecoxib, Viatris Healthcare Hong Kong). In the groups infected with T. canis and treated with celecoxib, each mouse received 2000 embryonated T. canis eggs through a stomach tube. In the celecoxib treatment group, celecoxib was administered intraperitoneally at a dose of 5 mg/kg/0.5 ml/day from day 0 of T. canis infection to day 7, with euthanasia of the mice on day 7 after infection.
Recovery of larvae from the lungs of T. canis-infected mice
The lung tissues were homogenized at a 1:30 ratio (tissue: artificial pepsin digestion solution containing pepsin from Sigma, USA) via a digestive juice grinder. The mixture was then evenly stirred and digested in a 37 °C incubator for 1 h. The lower sediment layer was carefully collected via a dropper and transferred to a glass plate, and the number of T. canis larvae was counted with an inverted microscope (Nikon, Tokyo, Japan).
Enzyme-linked immunosorbent assay
The concentrations of PGE2, INF-γ, TNF-α, IL1-β, IL-6 and NO in the homogeneous solution of the lungs were determined via commercially available enzyme immunoassay kits (Cayman Chemical, USA) according to the manufacturer’s instructions.
Casein/plasminogen zymography
PA activity was determined by casein/plasminogen zymography according to the Hou method with slight modifications23. In a typical procedure, lung proteins were subjected to 7.5% (mass/volume) SDS‒polyacrylamide gel electrophoresis after copolymerization with 0.1% casein (Sigma, USA) and plasminogen (13 mg/mL; American Diagnostica). The stacking gels were composed of 4% polyacrylamide (mass/volume). Electrophoresis was performed in running buffer (25 mM Tris, 250 mM glycine, and 1% SDS) at room temperature and 110 V for 1 h. The gel was washed twice at room temperature for 30 min each in 2.5% Triton X-100 and then twice with double distilled H2O for 10 min each. The gel was incubated in reaction buffer (50 mM Tris-HCl, pH 8.0, containing 10 mM CaCl2 and 0.01% NaN3) at 37 °C for 18 h. The gel was stained with 0.25% Coomassie Brilliant Blue R-250 (Sigma, USA) for 1 h and destained in 15% methanol/7.5% acetic acid. Quantitative analysis of these caseinolytic enzymes was performed via an iBright CL750 imaging system (Thermo Fisher Scientific, USA).
Western blot analysis
The proteins were extracted from lung tissues via radioimmunoprecipitation assay buffer containing protease inhibitors (Sigma, St. Louis, MO, USA), resolved via SDS‒polyacrylamide gel electrophoresis, and transferred to polyvinylidene difluoride membranes (Thermo Fisher Scientific, USA). The membranes were incubated for 1 h in blocking buffer (3% BSA in PBS) at room temperature. The membrane was washed three times with PBST (0.1% Tween-20 in phosphate-buffered saline). The following primary antibodies were used: tPA (1:500, American Diagnostica, USA), uPA (1:500, American Diagnostica, USA), COX-2 (1:1000, Santa Cruz, USA), COL6 (1:1000, Abcam, USA), PI3K (1:1000, Abcam, USA), p-PI3K (1:500, Abcam, USA), AKT (1:1000, Abcam, USA) and p-AKT (1:1000, Abcam, USA). β-Actin monoclonal antibody (1:5000, Sigma, St. Louis, MO, USA) was used as a loading control. The membrane was then incubated with a primary antibody and an HRP-conjugated secondary antibody (Jackson ImmunoResearch Laboratories, West Grove, PA) diluted 1:5000 at 37 °C for 1 h. Protein bands were detected via enhanced chemiluminescence (ECL) and visualized with an iBright CL750 imaging system (Thermo Fisher Scientific, USA).
Hematoxylin and eosin staining
Mouse lung slides were sequentially immersed in xylene, 100% ethanol, 95% ethanol, 75% ethanol, 50% ethanol, 25% ethanol, and deionized water for deparaffinization and rehydration. The nuclei were stained with hematoxylin, washed with deionized water, and then immersed in acid alcohol for differentiation. After another wash with deionized water, an eosin solution was used to stain the cytoplasm. Finally, the slides were washed with deionized water and air-dried at room temperature prior to mounting. The mounted slides were examined under a microscope (ZEISS, Axio Imager A2, Heidelberg, Germany). The pulmonary bleeding intensity was quantified via ImageJ software (version 1.54 g).
Pathological analysis
A semi-quantitative assessment of lung tissue inflammation was performed using the Szapiel scoring system, which grades alveolitis severity on a scale of 0–3: score 0: absence of alveolitis. Alveolitis. score 1: mild alveolitis, mononuclear cell infiltration was seen locally and in the near thorax, with an area of less than 20% of the whole lung, and the alveolar structure was more or less normal. Score 2: moderate alveolitis, with the lesion area accounting for 20–50% of the whole lung. Score 3: severe alveolitis and pulmonary fibrosis, with the area of the lesion being greater than 50% of the whole lung, with occasional mononuclear cells and/or hemorrhage in the alveolar lumen causing solid lesions.
Immunohistochemistry
Immunohistochemistry was performed to detect COX-2, tPA, uPA and COL6 proteins in lung sections from BALB/c mice infected with T. canis at 7 days post infection (dpi). In a typical procedure, paraffin-embedded section (10 μm) were deparaffinized, treated with 3% H2O2 in methanol for 10 min to inactivate any endogenous peroxidase, and washed with PBST. The sections were blocked for 1 h in blocking buffer (3% BSA in PBST) and incubated for 1 h with primary antibodies (COX-2, Santa Cruz, USA), tPA (American Diagnostica, USA), uPA (American Diagnostica, USA) and COL6 (Abcam, USA) diluted 1:50 in 1% BSA. Sections incubated with normal serum served as negative controls. The sections were washed three times for 10 min each in PBST and incubated for 1 h with an HRP-conjugated secondary antibody (Jackson ImmunoResearch Laboratories, West Grove, PA, USA) diluted 1:1000 in 1% BSA at 37 °C. The sections were finally incubated with 3,3′-diaminobenzidine (DAB) at room temperature for 3 min for color development. The sections were counterstained with diluted hematoxylin. Mounted slides were examined under a microscope (ZEISS, Axio Imager A2, Heidelberg, Germany).
Statistical analysis
The results from different groups of mice were compared via the nonparametric Kruskal‒Wallis test followed by post hoc testing via Dunn’s multiple comparison of means. All the results are presented as the mean ± standard deviation (SD). p values p values r > 0.8, p r p r > 0.8, p r p r values.






